PET and PET-CT for oncology applications in the abdomen and pelvis: Update and future directions in the age of molecular medicin
Images
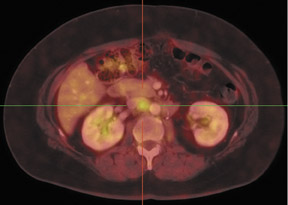
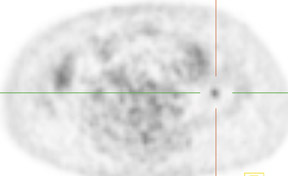

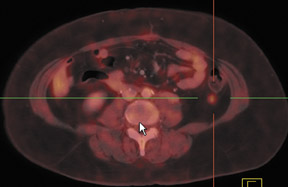

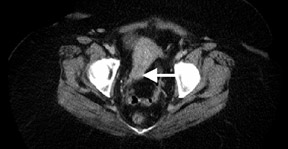





Dr. Franc is a member of Radiological Associates of Sacramento, Sacramento, CA, and is an Assistant Clinical Professor, Department of Radiology, University of California, San Francisco, CA.
Over the past several years, the significant utility of positron emission tomography (PET) in guiding the management of many cancersof the abdomen and pelvis has become apparent. PET has evolved as the leading molecular imaging modality in humans and is usedroutinely in the staging, restaging, and monitoring of many cancers throughout the body. When combined with computed tomog-raphy(CT) via software- or hardware-based fusion, PET/CT provides the anatomical localization of functional data from PET and the rapidacquisition of patient-specific attenuation correction data from CT. PET/CT peformed with intravenous contrast optimized for the specific organ of interest can enhance the value of the imaging study.1
The vast majority of clinical PET studies in the United States are performedfollowing the administration of 2-[fluorine-18 (18 F)]fluoro-2-deoxyglucose (18 F-FDG), as it is the most widely available PET radiopharmaceutical and is approved by the U.S.Food and Drug Administration (FDA).18 F-FDG is taken up into cells via glucose transporters and trapped after phosphorylation, thereby concentrating it in highly metabolically active tissues.18 F-FDG is not always the optimal PET imaging agent for cancers of the abdomen andpelvis. For example, the sensitivity of PET with18 F-FDG (FDG-PET) for detection of lymph node metastases from prostate cancer is notoriously low.2 Several other radio pharm aceuticals have been developed to characterize aspects of tumor metabolism apart from glycolysis, offering an alternative to18 F-FDG in the research setting. Unfortunately, many of these tracers are not widely available.
This review focuses on the application of PET and PET/CT with18 F-FDG in abdominal and pelvic cancers. In particular, it discussesthe implementation of PET in the diagnosis, staging, treatment planning, detection of recurrence, and monitoring of therapy. The role ofPET in guiding the management of co-lorectal cancer patients is well established, but significant data are being published on the use ofPET in other cancers of the abdomen and pelvis, particularly ovarian and cervical cancer.3 PET/CT has shown utility in identifying peritoneal metastases of several types of abdominal and pelvic tumors.4 In many other cancers of the abdomen, such as gastric carcinoma orpancreatic cancer, PET may be helpful as an adjunctive diagnostic test but is unlikely to play a central primary role in management deci-sions.5,6 Incidental uptake of FDG in the abdomen on PET studies performed for other indications often represents a clinically significantfinding but is not discussed in the present review.7 The integration of PET and PET/CT into oncology management algorithms has started,but the full potential of PET and PET/CT of abdominal and pelvic cancers has yet to be realized.
Diagnosing tumors of the abdomen and pelvis with PET and PET/CT
In contrast to the case of the solitary pulmonary nodule in thoracic imaging, only recently has PET found potential applications inassisting in the diagnosis of tumors of the abdomen.
Cancers of the colon and rectum
It has been recognized for several years that both benign and malignant lesions of the colon are metabolically active and that large adenomas of the colon may be identified incidentally on whole-body PET.8,9 Some studies that compared findings on FDG-PET directly toendoscopy found a threshold of detection for polyps of 1 cm on FDG-PET.10 However, at least one other study has identified focal uptake onPET that corresponds to lesions even smaller than 1 cm in size and has reported a greater sensitivity for lesions with a higher grade of dyspla-sia.11
CT colonoscopy (virtual colon oscopy) holds promise for making widespread screening for colorectal cancer more acceptable to patients andapproaches the detection capabilities of colonoscopy.12,13 The integration of data from PET into virtual colon oscopy, facilitated by PET/CT, mayhave the potential to allow in vivo characterization of lesions identified on CT colon oscopy and may even add to the sensitivity of identifyingpolyps and cancers, although results from 2 small initial studies did not support either of these hypotheses.14,15 Despite this, there is considerableexcitement in the imaging community regarding the potential of PET/ CT colonography, particularly in patients who are unable to undergo complete colonoscopy. The potential role for PET/ CT in staging colorectal cancers will be discussed below.
Ovarian cancer
While early studies on the use of PET for the characterization of pelvic masses provided mixed results and did not support a clear role for PETfor this indication,17-21 a recent study of pelvic tumors in 97 patients classified as high risk (based on CA-125 level, the results of an ultrasoundexamination, and menopausal status) has provided new evidence of the ability of PET/CT to accurately characterize these tumors. Although thelevel of18 F-FDG uptake in malignant versus benign tumors was not explicitly defined, the authors found a 100% sensitivity and a 92.5% (37 of40) specificity for the classification of tumors as malignant, and most of these cancers were ovarian carcinomas. The authors suggested thatPET/CT may become the modality of choice to characterize pelvic tumors seen on ultrasound, but more explicit guidelines defining malignancyare needed.22
Other cancers
Despite early encouraging results, it does not appear that PET is useful in differentiating between malignant and benign cystic lesionsof the pancreas.23 For the most part, the use of PET and PET/CT in the diagnosis of other cancers of the abdomen and pelvis remains on acase-by-case basis.
Staging and determination of disease extent
Cancers of the colon and rectum
While patients often receive treatment for colorectal cancer prior to receiving complete evaluation for disease extent, PET can be useful inpreoperative staging of colorectal patients and has been shown to lead to modification of the therapeutic approach in 50% of patients with unresectable disease, including identification of previously unknown disease, altered staging, or changed surgical plan.24 Although, most often,PET/CT appears to influence staging and management in the correct direction, there are a minority of cases in which suspicious areas of uptakein the liver or lymph node regions on PET correspond to benign findings at surgery, potentially exposing the patient to a greater extent of surgery than necessary.25 Other studies have found less impact of staging PET or PET/CT on the management of colorectal cancer, with no adverseimpact on patient outcome.26 Overall, colorectal cancers with low FDG uptake portend a better prognosis, despite which therapy the patientreceives.27 The integration of PET data into CT colonography for staging cancers of the colon has provided impact on both tumor (T) and nodal(N) components of the TNM staging system to varying degrees, depending on the specific study and nodal size staging.28
Bipat et al29 conducted a meta-analysis in which FDG-PET was shown to have the highest sensitivity (94.6%) and overall accuracy of tested imaging modalities in the identification of patients with co-lorectal metastases to the liver, but it was not significantly more accurate thanCT or magnetic resonance imaging (MRI) when analyzed on a lesion-by-lesion basis. Through more accurate identification of the cohort ofpatients who could benefit from hepatic resection and better delineation of the extent of that resection, PET can provide a significant impacton survival rate.30 Likewise, PET impacts treatment decisions in locally ablative therapy for metastases to the liver by defining diffuse hepatic disease or extrahepatic disease.31 When PET/CT is compared with CT alone for the evaluation of patients with colorectal cancer for metastatic liver disease, these modalities have similar sensitivities for the detection of intrahepatic metastases. However, whereas extrahepatic diseasewas missed on CT in one third of the cases (sensitivity 64%), PET/CT failed to detect extrahepatic lesions in only 11% of the cases (sensitivity 89%).32 In one of the first large studies comparing PET, in-line PET/CT, and software-fused PET and CT, Kim and colleagues33 found thatPET/CT was significantly more accurate in staging colorectal cancers than PET alone. They also reported that hardware-fused PET/CT provided less distance between key landmarks in the anatomic and functional images than did software-fused PET/CT and was not susceptible tooverall failure. However, when in-line PET/CT is not available, software fusion techniques (including a patient positioning device used in bothimage acquisitions) appear to be preferable to side-by-side reading of PET and CT.34 Despite the fact that both CT and PET missed the majority of small (<1 cm) hepatic metastases from co-lorectal cancer that were detected at surgery using intraoperative ultrasound in a study byWiering and colleagues,35 <7% of the resection plans were altered by the intraoperative findings. This suggests that the combination of PETand CT imaging data can provide the basis for the vast majority of surgical plans for resection of hepatic metastases.
