The Benefits of a Macrocyclic, Ionic Contrast Agent: An Expert Forum Summary
Images

Applied Radiology assembled a group of six radiologists experienced in adult and pediatric MR neuroimaging to discuss and debate their views on selection of contrast agents for contrast-enhanced neuro-MRI. This program was sponsored by Guerbet LLC.
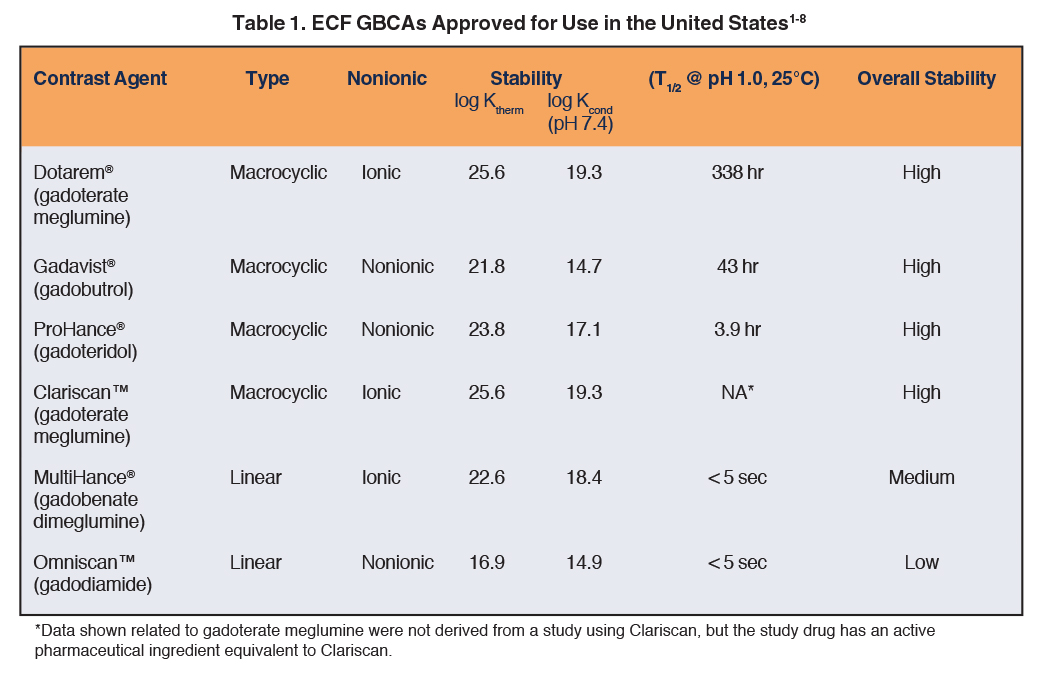
The properties of available ECF GBCAs are provided in Table 1.1-8 GBCAs consist of a Gd ion chelated to a carrier molecule. In terms of structure and stability, GBCAs may be macrocyclic or linear, and ionic or nonionic. In macrocyclic agents, the Gd ion is encircled by a chelate “cage,” and such complexes have been shown to be more stable than GBCAs with linear chelates.7,10,11 In addition, for GBCAs, ionicity also contributes to tighter binding between the Gd and chelate; ie, ionic GBCAs are less likely to dissociate and release the Gd ion.12 These concepts align with the prevailing theory that the most stable Gd-chelate complex would be ionic and have a macrocyclic backbone. In fact, Dotarem® (gadoterate meglumine), the first macrocyclic and ionic GBCA, demonstrates the highest thermodynamic and kinetic stability constants.1-8 (Table 1)
For neuroradiology applications, all the ECF GBCAs are indicated for MRI evaluation of the central nervous system (CNS) in adults and children over the age of 2 years. Dotarem, Gadavist® (gadobutrol), and MultiHance® (gadobenate dimeglumine; at half dose) are also indicated for evaluation of the CNS in term neonates and children 0-2 years old.1,2,5
Relaxivity, defined as the ability of the GBCA to create enhancement, is comparable among all the available GBCAs (with the exception of MultiHance, which is a bit higher), and stable over the range of magnetic fields used in clinical practice (0.2-1.5T).1-8 Another differential property is the formulation: all the ECF GBCAs have a concentration of 0.5 mmol/mL, with the exception of Gadavist, which is twice as concentrated as the others (ie, formulated at 1 mmol/mL), and thus requires half the volume to deliver the same amount of Gd as the other agents.2
