The lumbar epidural blood patch: A Primer
Images

Headaches secondary to leakage of cerebrospinal fluid (CSF), either related to recent dural puncture or spontaneous leakage, have been well described. These headaches are typically postural in character, can be severe, and may be associated with nausea, vomiting, neck stiffness, and dizziness. The autologous epidural blood patch (EBP) was first shown to be effective in the treatment of these low-pressure headaches in the 1970s.1 Since that time, multiple studies have shown efficacy rates ranging from 70-90% in the treatment of post-dural puncture headaches and 52-87% in the treatment of spontaneous intracranial hypotension.2,3 Currently, an image-guided EBP technique offers a safe and precise approach with the goal of improved efficacy, better patient tolerance, and decreased risk of complication relative to non-image-guided techniques, as well as accurate anatomic localization when a specific target location is required.
Indications
The goal of the epidural blood patch is to treat the symptoms, typically headache, related to cerebrospinal fluid leaks of any cause. Headaches due to low CSF pressure are stereotypically orthostatic in character with exacerbation in the upright position and relative improvement while supine. Recent lumbar puncture (LP) and suspected spontaneous intracranial hypotension (SIH) are the most frequent indications for EBP at our institution.
The reported incidence of post-dural puncture headache (PDPH) is up to 40% and typically occurs within the first few days’ post procedure.4 Conservative treatments include hydration, acetaminophen, caffeine, and limiting activity. While the headache tends to resolve within days if left untreated, there are reported cases of headaches lasting months.1 Several risk factors for PDPH have been described including larger needle size, cutting needle type, atraumatic tap, and not re-styletting the spinal needle prior to withdrawal.2
Similarly, SIH is believed to be caused by a spontaneous or occult CSF leak, most commonly in the spine where the nerve roots exit the meninges.5 In patients clinically suspected to have SIH, MR imaging of the brain may support the diagnosis by demonstrating indirect findings such as diffuse pachymeningeal thickening and enhancement, sagging brainstem, tonsillar descent, and enlargement of the pituitary. Alternatively, dedicated spine imaging (such as heavily T2-weighted fat-saturated MRI or CT myelography) may demonstrate direct evidence of a CSF leak.6 Whether or not a CSF leak is identified in patients with suspected SIH, empiric epidural blood patch has been shown to be effective in alleviating headaches in >50% of patients where no target leak was found.3 The proposed theories as to how the autologous EPB works in treating CSF leaks are twofold: first, it has been hypothesized to patch the rent in the dura, thus providing a potentially permanent seal to prevent further leakage. Second, the mass effect produced in the epidural space on the adjacent thecal sac is felt to result in an overall increase in CSF pressure, which translates intracranially and may explain the immediate relief most patients experience following the procedure.7 As such, even transient response to the procedure may be of diagnostic utility in supporting the suspected diagnosis of SIH when there is doubt.
Patient screening and contraindications
A careful review of the indication, patient history, laboratory values, and imaging is performed to determine the appropriateness of each procedure. The procedure is contraindicated in patients with active infection, coagulopathy, allergy to utilized medications, significant spinal stenosis, and pregnancy. Available spinal imaging should be reviewed prior to the procedure to evaluate for potential obstacles, specifically degenerative changes, spinal stenosis, variant anatomy, and evidence of prior surgery.