Cervical cancer
Cervical cancer is staged clinically using the International Federation of Obstetrics and Gynecology (FIGO) classification. Clinical stagingfor cervical cancer may understage the disease by failing to identify nodal disease, particularly para-aortic nodal metastases. In patients withFDG uptake indicating metastatic nodal disease, the 20-month disease-free survival has been found to be significantly poorer. Nodal FDG uptakeon pretreatment scanning was shown to predict disease recurrence and was hypothesized to be an important consideration in determining theinitial therapeutic management of these patients.36 Indeed, an initial small retrospective study reported a high accuracy of FDG-PET in distinguishing local from distant metastatic disease in both staging and restaging patients.37 However, when PET's ability to detect pelvic and para-aortic lymph node metastases preoperatively in FIGO IA2 and IIA patients was studied, its specificity was high (>90%) but its sensitivity wasonly 53% for pelvic and 25% for para-aortic lymph node metastases.38 In a study of patients with a broader range of stages (FIGO IB to IVA),region-specific nodal findings on PET showed a sensitivity of only 38% and a positive predictive value of 56%. PET had a sensitivity of 65%for nodal lesions >1 cm in diameter. The authors concluded that PET could not replace surgical staging but could still be useful for detectingmetastases in patients who could not undergo surgery because of other medical conditions.39 Initial findings of Chou et al40 in a study of earlystage (IA2 to IIA), nonbulky cervical cancers with no evidence of lymph node metastases on MRI found that PET had very poor sensitivity forthe detection of pelvic lymph node metastases, supporting the idea that PET alone may not contribute to staging of early cervical cancers.
PET/CT has demonstrated somewhat better results in patients with all stages of cervical cancer. Sironi et al41 reported that PET/CT hasimproved the characterization of lymph nodes <1 cm in diameter. In a group of stage IA or IB patients with 1081 histopathologically characterized lymph nodes, 18 nodes were found to be positive for malignancy, yielding a sensitivity of 72%, a specificity of 99.7%, and an accuracy of99.3% for PET/CT with all cancer-bearing nodes >0.5 cm in diameter correctly identified on noncontrast PET/CT.41 The study by Amit et al42 found slightly lower sensitivity (60%) and specificity (94%), potentially due to the number of false-negative findings for lymph nodes measuring 0.5 to 1.5 cm.42 Like dedicated PET, PET/CT cannot replace surgical staging altogether; however, in the described study PET/CT improvedthe diagnosis in 43% with somewhat fewer (10 of 32) patients with altered therapies by excluding nodal disease or by identifying distant or nodaldisease in at least one location.42 When Choi and colleagues43 compared PET/CT with MRI for cervical cancer staging, noncontrast PET/CT hadgreater sensitivity for lymph node metastases in all of the abdominal/pelvic nodal groups studied, but accuracies for both modalities were foundto be similar. This finding contrasted with a previous report that PET had better accuracy than MRI in defining pelvic lymph node involvement.44
In the Choi study, both modalities had relatively poor sensitivities, 30.3% for MRI and 57.6% for PET/CT.
A recent study by Loft et al45 may renew interest in the use of PET/CT to characterize lymph nodes during cervical cancer staging. In a groupof patients with all stages of cervical cancer, PET/CT with intravenous and bowel contrast enhancement had a sensitivity of 75% and a specificity of 96% in detecting pelvic lymph node metastases when compared with histologic data, and a sensitivity of 100% and a specificity of99% in detecting para-aortic lymph node disease. However, in this study, PET/CT suggested distant metastases in 19 pa-tients, 9 of whom didnot actually have metastatic disease. While contrast-enhanced PET/CT may im prove results for lymph node staging, findings suggesting othermetastases must be scrutinized carefully and corroborated with other imaging or pathologic data.
Other cancers
In advanced gastric cancer, PET can provide information related to nodal metastases and other cancers, and visualization of early intes-tinal-type gastric cancer primaries on PET suggests the potential for nodal spread.46,47 However, the variability of the sensitivity of PET fornodal involvement precludes the ability to utilize PET alone in making decisions regarding the extent of nodal dissection.48 PET may provide additional information over physical examination and CT on lymph node involvement in staging of anal cancer.49
In the preoperative evaluation of patients with endometrial cancer, the inability to image subcentimeter lymph nodes using PETlimits itsability to be used as a tool to guide the decision to perform retroperitoneal lymph node dissection. However, PET has demonstrated the ability to detect foci of extra uterine endometrial cancer not found on CT or MRI and can characterize anatomic lesions with high specificity.50 Therole of PET/CT in preoperative staging of endometrial cancer has yet to be defined.
Planning radiation therapy
Only a few years ago, the rationale for and parameters of radiation treatment plans were based solely on anatomical data, an approach that israpidly transforming to include functional imaging data. Currently, radiation oncologists are utilizing PET and PET/CT data in making decisionsregarding the use of aggressive treatment options and, at times, are incorporating functional data directly into the basis of the treatment plan.
High18 F-FDG uptake in pretreatment studies of cervical cancer patients may define a population in whom more aggressive therapy isrequired.51 There is growing evidence that PET/CT may change radiation treatment fields in a small number of cervical cancer patients.42 Guidelines have been published describing the treatment- and organ-specific parameters for the use of PET/CT in intensity-modulated radiotherapy (IMRT) for cervical cancer with involvement of the para-aortic lymph nodes.52 In planning radiation therapy for patients with rectalcancer using PET/CT, the inclusion of18 F-FDG or 3'-deoxy-3'-[18 F] fluorothymidine (FLT) uptake data significantly improved the interobserver nodal tumor volumes when compared with volumes determined by CT data alone.25
Initial investigations to define the role of PET imaging in patients undergoing brachytherapy for cervical cancer have been performed in smallnumbers of patients. Early data suggest that PET may be used to identify tumor response in individual patients53 and to define treatment plansthat offer improved dose coverage without a concomitant in crease in dose to critical organs.54
Selecting chemotherapy
Selecting appropriate chemotherapies based on radiotracer uptake on PET imaging was explored several years ago using18 F-fluorouracil18 F-FU) to predict the response of metastatic colorectal cancer to the chemotherapeutic regimen.55 This approach has recently gained re-newedinterest.56 Copper-64 (64 Cu)-labeled cetuximab, a chimeric anti- epidermal growth factor receptor (EGFR) monoclonal antibody (that is usedto quantitatively image EGFR expression levels), has identified the 20% to 25% of colorectal cancer patients in whom cetuximab therapy couldbe successful.57,58 The area of response prediction will likely grow rapidly in the coming years.