GBCA Structure and the Implications for Stability
Measuring GBCA Stability
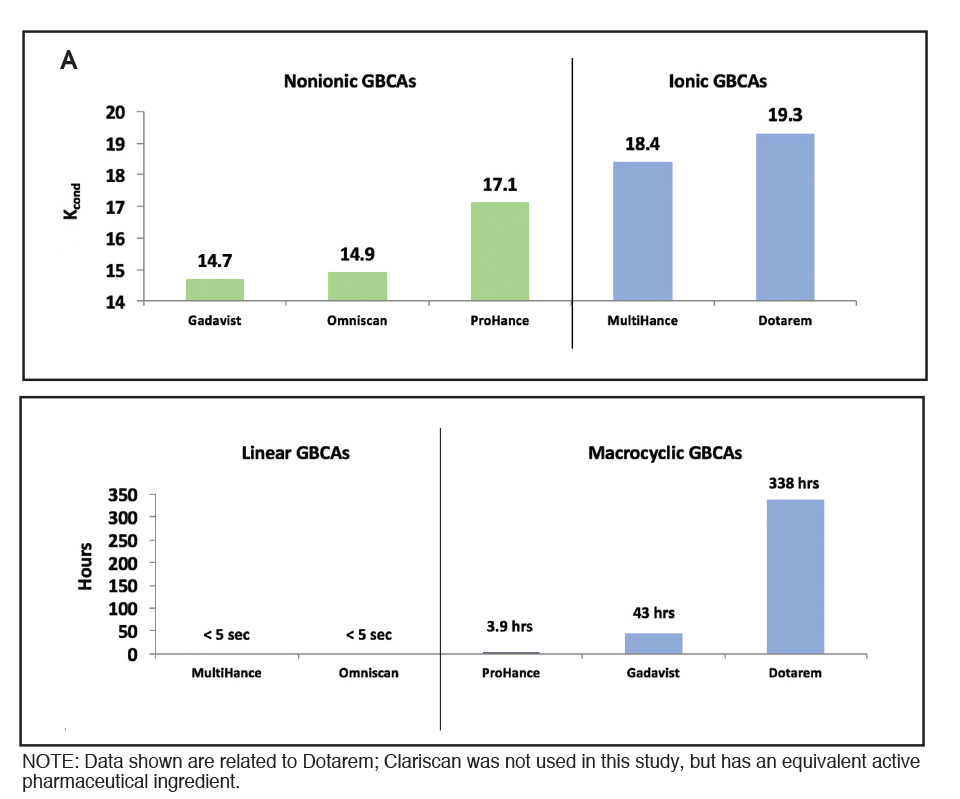
Stability of GBCAs can be assessed by measuring the conditional thermodynamic stability constant (log Kcond, measured at physiologic pH 7.4) and the half-life (T1/2).8,13 Figure 1A shows that the ionic contrast agents Dotarem and MultiHance have higher conditional thermodynamic stability compared with nonionic agents. Note that these constants are measured on a logarithmic scale and, therefore, small differences represent large differences in stability. In addition, the half-life, or T1/2, is substantially different among the GBCAs: the T1/2 of the linear agents is <5 seconds, while the T1/2 is 3.9 hours for ProHance® (gadoteridol), 43 hours for Gadavist, and up to 338 hours for Dotarem (the endpoint of the experiment).8 (Figure 1B) The same relationships were confirmed in a study performed in human plasma in the absence and presence of competitor ions:7 the presence of competitor ions in the plasma hastened the release of Gd, and the macrocyclic agents remained most resistant to transmetallation.
Nephrogenic Systemic Fibrosis (NSF)
It has been known for just over 10 years that the least stable GBCAs increase the risk for NSF among patients with severe renal dysfunction. NSF is a painful, potentially fatal disease in which severe fibrosis is seen in various organs, including the skin, muscle, and internal organs.14-17 In addition to end-stage renal disease, the main risk factors for developing NSF include administration of high and repeated doses of a low-stability GBCA.14,15 As NSF does not occur in the majority of patients with end-stage renal disease administered high/multiple doses of a low-stability GBCA, other unique and less well-understood physiologic or clinical factors are presumed to contribute to NSF incidence.