Pertinent anatomy
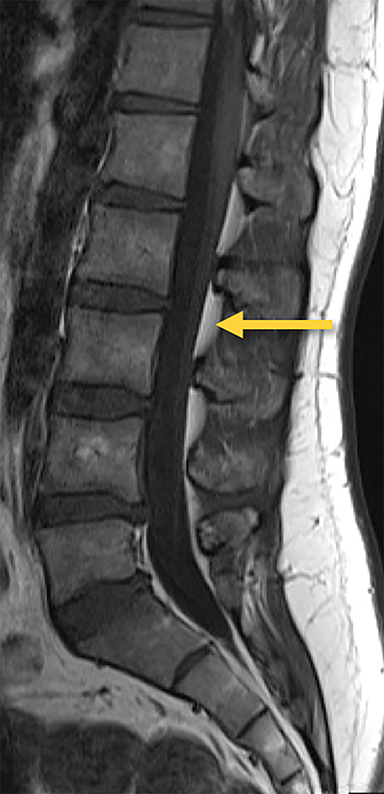
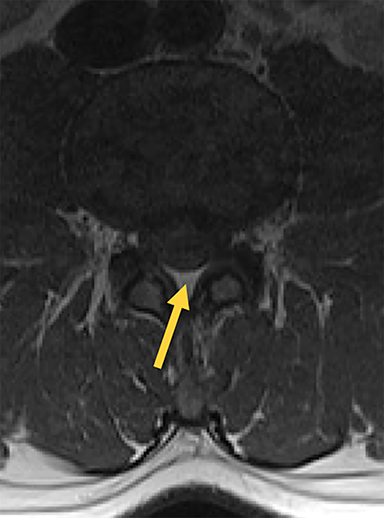
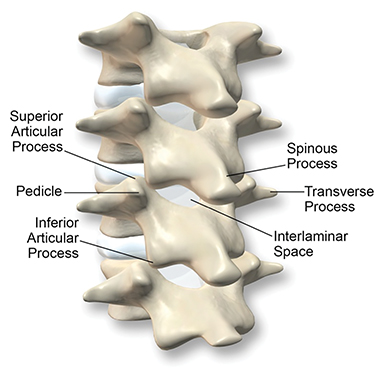
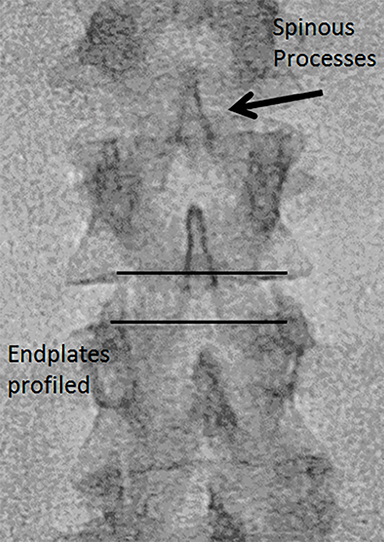
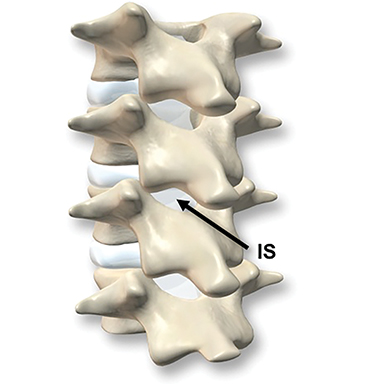
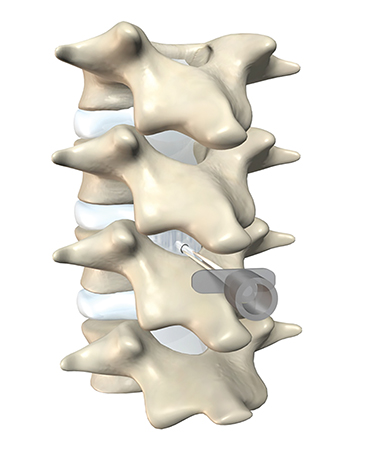
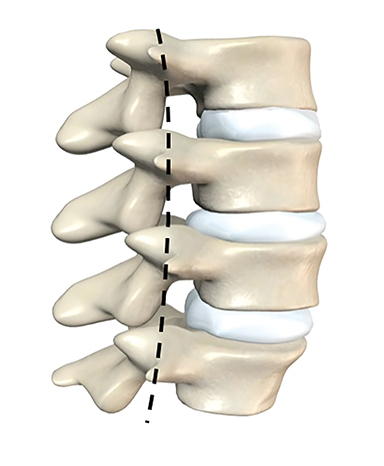
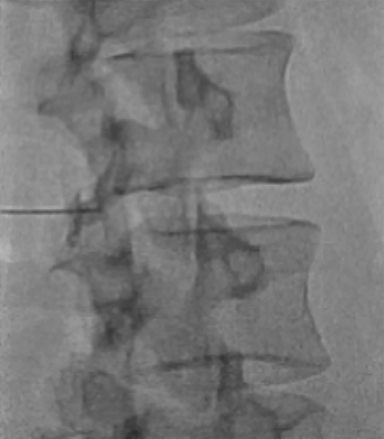
The dorsal epidural space (Figure 1) at the interlaminar level is the target site for injection, just deep to the ligamentum flavum. Relevant anatomic landmarks of the lumbar vertebrae including the posterior element components, interlaminar spaces, spinal canal margins, and disc spaces must all be identified by the operator (Figure 2). In the context of post-dural puncture headache, the level of prior puncture is targeted. In the setting of SIH without an identified leakage site, the L2-3 level is initially targeted assuming no anatomic impediments. Infrequently, we perform thoracic and cervical EBP under CT guidance if there is an identifiable leakage site; the CT-guided technique is beyond the scope of this article.