Detection of residual or recurrent cancer
Cancers of the colon and rectum
Although most patients who undergo surgical resection for colorectal cancers are thought to be potentially curable, approximately a thirdof patients will have recurrence of their cancer within 2 years, and half will have liver metastases within 5 years. Local recurrences or limited metastases to the lung or liver may be resectable in a large portion of these patients. In patients with symptoms or other markers of disease, such as a serum carcinoembryonic antigen (CEA) level, PET or PET/CT can be utilized to localize sites of recurrence when they arenot readily identified on single-modality anatomical imaging alone. PET/CT greatly adds to the accuracy of dedicated PET, with reportedsensitivities of 89% to 98% and specificities of 92% to 96% in the detection of recurrent colorectal cancer after resection.59,60 PET/CT is veryaccurate in distinguishing benign from malignant presacral abnormalities59 and is much more sensitive than CT in the identification of localrecurrence at the primary colorectal resection site.32
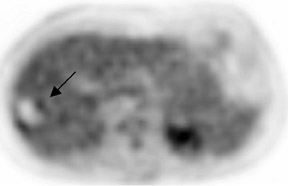
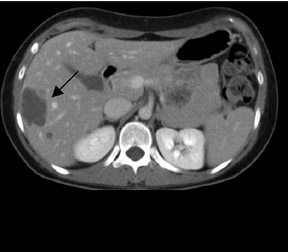
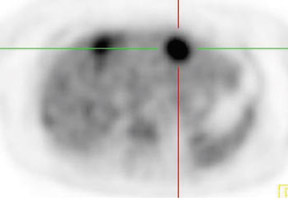
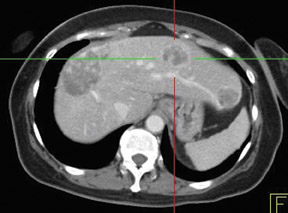
In patients with known cancer that is presumed to be isolated, the role of PET or PET/CT is to exclude other sites of disease that could render a patient incurable via surgery with or without a combination of other types of therapy. Early studies of PET indicated its usefulness in appropriately upstaging patients to avoid unnecessary surgery in up to 32% of cases.61 Data suggest that PET may be more sensitive than other imaging modalities for intrahepatic versus extrahepatic or peritoneal disease.18,62-65 PET/CT raises the specificity in the diagnosis of intrahepatic recurrence in patients with prior hepatic resection over CT alone from 50% to 100%.32 Likewise, PET/CT can be helpful in evaluating residual orrecurrent co-lorectal cancer in sites after radioablation or chemotherapy (Figure 1).
Ovarian cancer
The use of FDG-PET and PET/CT in the management of ovarian cancer patients has received unprecedented attention recently. This hasbeen a challenging question to address given the limited number of clinical situations in which pathology correlation of imaging findings ispossible. Recurrent ovarian cancer, regardless of histologic subtype, appears to be potentially FDG-avid and capable of being visualized onPET.6618 F-FDG uptake in recurrent ovarian cancers has been found to correlate most strongly with intratumor microvessel density and mitotic activity, but not histologic grade.67 PET has been reported to have high sensitivity for the detection of recurrent ovarian cancer; however,its reported specificities have varied widely.18,64,65 The sensitivity, specificity, and accuracy of FDG PET in the diagnosis of ovarian cancerrecurrence have been reported as 91.9%, 68.4%, and 84.8%, respectively.68 The accuracy of PET was found to be higher than that of conventional imaging methods or CA-125 levels in the diagnosis of recurrence. When compared with visual analysis, standard uptake values(SUVs) were not helpful in differentiating between benign and malignant tumors.68
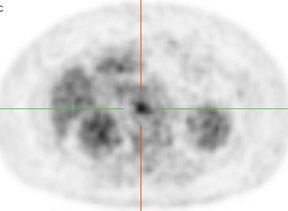
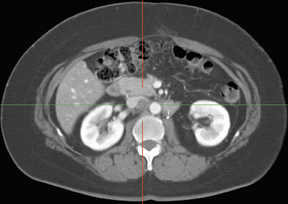
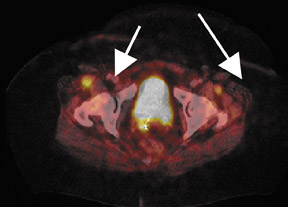
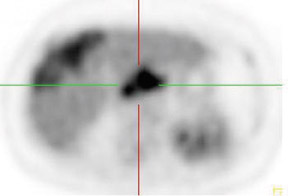
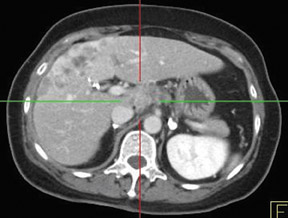
Whether through multimodal comparison or hardware-fusion, combining PET and CT appears to add to the value of either modality alone69,70 (Figure 2). PET/CT adds to the number of patients with recurrence found over CT alone71 and has shown significantly improved accuracy forthe detection of recurrent ovarian cancer in the chest and abdomen.72 However, an early retro spective study using fused PET/CT images to identify recurrent disease had poor specificity (40%). The sensitivity for lymph nodes was 100% in this study, but the sensitivity for small peritoneallesions (<1 cm) was extremely poor and only moderately better in diagnosing lesions >1 cm.73 Notably, only a fraction of the patients receivedoral contrast and none received intravenous contrast in this study. Another study of patients prior to second-look laparotomy in which PET/ CTwas performed without intravenous or oral contrast reported overall lesion-based sensitivity, specificity, and accuracy of 78%, 75%, and 89%,with the largest diameter lesion missed on PET/ CT being 0.5 cm.74 Chung and col-leagues75 presented a prospective study of 77 patients withsuspected recurrence of ovarian cancer of whom 58% had docu- mented recurrence found on surgical biopsy or clinical follow-up. The use ofPET/CT with oral contrast led to an overall sensitivity of 93.3%, specificity of 96.9%, and accuracy of 94.8% for the diagnosis of recurrent disease. In this population, PET/CT im pacted the diagnostic or treatment plan in 24.7% of patients, the majority of whom underwent un plannedtherapeutic procedures.75 These values were im proved significantly from those of earlier prospective studies using nonenhanced PET/CT.76 Contrast-enhanced PET/CT appears to dramatically improve the results of PET/CT in the diagnosis of recurrent ovarian cancer.
To properly determine the utility of PET and PET/CT in this setting, it is important to know what the oncologist does with the knowledgeof recurrent ovarian cancer. By indicating greater or lesser anatomical involvement or altering the known disease distribution in patients whopresent for detection of ovarian cancer recurrence, PET/CT can change management in the majority of cases because of its ability to distinguish between patients with localized disease and those with no evidence of disease.77 However, at least 1 study has reported no significant difference in the prognosis of patients at 2.5 years with positive versus negative PET scans following initial surgical treatment of ovarian can-cer.78 Whether or not to treat disease progression when it is identified in advanced ovarian cancer is a much debated topic. But an increasingnumber of less toxic therapeutic options are becoming available.79 Frequently, ovarian cancer pa-tients receive chronic regimens of chemotherapy with a variety of agents whose mechanisms and toxicities are complementary. Secondary cytoreductive surgery's role in treating recurrentovarian cancer remains controversial.80 In a small group of patients undergoing PET/CT for suspicion of ovarian cancer recurrence, Bristowand colleagues80 found a high positive predictive value of PET/CT for nodal involvement but found that PET/CT missed the majority of pathologically involved nodes in the same nodal basin as the target lymph nodes identified by PET/CT. The authors advocate PET/CT as a markerof re-gional node involvement and suggest that a single positive node on PET/CT is an indication for complete nodal excision of that basin.80 Using PET/CT with contrast administration as a first-line restaging tool in patients with suspected recurrence of ovarian cancer, Mangili andcoworkers71 have recently shown PET/CT that changed the management strategy in 44% of patients studied with 6 of 32 patients (19%) alteredfrom a "wait and see" approach to more aggressive management such as an instrument diagnostic procedure or surgical intervention.