Based on the number of unconfounded (ie, single agent) cases of NSF seen following administration of each GBCA, we know the risk of NSF is: highest with linear, nonionic agents; intermediate with linear, ionic agents; and lowest with macrocyclic agents. (Figure 2) Although factors such as market share and usage patterns undoubtedly contribute to some skew in the data,18-20 it is noteworthy that among the macrocyclics, no unconfounded cases of NSF have been associated with the highly stable macrocyclic and ionic GBCA Dotarem, despite extensive use in the “pre-NSF era” and after over 100 million doses have been administered worldwide. (Guerbet; data on file)
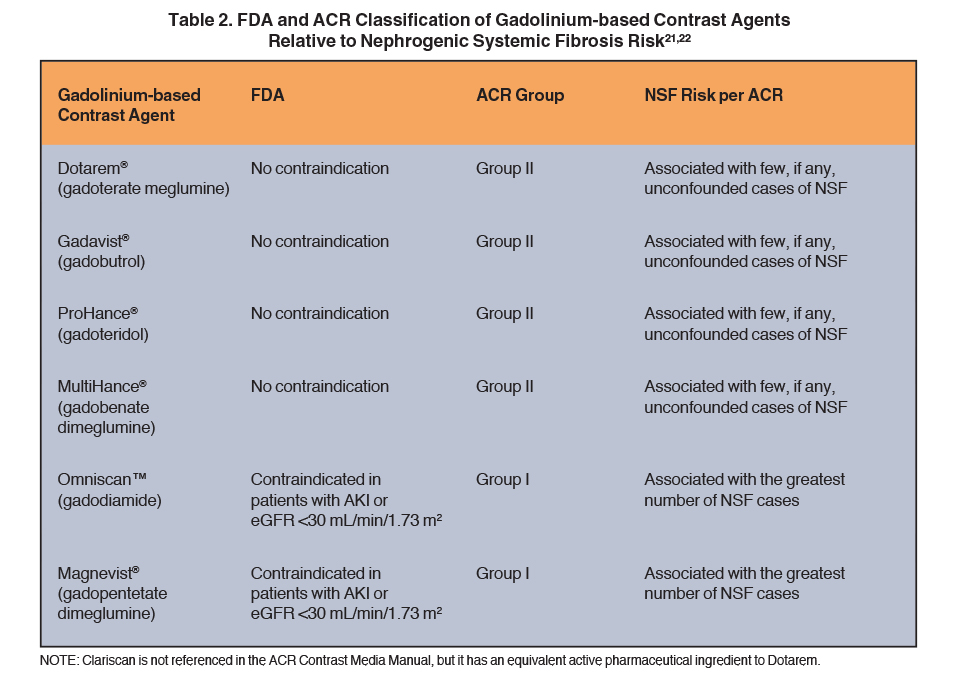
Since the association of GBCA use with NSF was reported, various government regulatory bodies and professional societies, including the U.S. Food and Drug Administration (FDA) and the American College of Radiology (ACR), have issued guidelines classifying the various GBCAs according to their inherent NSF risk, as well as recommendations for GBCA use in patients with various degrees of renal dysfunction.21,22 The recommendation by both groups is to avoid (contraindicated by the FDA) low-stability agents in patients with acute kidney injury or severe renal dysfunction (eGFR <30 mL/ min/1.73 m2). (Table 2) These guidelines, together with the ensuing changes in clinical practice patterns around GBCA administration, have resulted in a dramatic decrease in the number of newly reported NSF cases.
Administration of GBCAs in Patients with Known Renal Dysfunction
Patients with end-stage renal disease are at greatest risk for NSF. However, evidence shows that the risk of NSF is reduced when Group II agents are used. For example, in the RESCUE Study, Deray et al compared the safety of contrast-enhanced MRI with Dotarem to unenhanced MRI in 114 patients with chronic kidney disease, and found no difference in the incidence of NSF in the two patient groups.23 In the prospective ProFINEST Study, Amet et al assessed the incidence of NSF in long-term dialysis patients that either received or did not receive a GBCA.24 Of the 571 included patients, almost all (88.9%) received Dotarem and, after a 4-month follow-up period, no cases of NSF were detected in these patients, supporting the use of a stable, macrocyclic agent when contrast-enhanced MRI is indicated, in even the most renal compromised.
The general consensus among radiologists is that if a patient is on hemodialysis (but not peritoneal dialysis, as there is no evidence that it is protective), the contrast-enhanced exam should be scheduled so the next dialysis session immediately follows the imaging exam.22 The use of dialysis to minimize the risk of NSF has not been confirmed in any randomized, controlled trials, and it is generally agreed that initiating hemodialysis prior to or after GBCA administration in a patient not currently receiving hemodialysis is not warranted.