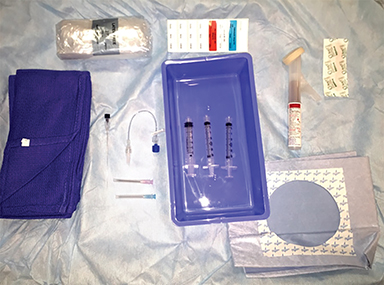
Equipment and table preparation
- Sterilization solution and draping.
- Preservative-free local anesthetic.
- Iodinated contrast approved for intrathecal administration (in case of inadvertent access).
- 22-gauge Quincke tip spinal needle or Tuohy needle (typically 3.5-inch, though 5- and 7- inch lengths may be necessary for larger patients.
- Short, low volume sterile tubing.
- Multiple sterile syringes for collection and administration of autologous blood.
- Nurse to draw autologous blood under sterile conditions.
See Figure 3.
Technique
Patient preparation, localization, and access
The patient should have intravenous access placed on arrival and the ability to withdraw blood easily should be confirmed. The patient is placed prone on the angiography/C-arm table with arms positioned above the head. The IV arm should be extended for access and to prevent kinking of the catheter. The IV site must be prepped and draped for sterility. The IV should be tested again with the patient properly positioned to ensure adequate access, as prone positioning often affects access quality and sometimes necessitates new IV placement.
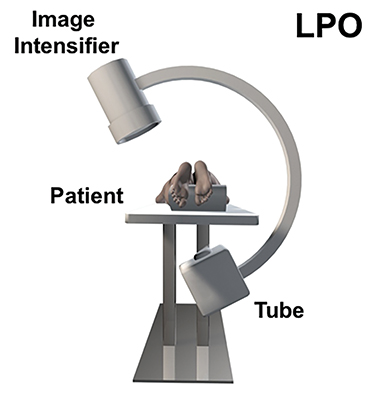
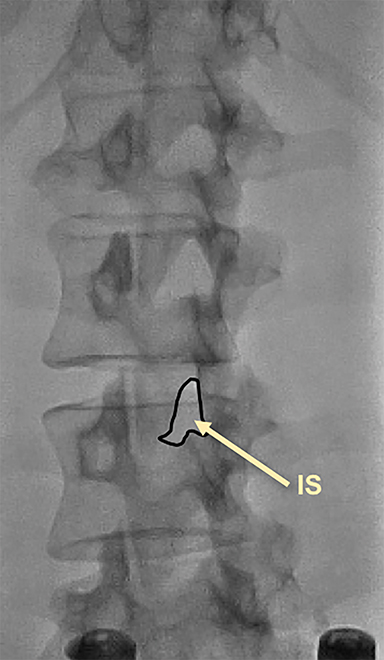
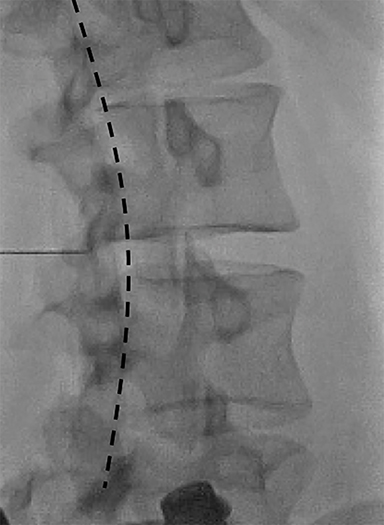
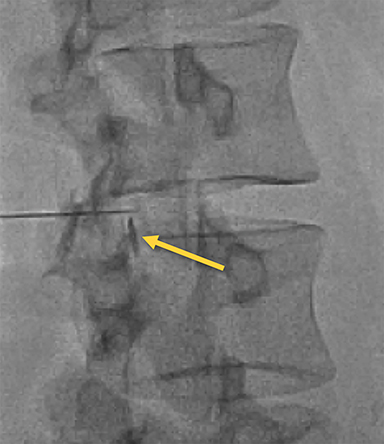
Fluoroscopy is used to target the appropriate interlaminar space. For post-dural puncture patients, the level of prior LP should be targeted. For patients with suspected SIH, we empirically select the L2-L3 level as the initial target site. The L2-L3 level is typically inferior to the conus in cases of accidental dural puncture, usually less affected by impeding degenerative changes, and is a relatively superior starting point given that most cases of SIH are associated with upper spine leaks. Once the target level is identified in the AP view (Figure 4), the image intensifier (II) is then rotated into a left posterior oblique (LPO) position to “open” the interlaminar space (Figure 5). This will be described as the “trajectory” view and corresponds to the course and trajectory that the spinal needle will follow. The tip of a hemostat or metallic pointer is used to mark the center of the interlaminar space on the skin surface. The site can then be prepped, draped, and anesthetized.