Cervical and other uterine cancers
The use of FDG-PET for recurrent cervical cancer has been reported to have a sensitivity of 96.1%, a specificity of 84.4%, and an accuracy of 91.7%. In the same retrospective study, FDG-PET showed abnormalities in 85% of the patients with asymptomatic recurrent disease.Patients without evidence of distant metastases on PET underwent pelvic exenteration, and 37.5% of patients had a complete response.81 Otherreports have supported the use of PET in the diagnosis of recurrent cervical cancer in both symptomatic and asymptomatic women.82 In patientsundergoing PET following radiation treatment, a high sensitivity for extra pelvic lymph node disease was found, with a poorer sensitivity andspecificity for distant metastatic disease in the lung and bone.83 When data from PET is used to interpret findings on CT and MRI, significantimprovements in the false-negative and false-positive rates can be realized.84 Even in patients who have had therapy for recurrent disease, PEThas a role in defining persistent disease or future recurrences and may be more sensitive than MRI in detecting these entities.85
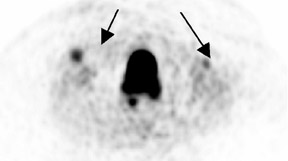
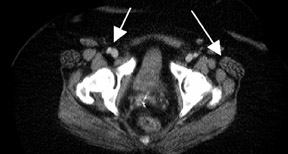
Chung and colleagues75 investigated the use of PET/CT in detecting recurrent cervical cancer and found the sensitivity, specificity, and accuracy to be 90.3%, 81.0%, and 86.5%, respectively. The authors found that PET/CT changed the management in 23.1% of patients by alteringthe treatment plan, initiating unplanned treatment, or canceling a planned diagnostic procedure. A recent study of 25 patients with treated cervical or endometrial cancers showed improved patient-specific and lesion-based accuracies for the detection of cancer recurrence using PET/CT86 (Figure 3).
Other cancers
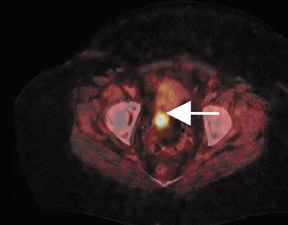
PET and PET/CT have been shown to have high sensitivity and specificity for the detection of recurrent cholangiocarcinoma87 (Figure4) and are useful in identifying locally recurrent pancreatic cancer.88
Using tumor markers to select patients for PET and PET/CT
Are there nonimaging indicators of recurrence that can better guide the selection of patients who may benefit from a PET/CT study? Manyreferrals for imaging cite the increase in a tumor marker-a molecule typically detected in excess in the blood of a patient in the presenceof tumor-as an indication for PET or PET/CT to investigate or localize recurrent disease. Important tumor markers in abdominal and pelvicmalignancies include (but are not limited to) glycoprotein markers such as prostate- specific antigen (PSA) for prostate cancer, CEA for colorectal and ovarian cancer, human chorionic gonadotropin (hCG), alpha-fetoprotein (AFP) for hepatocellular carcinoma and mucinous glycoprotein markers such as cancer antigen (CA)-19-9, CA-50, and CA-195 for pancreatic cancer and CA-125 for ovarian cancer.
Ideally, tumor markers would increase only in the presence of and in proportion to the bulk of malignancy, would respond rapidly to alterations in tumor size, would be specific for the suspected type of malignancy, and would guide other types of diagnostic investigations, such asPET/CT. Tumor markers of cancers in the abdomen and pelvis vary in their abilities to accomplish these tasks. Carcinoembryonic antigenmeasurements can be very useful in selecting patients for localization of colon cancer recurrence with PET/CT. Levels of CEA are typically repeated every 2 to 3 months following resection and raise concerns of cancer recurrence if they are increased from baseline levels on 2 re-peated occasions. A reported 59% to 76% of patients with recurrent disease will have an increase in serum CEA level witha specificity of approximately 80%. Between 60% and 90% of patients with an increased serum CEA level and the absence of disease on CTwill have recurrent disease found at laparotomy.89,90
Even though CEA levels may detect recurrent colon cancer several months before it can be identified on a CT scan, the benefit of this serumscreening procedure to patient survival or quality of life has not been firmly established, possibly because earlier detection does not necessarilybenefit the patient without concurrent localization. Early studies established the utility of PET in identifying a source of elevated CEA in a veryhigh fraction of patients who had negative findings on CT.61,91 More recent studies have corroborated the high patient-based sensitivity of PET inthis population. However, lower lesion-based sensitivities (57% to 75%) have been reported.92,93 At least 1 study has indicated that the volume ofFDG-avid findings on PET correlates with the level of serum CEA.94 Overall, in cases of rising CEA levels, PET and PET/CT may be useful tolocalize recurrent cancer with a high positive predictive value but, in a fraction of patients, may fail to localize all sites of disease or may fail tolocalize disease altogether. PET can identify approximately 90% of those patients who have unresectable disease and 81% of those patients withresectable disease prior to laparotomy.93
CA-125, a glycoprotein marker for ovarian cancer, is made by many normal tissues, including the endometrium, endothelium of the fallopian tubes, endocervix, normal ovary, and meso thelial cells of the pleura, pericardium, and peritoneum.95 Elevated levels can be found secondary to menstruation, pregnancy, endometriosis, peritonitis, and cirrhosis.96 CA-125 levels are elevated in the setting of vascular invasion, tissue destruction, and inflammation associated with malignant disease. Thus, not surprisingly, only 50% of stage 1 epithelial ovarian cancershave an elevated serum CA-125 versus 75% to 90% of patients with advanced disease. Following primary treatment, relapse or progressionof disease is associated with a doubling of CA-125 or a rise above normal levels in the vast majority of cases, and clinical criteria includingCA-125 as an indicator of disease can provide a lead time of >2 months in identifying the progression of disease.97,98 Some small studies suggest that PET is at least as sensitive and accurate as serum CA-125 determination in the identification of recurrent ovarian cancer,99,100 and recurrent disease can be found on PET even in the absence of an elevated level of CA-125.99 There may be threshold values of tumor markers abovewhich imaging physicians are more likely to localize a focus of recurrent cancer using PET/CT101 ; however, this has not been well establishedfor either CEA or CA-125.
Data regarding the value of other serum-based tumor markers in selecting patients for PET or PET/CT to detect recurrent abdominal orpelvic cancers are sparse. Schöder and colleagues102 have reported localization of local or metastatic recurrent prostate cancer in 31% of patients referred with PSA relapse and determined a relationship between PSA level and detection ability. In detecting recurrence of hepatocellular carcinoma in patients referred with a rising AFP, Chen and colleagues103 have reported the sensitivity, specificity,and accuracy of PET to be 73.3%, 100%, and 74.2%, respectively. Recently, investigators have explored a novel approach using biomarkers from the primary tumor to identify patients in whom a PET could be valuable in the detection of recurrence. For example, in cervicalcancer, microvessel density and p53 profiles in the primary tumor have been found to be potentially helpful in preselecting patients in whomrecurrence can be identified on FDG-PET.104 In the era of "personalized medicine," this type of patient-specific, biomarker-based diagnostic selection process likely represents the future of molecular markers and PET/CT imaging.
Monitoring treatment with chemotherapy, biologic agents, or radiation
The use of PET to identify biologic response to therapy prior to an anatomic response detectable by anatomic imaging modalities is agrowing area of research and clinical applications. Pharmaceutical companies are investigating ways to optimize drug candidate trials, andphysicians seek reassurance of the effects of cytostatic drugs and information to decrease the toxicity of therapeutic regimens that are notbeneficial to the patient. The exact methods of defining treatment response with PET are still evolving. Currently, oncologists use a changein the SUV between the initial pretherapy study and a study performed following some increment of therapy (eg, 3 cycles of chemotherapy). While complex quantitative methods of PET data acquisition and analysis may provide enhanced ability to monitor response to therapy, these are not practiced routinely in the clinical setting.105
One of the first and most studied applications of PET in monitoring therapy of an abdominal cancer was for cases of gastrointestinal stromal tumor (GIST). In multiple studies, PET has been shown to separate responders from nonresponders very early in the course of treatmentwith the inhibitor to tyrosine kinase growth factor receptor, imatinib mesylate. Jager and colleagues106 reported changes in18 F-FDG uptakeafter 1 week of treatment that exceeded the magnitude of tumor volume changes on CT at 8 weeks. PET/CT may become the next standardfor determining response to therapy in this population.107
In a cohort of patients undergoing 3 cycles of carboplatin-based neoadjuvant chemotherapy followed by cyto-reductive surgery foradvanced-stage ovarian cancer, metabolic response defined on FDG-PET studies performed after the first or third cycle correlated with overall survival and was more accurate than clinical or histopathologic response criteria.108 The threshold for decrease in SUV from baseline asan indicator of positive response was 20% after the first cycle and 55% after the third cycle in this study.