Neuroimaging in the Adult Population
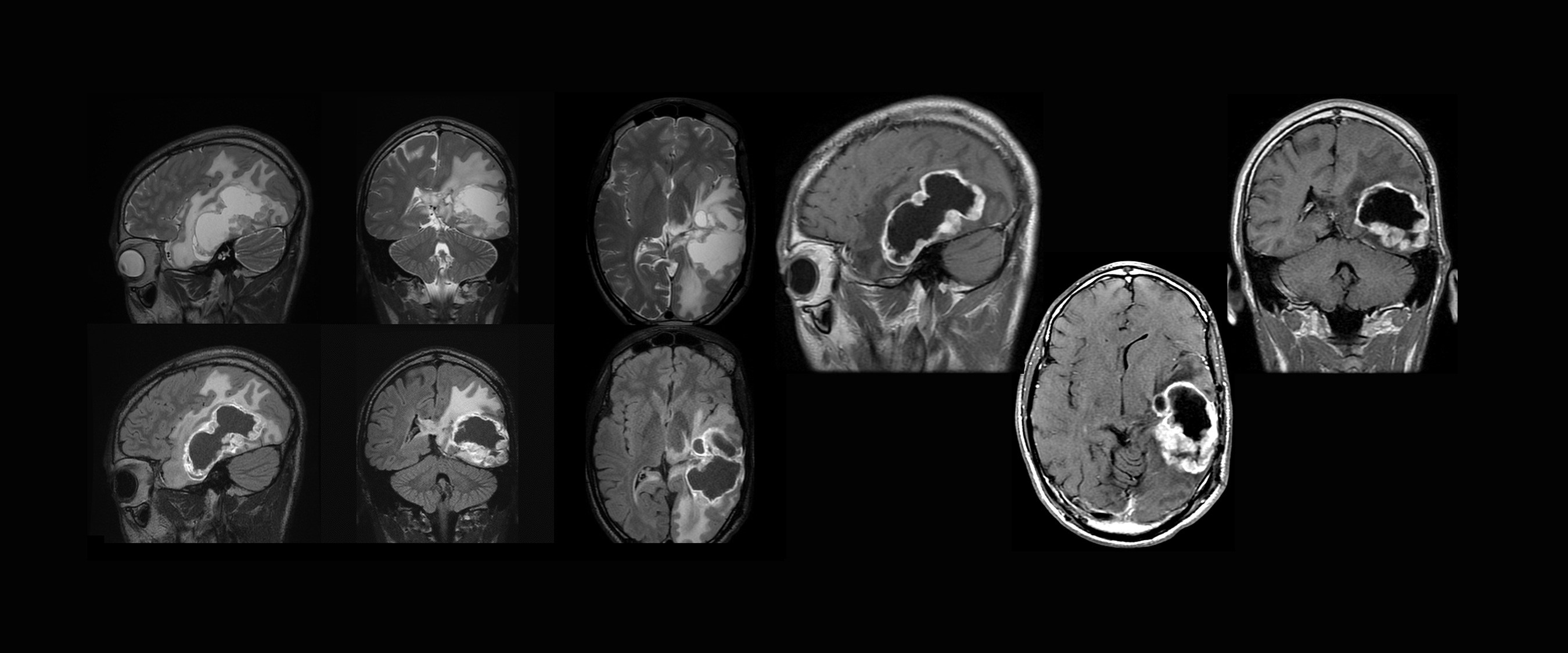
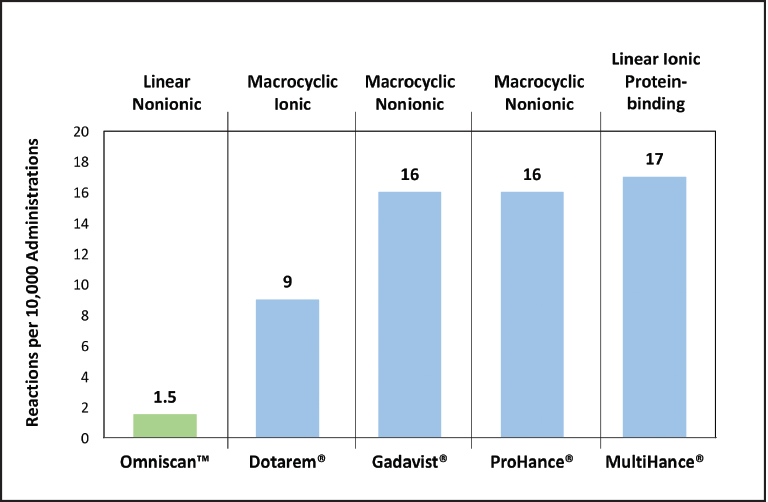
Contrast-enhanced MRI in patients with malignant brain tumors provides information critical to diagnosis, treatment-related decisions, and treatment follow-up. (Figure 3) Historically, MRI contrast agents have been viewed as having a favorable safety profile, with low adverse reaction rates.25,26 (Figure 4) However, when using a GBCA for imaging adult patients with neurologic disease, several patient populations are at increased risk of clinical sequelae, particularly when less stable GBCAs are used.