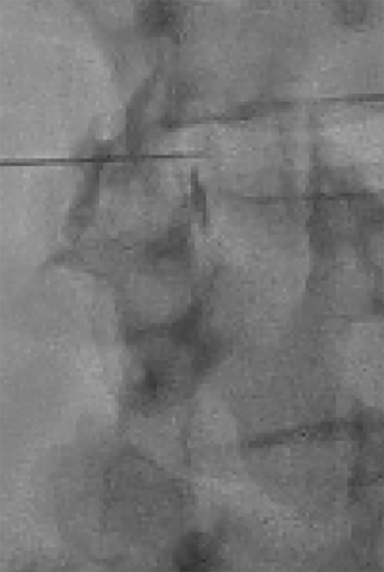
The spinal needle should be oriented “down the barrel” parallel to the X-ray beam and advanced slowly under intermittent fluoroscopy through the subcutaneous tissues into the paraspinal musculature but superficial to the ligamentum flavum (Figure 6). The II should then be rotated to the contralateral oblique to obtain the “depth” view (Figure 7). This view will illicit location of the needle tip and its relationship to the spinolaminar line.
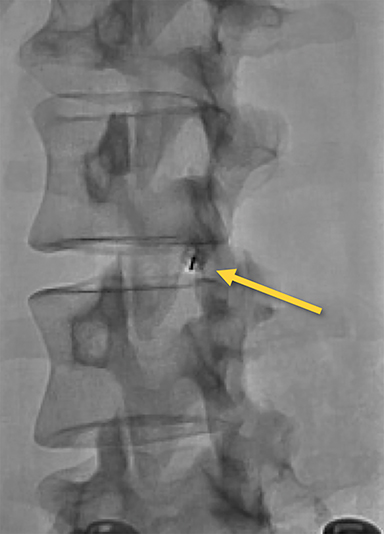
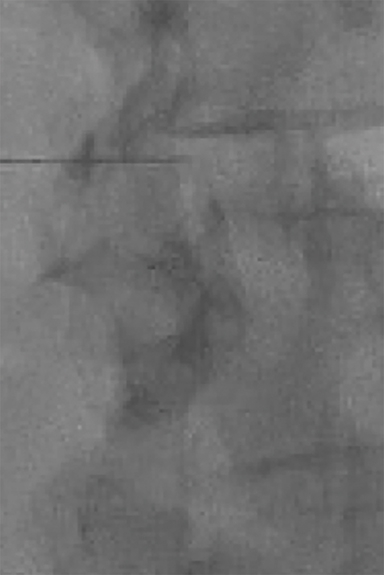
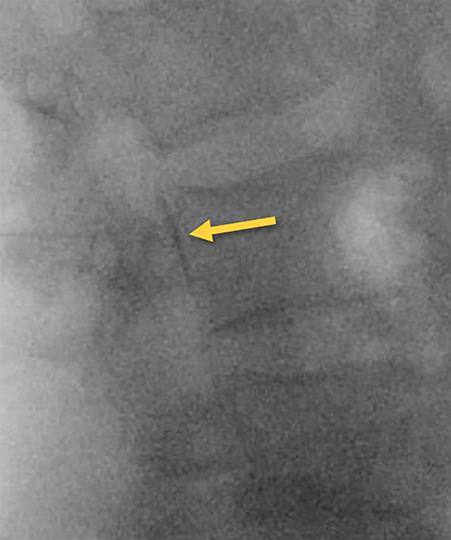
Contrast and primed tubing can then be attached to the spinal needle for a test injection under fluoroscopy to ensure that the needle is superficial to the ligament (Figure 8A). The needle can then be advanced under real-time fluoroscopy with continuous gentle pressure on the plunger. Once the needle has entered the ligament, resistance will noticeably increase and contrast flow will cease. As the needle traverses the ligament into the epidural space, a “loss of resistance” occurs and epidural contrast will then be visualized as a nondispersing oval just deep to the spinolaminar line (Figure 8B). This represents the appropriate location for autologous blood administration.