Only very initial work has been published in the evaluation of response to neoadjuvant chemotherapy for cervical cancer. In some cases,response defined by changes in SUV on PET may correspond to histopathologic changes better than changes seen on MRI during the courseof treatment.109 Response of cervical cancer to external radiation has been measured using PET and has demonstrated a 50% decrease inphysiologic tumor volume within 20 days of beginning therapy.110
Using sophisticated quantitative methods, response to colorectal cancer therapy can be measured, even following completion of asingle treatment cycle.111 In rectal cancer, findings on PET 6 weeks after completion of neoadjuvant radiochemotherapy have beenshown to be an independent prognostic predictor of overall and disease-free survival.112 A 66% decrease in SUV on PET/CT 5 to 6 weeksfollowing neoadjuvant therapy provided an 80% accuracy in defining responders versus nonresponders.113 A change of 52% in SUV separates responders from nonresponders on PET 12 days following completion of neoadjuvant therapy.114 Kalff and colleagues115 studiedthe prognostic information from the change in FDG uptake following chemoradiation before radical curative surgery in patients withlocally ad-vanced rectal cancer. Using a classification of complete response, partial response, stable, or progressive disease, they foundthat response on PET was associated with overall survival duration and time to progression but were unable to identify a threshold of maximum SUV change predicting survival in this patient population.115 Complete pathologic response was the only other predictorof survival.
PET may also be used to defind the response of gastric cancer to chemo therapy,6 in monitoring the response of unresectable hepatic tumors toY-90 microsphere therapy for unresectable hepatic tumors,116 and in many other applications that have yet to be elucidated. Undoubtedly, as molecular therapies gain greater targeting specificity, the use of PET to monitor therapy will grow.117
Future directions
Few cost analyses to determine the utility of PET in diagnosing, staging, or restaging abdominal cancers have included the scenario ofPET and CT used together. Two recent studies suggest a significant cost savings in pa-tients undergoing PET for staging colorectal carcinoma with potential liver metastases when compared with the use of CT alone.118 The study by Lejeune and colleagues119 found a majorrisk reduction in terms of decreased exploratory surgery when PET and CT are implemented together. Further studies must explore theefficacy and cost effectiveness of using PET/CT in the areas of treatment planning and monitoring as well as in more controversial areassuch as PET/CT colonography and whole-body PET/CT screening for cancers in asymptomatic, otherwise healthy patients.120,121
As new molecular therapies are realized, PET/CT imaging will adapt to more specifically characterize molecular and cellular processes forimproved therapeutic efficiency. Thisexpanded role of molec- ularimaging will require new radiopharmaceuticals for PET. Intriguing work in the synthesis and characterization of antibody- and antibody-fragment-based PET agents has yielded an array of agents capable of specifictumor targeting.122-128 In addition, agents characterizing specific molecular processes such as tumor hypoxia and tyrosine kinase modulation129,131 will be useful in planning therapies. Labeled viruses, macrophages, and stem effector cells will be helpful in tracking oncolytic viral therapies,immunotherapies, and stem-cell-based therapies, respectively.131-133
Conclusion
PET/CT is rapidly becoming the new standard of functional imaging in the abdomen and pelvis. PET/CT assists in localization of normalphysiologic uptake, such as that in the rectum or in the corpus luteum in the periovulatory period134 (Figure 5). Several recognized artifactsare introduced by using CT as the basis for attenuation correction in PET. Generally, these are density artifacts secondary to barium oral contrast or metal prostheses135 and are easily identified.
Given the mounting evidence of greater diagnostic accuracy of PET/CT performed with contrast enhancement, intravenous and posi-tive/negative bowel contrast should be provided to patients undergoing PET/CT for evaluation of abdominal or pelvic tumors whenever possible. Additional benefit may be derived from disease-specific protocols, including multiphase CT imaging in patients with potential livermetastases or PET/CT with colonographic reconstructions when staging colon cancer. Interpretation of PET/CT should include review ofeach full dataset (PET and CT) as well as correlation of functional and anatomic findings on fusion images or through intra-dataset triangulation tools provided on modern systems. Future technical improvements are required to better coregister PET and CT images of theabdomen, as they are subject to misalignment secondary to respiratory motion artifact or shifts in organ position between scans.
Molecular imaging of abdominal and pelvic cancers with PET and PET/CT promises to remain an exciting area of research and clinicalimaging practice as the promise of molecular-based medicine is translated into clinical care.
REFERENCES
- Pfannenberg AC, Aschoff P, Brechtel K, et al. Value of contrast-enhanced multiphase CT in combined PET/CT protocols for oncological imaging. Br J Radiol. 2007;80:437-450.
- Schöder H, Larson SM. Positron emission to-mography for prostate, bladder, and renal cancer. Semin Nucl Med. 2004; 34:274-292.
- Podoloff DA, Advani RH, Allred C, et al. NCCN task force report: Positron emission tomography (PET)/computed tomography (CT). Scanning in cancer. J Natl Compr Canc Netw. 2007;5 suppl 1:S1-S22.
- Yang QM, Bando E, Kawamura T, et al. The diagnostic value of PET-CT for peritoneal dissemination of abdominal malignancies. Gan To Kagaku Ryoho. 2006;33:1817-1821.
- Borbath I, Van Beers BE, Lonneux M, et al. Preoperative assessment of pancreatic tumors using magnetic resonance imaging, endoscopic ultrasonography, positron emission tomography and laparoscopy. Pancreatology. 2005;5:553-561.
- Lim JS, Yun MJ, Kim MJ, et al. CT and PET in stomach cancer: Preoperative staging and monitoring of response to therapy. Radiographics. 2006;26:143-156.
- Israel O, Yefremov N, Bar-Shalom R, et al. PET/CT detection of unexpected gastrointestinal foci of 18F-FDG uptake: Incidence, localization patterns, and clinical significance. J Nucl Med. 2005;46:758-762.
- Nguyen BD. PET imaging of concomitant colonic hyperplastic polyp and tubulovillous adenoma. Clin Nucl Med. 2005;30:48-50.
- Yasuda S, Fujii H, Nakahara T, et al. 18F-FDG PET detection of colonic adenomas. J Nucl Med 2001;42:989-992.
- Drenth JP, Nagengast FM, Oyen WJ. Evaluation of (pre-)malignant colonic abnormalities: endoscopic validation of FDG-PET findings. Eur J Nucl Med. 2001;28:1766-1769.
- van Kouwen MC, Nagengast FM, Jansen JB, et al. 2-(18F)-fluoro-2-deoxy-D-glucose positron emission tomography detects clinical relevant adenomas of the colon: Aprospective study. J Clin Oncol. 2005;23:3713-3717.
- Pickhardt PJ, Choi JR, Hwang I, et al. Computed tomographic virtual colonoscopy to screen for colorectal neoplasia in asymptomatic adults. N Engl J Med. 2003; 349:2191-2200. Comments in: ACP J Club. 2004;141(1):22-23; CMAJ. 2004;170: 1392; Gastroenterology. 2004;126:1910-1911; discussion 1911-1912; Korean J Gastroenterol. 2004;43:71-73; N Engl J Med. 2003;349: 2261-2264; N Engl J Med. 2004;350:1148-1150; author reply 1148-1150; Rev Gastroenterol Disord. 2005; 5:227-229.