Even in low-risk patients, it is often beneficial to evaluate clinical situations when it might be possible to avoid contrast use — eg, where the contrast may not actually contribute to the diagnosis or surveillance. MR contrast may be helpful for diagnosis and treatment planning, but consideration should be given to its value in assessing interval changes in lesion size. The ability to routinely surveil primary brain lesions without contrast is somewhat more controversial. However, if the primary brain lesion does not enhance, contrast may be less likely to contribute important information during surveillance. Imaging the blood-brain barrier with contrast is also of limited value in distinguishing between treatment-related findings and recurrent tumor. In such cases, alternative methods, such as arterial spin labeling, could be helpful in lesion interrogation and characterization.
Contrast-enhanced Neuroimaging: A Pediatric Perspective
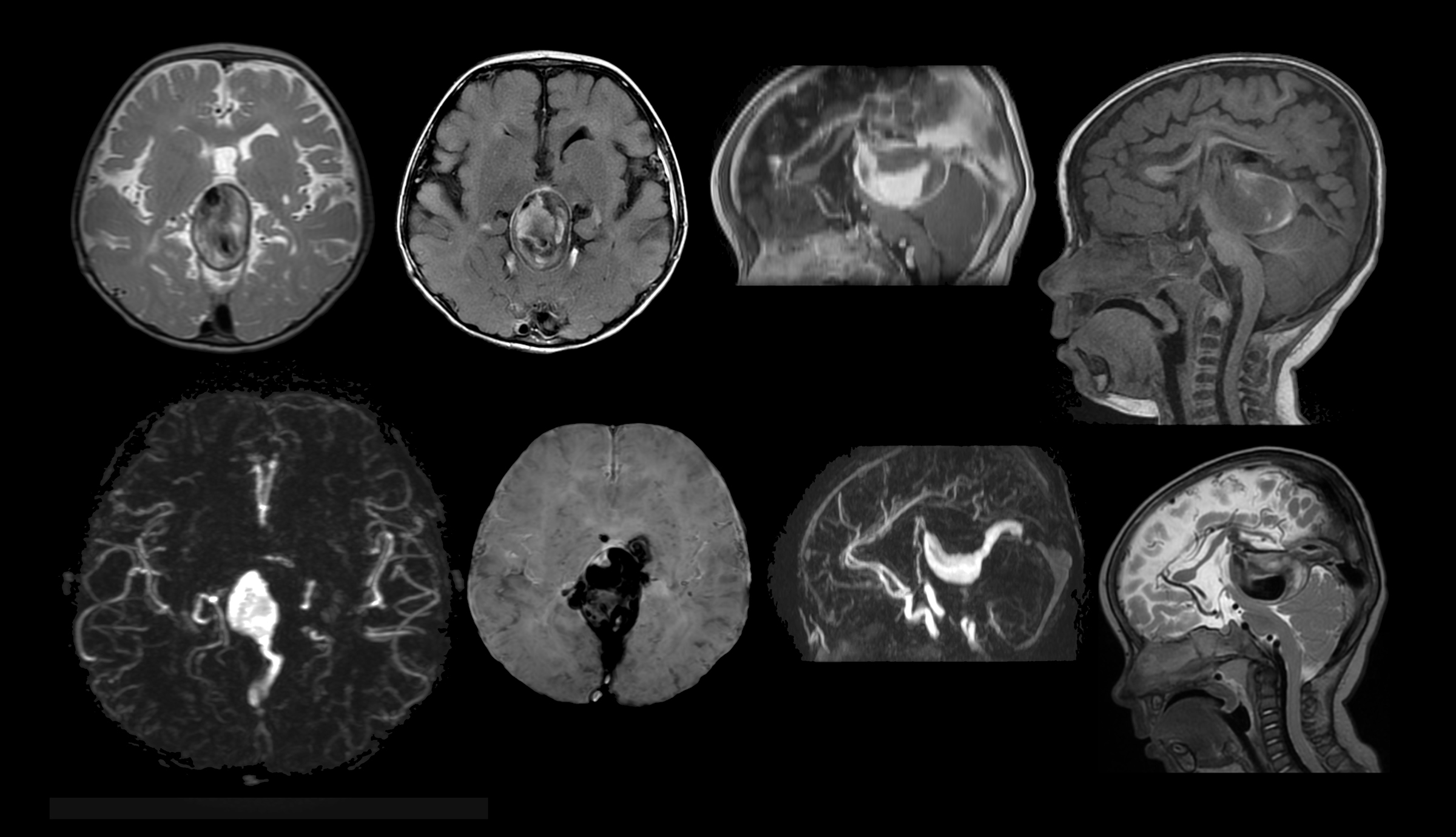
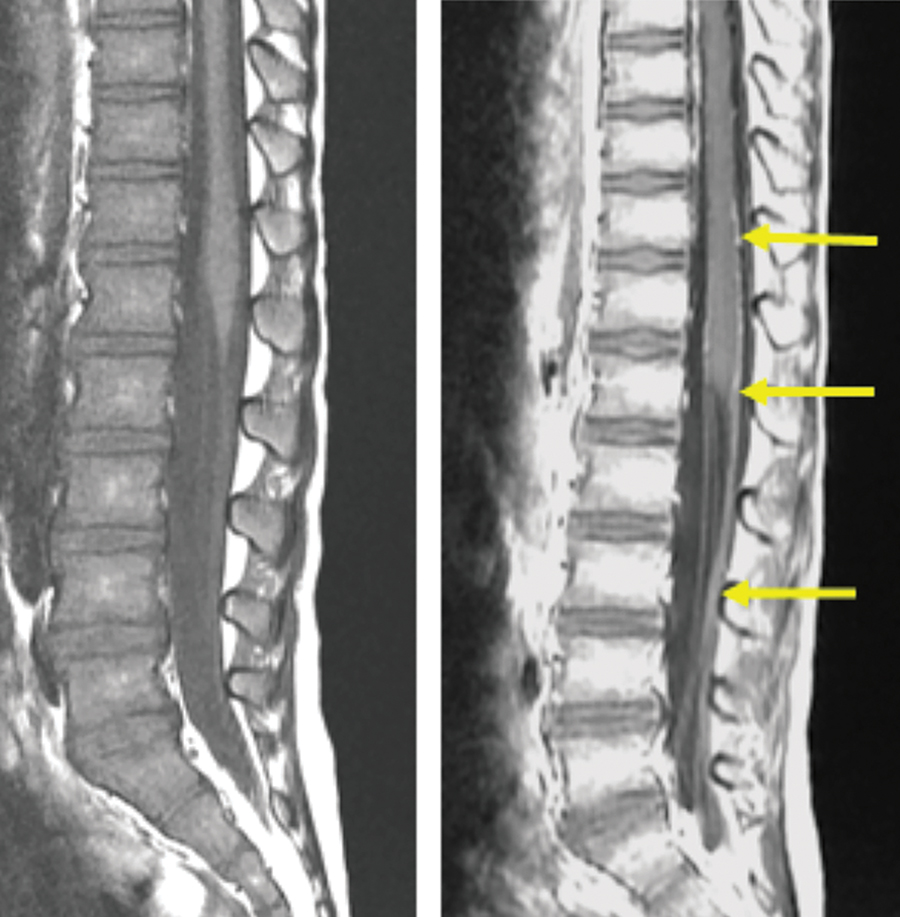
Contrast-enhanced imaging is indicated in children for a wide variety of situations, including evaluation of intracranial, spine, and head and neck pathologies. (Figures 5, 6) In fact, in many children, asymptomatic recurrence of brain tumors is detectable only with postcontrast imaging, justifying contrast-enhanced MRI for surveillance neuroimaging studies.27,28 However, when deciding whether to administer a GBCA, concerns for pediatric patients are somewhat different from those for adults. Surveillance imaging in children can potentially lead to a much larger cumulative burden of Gd exposure over the course of a lifetime. In addition, other challenges to contrast administration in children include the difficulty in administering small doses accurately (often <1 mL), and the desire to avoid initiating an IV or using sedation.
Finally, an important consideration when deciding whether to administer contrast in a child is the uncertain value of measuring eGFR in the very young. In the newborn, the GFR is only approximately 40 mL/min/1.73 m2, and it doesn’t reach adult levels of 100–125 mL/min/1.73 m2 until the child is about 2 years of age.29 As a result, eGFR thresholds typically used as guidelines for GBCA use in adults are not appropriate for pediatric patients. Rather, assuming the child is not known to have renal dysfunction, patient age is a better indicator of renal function than eGFR and often, waiting even 1 week before administering contrast can allow for substantial maturation in renal function. Although there are very few, if any, documented cases of NSF in children <2 years of age, their immature renal function potentially does, in theory, represent an NSF risk.30 Therefore, it is important, particularly in children, (1) to administer a GBCA only when clinically necessary, (2) to document the number of doses administered to a patient, (3) to consider temporal spacing between exams to permit full clearance, and (4) when indicated, to use a high-stability agent.