Injection of autologous blood
Between 3mL and 5 mL aliquots of fresh, sterilely drawn, autologous blood is utilized for epidural injection. Small aliquots and fresh blood are used to avoid clotting. At the start of injection, a quick fluoroscopic image should be obtained to visualize the injection of the residual contrast within the tubing and needle to ensure that the needle has not moved. Intermittent fluoroscopy during the initial aliquot injection can then be performed to show that the contrast has dispersed in the epidural space (Figure 9). The remainder of the injection should be performed slowly to avoid patient discomfort. The operator should assess the patient during administration for any symptoms of local pain/pressure, radiculopathy, or numbness.
For post-dural puncture headaches, we recommend a target of 10mL-15 mL. In patients with SIH, we recommend a goal of 20 mL initially and up to 30 mL on subsequent encounters. However, there is significant variability in the volume of blood tolerated; the final volume is largely determined by the patient’s comfort level. If the patient feels excessive discomfort or pressure, the procedure is terminated and the amount of blood injected is documented.
Troubleshooting
If there is concern for intrathecal needle placement, check for CSF return or look for a myelographic appearance during contrast administration: on the depth view, small intrathecal injections will first fall dependently and outline the anterior thecal sac (Figure 10). Larger injections will outline the nerve roots as in a typical myelogram. If there is quick dispersion of contrast material, then the needle should be retracted into the epidural space and a repeat test injection performed.
If the patient feels significant pain during administration, then slight Trendelenburg positioning may allow some of the blood to move cephalad (particularly in the higher volume SIH patients). This may allow for a slightly greater volume to be administered. In addition, the operator can slow the injection or take short breaks to allow the discomfort to abate.
At times, there may be issues with blood coagulating in the 5 mL syringes prior to injection. If this occurs, the operator can switch to 3 mL sterile syringes for the remainder of the procedure.
Complications
Commonly reported side effects and/or complications include transient paresthesias, transient radicular lower-extremity pain, local back pain, and/or pressure at the site of the injection (which may last up to 2 days), dizziness, vertigo, tinnitus, and rebound intracranial hypertension. A low-grade fever following injection has also been reported. Two rare complications of the epidural blood patch are epidural abscess and nerve compression resulting in transient, or less frequently, permanent neurologic damage.1 Ineffectiveness of the procedure to resolve the headache or to result in only a partial response has also been observed. This is more likely to occur in patients with SIH where no target site was identified. In this subclass of patients, we may repeat the EBP a second and sometimes a third time within a 2-week to 3-week period at higher vertebral body levels. If there is no change in patient’s symptoms after these additional attempts, other etiologies for the headache and alternative therapeutic options should be explored.
Post-procedure care
The patient should be placed on bedrest for at least 1-2 hours (optimally 24 hours post procedure) to allow for coagulation and a dural seal to develop. A slight Trendelenburg position initially may help the injection spread cranially, which may be useful in empiric SIH treatment. Emergency precautions should be given to include fever, worsening focal pain, bruising/bleeding, lower extremity numbness or weakness, or any other signs of cord compression.
Conclusion
The EBP continues to be a commonly requested, effective procedure for the treatment of headache due to CSF leak. We have presented a detailed technique for the standard fluoroscopic-guided approach which has proven to be safe and effective in our patient population.
References
- Brodsky JB. Epidural blood patch – A safe, effective treatment for postlumbar-puncture headaches. West J. Med. 1978;129(1):85-87.
- Turnbull DK, Shepherd DB. Post-dural puncture headache: pathogenesis, prevention and treatment. Br J Anaesth. 2003;91(5):718-729.
- Cho KI, Moon HS, Jeon HJ, et al. Spontaneous intracranial hypotension: efficacy of radiologic targeting versus blind blood patch. Neurology. 2011;76(13):1139-1144.
- Oedit R, van Kooten F, Bakker SL, et al. Efficacy of the epidural blood patch for the treatment of post lumbar puncture headache BLOPP: a randomised, observer-blind, controlled clinical trial [ISRCTN 71598245]. BMC Neurol. 2005;5(1):12.
- Couch JR, Persson J. Treatment of spontaneous intracranial hypotension with epidural blood patch: is a complex approach necessary or better than a simple one? Acta Anaesthesiol Scand. 2012;56(10):1207–1209.
- Schievink WI. Spontaneous spinal cerebrospinal fluid leaks and intracranial hypotension. JAMA. 2006;295(19):2286-2296.
- Safa-Tisseront V, Thormann F, Malassine P, et al. Effectiveness of epidural blood patch in the management of post-dural puncture headache. Anesthesiology. 2001;95(2):334-339.
Citation
B W, V L, D C, D S, E S, W M.The lumbar epidural blood patch: A Primer. Appl Radiol. 2019; (2):25-30.
March 28, 2019