- Yee J, Akerkar GA, Hung RK. Colorectal neoplasia: Performance characteristics of CT colonography for detection in 300 patients. Radiology. 2001;219:685-692.
- Gollub MJ, Akhurst T, Markowitz AJ, et al. Combined CT colonography and 18F-FDG PET of colon polyps: Potential technique for selective detection of cancer and precancerous lesions. AJR Am J Roentgenol. 2007;188:130-138.
- Mainenti PP, Salvatore B, D'Antonio D, et al. PET/CT colonography in patients with colorectal polyps: A feasibility study. Eur J Nucl Med Mol Imaging. 2007;34:1594-1603.
- Veit P, Kühle C, Beyer T, et al. Whole-body positron emission tomography/computed tomography (PET/CT) tumour staging with integrated PET/CT colonography: Technicalfeasibility and first experiences in patients with colorectal cancer. Gut. 2006;55:68-73.
- Grab D, Flock F, Stöhr I, et al. Classification of asymptomatic adnexal masses by ultrasound, magnetic resonance imaging, and positron emission tomography. Gynecol Oncol. 2000;77:454-459.
- Hubner KF, McDonald TW, Niethammer JG, et al. Assessment of primary and metastatic ovarian cancer by positron emission tomography (PET) using 2-[18F]deoxyglucose(2-[18F]FDG). Gynecol Oncol. 1993;51:197-204.
- Kawahara K, Yoshida Y, Kurokawa T, et al. Evaluation of positron emission tomography with tracer 18-fluorodeoxyglucose in addition to magnetic resonance imaging in thediagnosis of ovarian cancer in selected women after ultraso- nography. J Comput Assist Tomogr. 2004; 28:505-516.
- Kubik-Huch RA, Dörffler W, von Schulthess GK, et al. Value of (18F)-FDG positron emission tomography, computed tomography, and magnetic resonance imaging in diagnosing primary and recurrent ovarian carcinoma. Eur Radiol. 2000; 10:761-767.
- Fenchel S, Grab D, Nuessle K, et al. Asymptomatic adnexal masses: Correlation of FDG PET and histopathologic findings. Radiology. 2002; 223:780-788.
- Risum S, Hogdall C, Loft A, et al. The diagnostic value of PET/CT for primary ovarian cancer-a prospective study. Gynecol Oncol. 2007;105: 145-149.
- Mansour JC, Schwartz L, Pandit-Taskar N, et al. The utility of F-18 fluorodeoxyglucose whole body PET imaging for determining malignancy in cystic lesions of the pancreas. J Gastrointest Surg. 2006;10:1354-1360.
- Llamas-Elvira JM, Rodríguez-Fernández A, et al. Fluorine-18 fluorodeoxyglucose PET in the preoperative staging of colorectal cancer. Eur J Nucl Med Mol Imaging. 2007;34:859-867.
- Park IJ, Kim HC, Yu CS, et al. Efficacy of PET/CT in the accurate evaluation of primary colorectal carcinoma. Eur J Surg Oncol. 2006; 32:941-947.
- Joyce DL, Wahl RL, Patel PV, et al. Preoperative positron emission tomography to evaluate potentially resectable hepatic colorectal metastases. Arch Surg. 2006;141:1220-1226; discussion 1227.
- de Geus-Oei LF, Wiering B, Krabbe PF, et al. FDG-PET for prediction of survival of patients with metastatic colorectal carcinoma. Ann Oncol. 2006;17:1650-1655.
- Kinner S, Antoch G, Bockisch A, Veit-Haibach P. Whole-body PET/CT-colonography: A possible new concept for colorectal cancer staging. Abdom Imaging. 2007;32:606-612.
- Bipat S, van Leeuwen MS, Comans EF, et al. Colorectal liver metastases: CT, MR imaging, and PET for diagnosis-meta-analysis. Radiology. 2005;237:123-131.
- Fernandez FG, Drebin JA, Linehan DC, et al. Five-year survival after resection of hepatic metastases from colorectal cancer in patients screened by positron emissiontomography with F-18 fluorodeoxyglucose (FDG-PET). Ann Surg. 2004; 240:438-447; discussion 447-450.
- Amthauer H, Denecke T, Hildebrandt B, et al. Evaluation of patients with liver metastases from colorectal cancer for locally ablative treatment with laser inducedthermotherapy. Impact of PET with 18F-fluorodeoxyglucose on therapeutic decisions. Nuklearmedizin. 2006;45:177-184.
- Selzner M, Hany TF, Wildbrett P, et al. Does the novel PET/CT imaging modality impact on the treatment of patients with metastatic colorectal cancer of the liver? Ann Surg. 2004;240:1027-1034; discussion 1035-1036.
- Kim JH, Czernin J, Allen-Auerbach MS, et al. Comparison between 18F-FDG PET, in-line PET/CT, and software fusion for restaging of recurrent colorectal cancer. J Nucl Med. 2005; 46:587-595.
- Nakamoto Y, Sakamoto S, Okada T, et al. Clinical value of manual fusion of PET and CT images in patients with suspected recurrent colorectal cancer. AJR Am J Roentgenol. 2007;188:257-267.
- Wiering B, Ruers TJ, Krabbe PF, et al. Comparison of multiphase CT, FDG-PET and intra-operative ultrasound in patients with colorectal liver metastases selected for surgery. Ann Surg Oncol. 2007;14:818-826.
- Unger JB, Lilien DL, Caldito G, et al. The prognostic value of pretreatment 2-[18F]-fluoro-2-deoxy-D-glucose positron emission tomography scan in women with cervicalcancer. Int J Gynecol Cancer. 2007;17:1062-1067.
- Wong TZ, Jones EL, Coleman RE. Positron emission tomography with 2-deoxy-2-[(18)F] fluoro-D-glucose for evaluating local and distant disease in patients with cervicalcancer. Mol Imaging Biol. 2004;6:55-62.
- Wright JD, Dehdashti F, Herzog TJ, et al. Preoperative lymph node staging of early-stage cervical carcinoma by [18F]-fluoro-2-deoxy-D-glucose- positron emission tomography. Cancer. 2005; 104:2484-2491.
- Roh JW, Seo SS, Lee S, et al. Role of positron emission tomography in pretreatment lymph node staging of uterine cervical cancer: A prospective surgicopathologic correlation study. Eur J Cancer. 2005;41:2086-2092.
- Chou HH, Chang TC, Yen TC, et al. Low value of [18F]-fluoro-2-deoxy-D-glucose positron emission tomography in primary staging of early-stage cervical cancer beforeradical hysterectomy. J Clin Oncol. 2006;24:123-128.
- Sironi S, Buda A, Picchio M, et al. Lymph node metastasis in patients with clinical early-stage cervical cancer: Detection with integrated FDG PET/CT. Radiology. 2006;238:272-279.
- Amit A, Beck D, Lowenstein L, et al. The role of hybrid PET/CT in the evaluation of patients with cervical cancer. Gynecol Oncol. 2006;100:65-69.
- Choi HJ, Roh JW, Seo SS, et al. Comparison of the accuracy of magnetic resonance imaging and positron emission tomography/computed tomography in the presurgicaldetection of lymph node metastases in patients with uterine cervical carcinoma: A prospective study. Cancer. 2006; 106:914-922.
- Park W, Park YJ, Huh SJ, et al. The usefulness of MRI and PET imaging for the detection of parametrial involvement and lymph node metastasis in patients with cervicalcancer. Jpn J Clin Oncol. 2005;35:260-264.
- Loft A, Berthelsen AK, Roed H, et al. The diagnostic value of PET/CT scanning in patients with cervical cancer: A prospective study. Gynecol Oncol. 2007;106:29-34.
- Mukai K, Ishida Y, Okajima K, et al. Usefulness of preoperative FDG-PET for detection of gastric cancer. Gastric Cancer. 2006;9:192-196.
- Kim SK, Kang KW, Lee JS, et al. Assessment of lymph node metastases using 18F-FDG PET in patients with advanced gastric cancer. Eur J Nucl Med Mol Imaging. 2006;33:148-155.