Recent practice-pattern surveys confirm a trend among pediatric radiologists to switch from linear agents to macrocyclics.31 Specifically, in a survey of over 5,000 physicians in various pediatric specialties conducted between January 2016 and March 2016, (overall response rate 690/5,390 [13%]), 80% of responding pediatric hospitals were found to use a macrocyclic GBCA. Of those pediatric radiology departments that switched in the last year (15/26 [58%]), most (8/15 [53%] switched to Dotarem, and the most common reasons for switching included brain gadolinium deposition concerns (11/15 [73%]), improved safety profile (7/15 [47%]), and improved stability (5/15 [33%]). Previously, GBCAs were used off-label in children <2 years of age; however, Dotarem, Gadavist, and MultiHance are all currently FDA-approved for use in neonates and young children.1,2,5
Administrative Considerations
When selecting a contrast agent, it is prudent practice that physicians with knowledge and experience be part of the decision-making team.
Upon switching agents, care must be taken to ensure a smooth transition, since increases in reports of adverse reactions shortly after a transition can occur due to staff on the “look-out” for new reactions (Weber effect), a phenomenon that has been recognized to occur after a GBCA switch.32,33 Educating nurses and technologists regarding the history of (ie, number of administrations worldwide) and safety profile of the newer agent can often ensure and boost comfort. An updated and current department-wide contrast agent policy that provides clear guidelines on which agent to use in which patients, when to screen for renal function, when to pretreat, etc., is essential.
Conclusions
In summary, the currently available ECF GBCAs have varying structures that impact their stability, and thus their propensity to release Gd from the chelate in vivo. Therefore, when a risk-benefit analysis supports the use of contrast with MRI, stability is a very important consideration when selecting from among the currently available GBCAs.
References
- Dotarem® (gadoterate meglumine) [prescribing information]. Princeton, NJ: Guerbet LLC; October, 2019.
- Gadavist® (gadobutrol) injection [prescribing information]. Wayne, NJ; Bayer HealthCare Pharmaceuticals; April 2016.
- ProHance® (Gadoteridol) Injection, [prescribing information]. Princeton, NJ: Bracco Diagnostics Inc.; November 2013.
- Clariscan™ (gadoterate meglumine) [prescribing information]. Marlborough, MA: GE Healthcare Inc.; November, 2019.
- MultiHance® (gadobenate dimeglumine) injection [prescribing information]. Princeton, NJ; Bracco Diagnostics Inc.; July 2013.
- Omniscan™ (gadodiamide) injection [prescribing information]. Princeton, NJ: GE Healthcare; August 2013.
- Frenzel T, Lengsfeld P, Schirmer H, Hütter J, Weinmann HJ. Stability of gadolinium-based magnetic resonance imaging contrast agents in human serum at 37 degrees C. Invest Radiol. 2008; 43:817-828.
- Port M, Idée JM, Medina C, Robic C, Sabatou M, Corot C. Efficiency, thermodynamic and kinetic stability of marketed gadolinium chelates and their possible clinical consequences: a critical review. Biometals. 2008; 21:469-490.
- Rogosnitzky M, Branch S. Gadolinium-based contrast agent toxicity: a review of known and proposed mechanisms. Biometals. 2016; 29:365-376.
- Puttagunta NR, Gibby WA, Smith GT. Human in vivo comparative study of zinc and copper transmetallation after administration of magnetic resonance imaging contrast agents. Invest Radiol. 1996;31: 739-742.
- White GW, Gibby WA, Tweedle MF. Comparison of Gd (DTPA-BMA) (Omniscan) versus Gd(HP-DO3A) (ProHance) relative to gadolinium retention in human bone tissue by inductively coupled plasma mass spectroscopy. Invest Radiol. 2006;41: 272-278.
- Morcos SK. Nephrogenic systemic fibrosis following the administration of extracellular gadolinium based contrast agents: is the stability of the contrast agent molecule an important factor in the pathogenesis of this condition? Br J Radiol. 2007;80: 73-76. Erratum in: Br J Radiol. 2007; 80:586.
- Idée JM, Port M, Robic C, Medina C, Sabatou M, Corot C. Role of thermodynamic and kinetic parameters in gadolinium chelate stability. J Magn Reson Imaging. 2009; 30:1249-1258.
- Kanal E, Barkovich AJ, Bell C, et al; ACR Blue Ribbon Panel on MR Safety. ACR guidance document for safe MR practices: 2007. AJR Am J Roentgenol. 2007; 188:1447-1474.
- Thomsen HS, Marckmann P, Logager VB. Nephrogenic systemic fibrosis (NSF): a late adverse reaction to some of the gadolinium based contrast agents. Cancer Imaging. 2007; 7:130-137.
- Perazella MA. Nephrogenic systemic fibrosis, kidney disease, and gadolinium: is there a link? Clin J Am Soc Nephrol. 2007; 2:200-202.
- Aime S, Caravan P. Biodistribution of gadolinium-based contrast agents, including gadolinium deposition. J Magn Reson Imaging. 2009;30: 1259-1267.
- Edwards BJ, Laumann AE, Nardone B, et al. Advancing pharmacovigilance through academic-legal collaboration: The case of gadolinium-based contrast agents and nephrogenic systemic fibrosis— Research on Adverse Drug Events and Reports (RADAR) report. Br J Radiol. 2014; 87:20140307.
- Food and Drug Administration (FDA). Advisory Committee Briefing Document, Dotarem® (gadoterate meglumine). Available at: https://webcache.googleus-ercontent.com/search?q=cache:pNy3Rdj4lJwJ:https:// www.fda.gov/downloads/AdvisoryCommittees/UCM339378.pdf+&cd=1&hl=en&ct=clnk&gl=us. February, 2013.
- Food and Drug Administration (FDA). Center for Drug Evaluation and Research (CDER). Summary Review. Gadovist. Available at: https://www. accessdata.fda.gov/drugsatfda_docs/nda/2011/ 201277Orig1s000SumR.pdf. March, 2011.
- Food and Drug Administration (FDA). FDA Drug Safety Communication: New warnings for using gadolinium-based contrast agents in patients with kidney dysfunction. Available at: https://www.fda.gov/Drugs/ DrugSafety/ucm223966.htm. December, 2010.
- American College of Radiology (ACR) Committee on Drugs and Contrast Media. ACR Manual on Contrast Media, 2020. Available at: https://www.acr.org/Clinical-Resources/Contrast-Manual.
- Deray G, Rouviere O, Bacigalupo L, et al Safety of meglumine gadoterate (Gd-DOTA)-enhanced MRI compared to unenhanced MRI in patients with chronic kidney disease (RESCUE study). Eur Radiol. 2013; 23:1250-1259.
- Amet S, Launay-Vacher V, Clément O, et al Incidence of nephrogenic systemic fibrosis in patients undergoing dialysis after contrast-enhanced magnetic resonance imaging with gadolinium-based contrast agents: the Prospective Fibrose Nephrogénique Systémique study. Invest Radiol. 2014; 49:109-115.
- Beckett KR, Moriarity AK, Langer JM. Safe Use of Contrast Media: What the Radiologist Needs to Know. Radiographics. 2015; 35:1738-1750.
- Behzadi AH, Zhao Y, Farooq Z, Prince MR. Immediate Allergic Reactions to Gadolinium-based Contrast Agents: A Systematic Review and Meta-Analysis. Radiology. 2018; 286:471-482.
- Korones DN, Butterfield R, Meyers SP, Constine LS. The role of surveillance magnetic resonance imaging (MRI) scanning in detecting recurrent brain tumors in asymptomatic children. J Neurooncol. 2001; 53:33-38.
- Udaka YT, Yeh-Nayre LA, Amene CS, VandenBerg SR, Levy ML, Crawford JR. Recurrent pediatric central nervous system low-grade gliomas: the role of surveillance neuroimaging in asymptomatic children. J Neurosurg Pediatr. 2013; 11: 119-126.
- Sulemanji M, Vakili K. Neonatal renal physiology. Semin Pediatr Surg. 2013; 22:195-198.
- Thomsen HS, Morcos SK, Almén T, et al. Nephrogenic systemic fibrosis and gadolinium-based contrast media: updated ESUR Contrast Medium Safety Committee guidelines. Eur Radiol. 2013;23:307-318.
- Mithal LB, Patel PS, Mithal D, Palac HL, Rozenfeld MN. Use of gadolinium-based magnetic resonance imaging contrast agents and awareness of brain gadolinium deposition among pediatric providers in North America. Pediatr Radiol. 2017; 47:657-664.
- Abujudeh HH, Kosaraju VK, Kaewlai R. Acute adverse reactions to gadopentetate dimeglumine and gadobenate dimeglumine: experience with 32,659 injections. AJR Am J Roentgenol. 2010; 194:430-434.
- Davenport MS, Dillman JR, Cohan RH, et al. Effect of abrupt substitution of gadobenate dimeglumine for gadopentetate dimeglumine on rate of allergic-like reactions. Radiology. 2013; 266:773-782.
Citation
The Benefits of a Macrocyclic, Ionic Contrast Agent: An Expert Forum Summary. Appl Radiol.
November 5, 2020