- Yun M, Lim JS, Noh SH, et al. Lymph node staging of gastric cancer using (18)F-FDG PET: A comparison study with CT. J Nucl Med. 2005; 46:1582-1588.
- Cotter SE, Grigsby PW, Siegel BA, et al. FDG-PET/CT in the evaluation of anal carcinoma. Int J Radiat Oncol Biol Phys. 2006;65:720-725.
- Suzuki R, Miyagi E, Takahashi N, et al. Validity of positron emission tomography using fluoro-2-deoxyglucose for the preoperative evaluation of endometrial cancer. Int J Gynecol Cancer. 2007; 17:890-896.
- Xue F, Lin LL, Dehdashti F, et al. F-18 fluorodeoxyglucose uptake in primary cervical cancer as an indicator of prognosis after radiation therapy. Gynecol Oncol. 2006;101:147-151.
- Esthappan J, Mutic S, Malyapa RS, et al. Treatment planning guidelines regarding the use of CT/PET-guided IMRT for cervical carcinoma with positive paraaortic lymph nodes. Int J Radiat Oncol Biol Phys. 2004;58:1289-1297.
- Lin LL, Mutic S, Malyapa RS, et al. Sequential FDG-PET brachytherapy treatment planning in carcinoma of the cervix. Int J Radiat Oncol Biol Phys. 2005;63:1494-1501.
- Lin LL, Mutic S, Low DA, et al. Adaptive brachytherapy treatment planning for cervical cancer using FDG-PET. Int J Radiat Oncol Biol Phys. 2007;67:91-96.
- Dimitrakopoulou-Strauss A, Strauss LG, Schlag P, et al. Fluorine-18-fluorouracil to predict therapy response in liver metastases from colorectal carcinoma. J Nucl Med. 1998;39:1197-1202.
- Gupta N, Saleem A, Kötz B, et al. Carbogen and nicotinamide increase blood flow and 5-fluorouracil delivery but not 5-fluorouracil retention in colorectal cancer metastasesin patients. Clin Cancer Res. 2006;12:3115-3123.
- Cai W, Chen K, He L, et al. Quantitative PET of EGFR expression in xenograft-bearing mice using (64)Cu-labeled cetuximab, a chimeric anti-EGFR monoclonal antibody. Eur J Nucl Med Mol Imaging. 2007; 34:850-858.
- Pantaleo MA, Fanti S, Lollini PL, et al. PET detection of epidermal growth factor receptor in colorectal cancer: A real predictor of response to cetuximab treatment? Eur J Nucl Med Mol Imaging. 2007;34:1510-1511. Comment on: Eur J Nucl Med Mol Imaging. 2007;34:850-858.
- Even-Sapir E, Parag Y, Lerman H, et al. Detection of recurrence in patients with rectal cancer: PET/CT after abdominoperineal or anterior resection. Radiology. 2004;232:815-822.
- Votrubova J, Belohlavek O, Jaruskova M, et al. The role of FDG-PET/CT in the detection of recurrent colorectal cancer. Eur J Nucl Med Mol Imaging. 2006;33:779-784.
- Valk PE, Abella-Columna E, Haseman MK, et al. Whole-body PET imaging with [18F]fluoro-deoxyglucose in management of recurrent co-lorectal cancer. Arch Surg. 1999;134:503-511; discussion 511-503.
- Schiepers C, Penninckx F, De Vadder N, et al. Contribution of PET in the diagnosis of recurrent colorectal cancer: Comparison with conventional imaging. Eur J Surg Oncol. 1995;21:517-522.
- Tanaka T, Kawai Y, Kanai M, et al. Usefulness of FDG-positron emission tomography in diagnosing peritoneal recurrence of colorectal cancer. Am J Surg. 2002;184:433-436.
- Hubner KF, McDonald TW, Smith GT, Thie JA. Detection of recurrent ovarian cancer by PET using FDG. Clin Positron Imaging. 1999;2:346.
- Nakamoto Y, Saga T, Ishimori T, et al. Clinical value of positron emission tomography with FDG for recurrent ovarian cancer. AJR Am J Roentgenol. 2001;176:1449-1454.Comment in: AJR Am J Roentgenol. 2002;178:1030.
- Martínez-Román S, Ramirez PT, Oh J, et al. Combined positron emission tomography and computed tomography for the detection of recurrent ovarian mucinous adenocarcinoma. Gynecol Oncol. 2005;96:888-891.
- Cho SM, Park YG, Lee JM, et al. 18F-fluorodeoxyglucose positron emission tomography in patients with recurrent ovarian cancer: In comparison with vascularity, Ki-67,p53, and histologic grade. Eur Radiol. 2007;17:409-417.
- García-Velloso MJ, Jurado M, Ceamanos C, et al. Diagnostic accuracy of FDG PET in the follow-up of platinum-sensitive epithelial ovarian carcinoma. Eur J Nucl Med Mol Imaging. 2007;34: 1396-1405.
- Hauth EA, Antoch G, Stattaus J, et al. Evaluation of integrated whole-body PET/CT in the detection of recurrent ovarian cancer. Eur J Radiol. 2005;56:263-268.
- Picchio M, Sironi S, Messa C, et al. Advanced ovarian carcinoma: Usefulness of [(18)F]FDG-PET in combination with CT for lesion detection after primary treatment. Q J Nucl Med. 2003;47:77-84.
- Mangili G, Picchio M, Sironi S, et al. Integrated PET/CT as a first-line re-staging modality in patients with suspected recurrence of ovarian cancer. Eur J Nucl Med Mol Imaging. 2007;34:658-666.
- Sebastian S, Lee SI, Horowitz NS, et al. PET-CT vs. CT alone in ovarian cancer recurrence. Abdom Imaging. 2008;33:112-118.
- Pannu HK, Cohade C, Bristow RE, et al. PET-CT detection of abdominal recurrence of ovarian cancer: Radiologic-surgical correlation. Abdom Imaging. 2004;29:398-403.
- Sironi S, Messa C, Mangili G, et al. Integrated FDG PET/CT in patients with persistent ovarian cancer: Correlation with histologic findings. Radiology. 2004;233:433-440.
- Chung HH, Jo H, Kang WJ, et al. Clinical impact of integrated PET/CT on the management of suspected cervical cancer recurrence. Gynecol Oncol. 2007;104:529-534.
- Nanni C, Rubello D, Farsad M, et al. (18)F-FDG PET/CT in the evaluation of recurrent ovarian cancer: A prospective study on forty-one patients. Eur J Surg Oncol. 2005;31:792-797.
- Simcock B, Neesham D, Quinn M, et al. The impact of PET/CT in the management of recurrent ovarian cancer. Gynecol Oncol. 2006;103:271-276.
- Kurosaki H, Oriuchi N, Okazaki A, et al. Prognostic value of FDG-PET in patients with ovarian carcinoma following surgical treatment. Ann Nucl Med. 2006;20:171-174.
- Herzog TJ. Recurrent ovarian cancer: How important is it to treat to disease progression? Clin Cancer Res. 2004;10:7439-7449.
- Bristow RE, Giuntoli RL 2nd, Pannu HK, et al. Combined PET/CT for detecting recurrent ovarian cancer limited to retroperitoneal lymph nodes. Gynecol Oncol . 2005;99:294-300. Comment in: Gynecol Oncol. 2006;101:550-551; author reply 551-552.
- Chung HH, Kim SK, Kim TH, et al. Clinical impact of FDG-PET imaging in post-therapy surveillance of uterine cervical cancer: From diagnosis to prognosis. Gynecol Oncol. 2006;103: 165-170.
- Unger JB, Ivy JJ, Connor P, et al. Detection of recurrent cervical cancer by whole-body FDG PET scan in asymptomatic and symptomatic women. Gynecol Oncol. 2004;94:212-216.
- Sakurai H, Suzuki Y, Nonaka T, et al. FDG-PET in the detection of recurrence of uterine cervical carcinoma following radiation therapy-tumor volume and FDG uptakevalue. Gynecol Oncol. 2006;100:601-607.
- Yen TC, Lai CH, Ma SY, et al. Comparative benefits and limitations of (18)F-FDG PET and CT-MRI in documented or suspected recurrent cervical cancer. Eur J Nucl Med Mol Imaging. 2006; 33:1399-1407.
- Lin CT, Yen TC, Chang TC, et al. Role of [18F]fluoro-2-deoxy-D-glucose positron emission tomography in re-recurrent cervical cancer. Int J Gynecol Cancer. 2006;16:1994-2003.
- Sironi S, Picchio M, Landoni C, et al. Post-therapy surveillance of patients with uterine cancers: Value of integrated FDG PET/CT in the detection of recurrence. Eur J Nucl Med Mol Imaging. 2007; 34:472-479.
- Jadvar H, Henderson RW, Conti PS. [F-18]fluorodeoxyglucose positron emission tomography and positron emission tomography: Computed tomography in recurrent andmetastatic cholangiocarcinoma. J Comput Assist Tomogr. 2007;31: 223-228.
- Ruf J, Lopez Hänninen E, Oettle H, et al. Detection of recurrent pancreatic cancer: Comparison of FDG-PET with CT/MRI. Pancreatology. 2005(2-3);5:266-272.
- Minton JP, Hoehn JL, Gerber DM, et al. Results of a 400-patient carcinoembryonic antigen second-look colorectal cancer study. Cancer. 1985;55:1284-1290.
- Sardi A, Nieroda CA, Siddiqi MA, et al. Carcinoembryonic antigen directed multiple surgical procedures for recurrent colon cancer confined to the liver. Am Surg. 1990;56:255-259.
- Flanagan FL, Dehdashti F, Ogunbiyi OA, et al. Utility of FDG-PET for investigating unexplained plasma CEA elevation in patients with colorectal cancer. Ann Surg. 1998;227:319-323.
- Flamen P, Hoekstra OS, Homans F, et al. Unexplained rising carcinoembryonic antigen (CEA) in the postoperative surveillance of colorectal cancer: The utility of positronemission tomography (PET). Eur J Cancer. 2001;37:862-869.
- Libutti SK, Alexander HR Jr, Choyke P, et al. A prospective study of 2-[18F] fluoro-2-deoxy-D-glucose/positron emission tomography scan, 99mTc-labeled arcitumomab(CEA-scan), and blind second-look laparotomy for detecting colon cancer recurrence in patients with increasing carcinoembryonic antigen levels. Ann Surg Oncol. 2001; 8:779-786. Comment in: Ann Surg Oncol. 2001;8:752-753.
- Choi MY, Lee KM, Chung JK, et al. Correlation between serum CEA level and metabolic volume as determined by FDG PET in postoperative patients with recurrent colorectal cancer. Ann Nucl Med. 2005;19:123-129.
- Meyer T, Rustin GJ. Role of tumour markers in monitoring epithelial ovarian cancer. Br J Cancer 2000;82:1535-1538.
- Moss EL, Hollingworth J, Reynolds TM. The role of CA125 in clinical practice. J Clin Pathol. 2005;58:308-312.
- Rustin GJ, Nelstrop AE, Tuxen MK, Lambert HE. Defining progression of ovarian carcinoma during follow-up according to CA 125: A North Thames Ovary Group Study. Ann Oncol. 1996;7:361-364.
- van der Burg ME, Lammes FB, Verweij J. The role of CA 125 in the early diagnosis of progressive disease in ovarian cancer. Ann Oncol. 1990;1:301-302.
- García Velloso MJ, Boán García JF, Villar Luque LM, Aramendia Beitia JM, Lopez Garcia G, Richter Echeverria JA. [F-18-FDG positron emission tomography in the diagnosis of ovarian recurrence. Comparison with CT scan and CA 125]. Article in Spanish. Rev Esp Med Nucl. 2003; 22:217-223.
- Torizuka T, Nobezawa S, Kanno T, et al. Ovarian cancer recurrence: Role of whole-body positron emission tomography using 2-[fluorine-18]-fluoro-2-deoxy-D-glucose. Eu J Nucl Med Mol Imaging. 2002;29:797-803.
- Menzel C, Dobert N, Hamscho N, et al. The influence of CA 125 and CEA levels on the results of (18)F-deoxyglucose positron emission tomography in suspected recurrence of epithelial ovarian cancer. Strahlenther Onkol. 2004;180:497-501.
- Schöder H, Herrmann K, Gönen M, et al. 2-[18F]fluoro-2-deoxyglucose positron emission tomography for the detection of disease in patients with prostate-specific antigen relapse after radical prostatectomy. Clin Cancer Res. 2005;11:4761-4769.
- Chen YK, Hsieh DS, Liao CS, et al. Utility of FDG-PET for investigating unexplained serum AFP elevation in patients with suspected hepatocellular carcinoma recurrence. Anticancer Res. 2005;25:4719-4725.
- van der Veldt AA, Hooft L, van Diest PJ, et al. Microvessel density and p53 in detecting cervical cancer by FDG PET in cases of suspected recurrence. Eur J Nucl Med Mol Imaging. 2006;33: 1408-1416.
- Strauss LG, Klippel S, Pan L, et al. Assessment of quantitative FDG PET data in primary colorectal tumours: Which parameters are important with respect to tumour detection? Eur J Nucl Med Mol Imaging. 2007;34:868-877.
- Jager PL, Gietema JA, van der Graaf WT. Imatinib mesylate for the treatment of gastrointestinal stromal tumours: Best monitored with FDG PET. Nucl Med Commun. 2004;25:433-438. Comment in: Nucl Med Commun. 2004;25:429-432.
- Goerres GW, Stupp R, Barghouth G, et al. The value of PET, CT and in-line PET/CT in patients with gastrointestinal stromal tumours: Long-term outcome of treatment with imatinib mesylate. Eur J Nucl Med Mol Imaging. 2005;32: 153-162.
- Avril N, Sassen S, Schmalfeldt B, et al. Prediction of response to neoadjuvant chemotherapy by sequential F-18-fluorodeoxyglucose positron emission tomography in patients with advanced-stage ovarian cancer. J Clin Oncol. 2005;23:7445-7453. Erratum in: J Clin Oncol. 2005;23:9445. Comment in: J Clin Oncol. 2005;23:7385-73857.
- Yoshida Y, Kurokawa T, Kawahara K, et al. Metabolic monitoring of advanced uterine cervical cancer neoadjuvant chemotherapy by using [F-18]-Fluorodeoxyglucose positron emission tomography: Preliminary results in three patients. Gynecol Oncol. 2004;95:597-602.
- Lin LL, Yang Z, Mutic S, et al. FDG-PET imaging for the assessment of physiologic volume response during radiotherapy in cervix cancer. Int J Radiat Oncol Biol Phys. 2006;65:177-181.
- Dimitrakopoulou-Strauss A, Strauss LG, Rudi J. PET-FDG as predictor of therapy response in patients with colorectal carcinoma. Q J Nucl Med. 2003;47:8-13.
- Capirci C, Rubello D, Chierichetti F, et al. Long-term prognostic value of 18F-FDG PET in patients with locally advanced rectal cancer previously treated with neoadjuvant radiochemotherapy. AJR Am J Roentgenol. 2006;187:W202-208.
- Capirci C, Rampin L, Erba PA, et al. Sequential FDG-PET/CT reliably predicts response of locally advanced rectal cancer to neo-adjuvant chemo-radiation therapy. Eur J Nucl Med Mol Imaging. 2007;34:1583-1593. Comment in: Eur J Nucl Med Mol Imaging. 2007;34:1576-1582.
- Cascini GL, Avallone A, Delrio P, et al. 18F-FDG PET is an early predictor of pathologic tumor response to preoperative radiochemotherapy in locally advanced rectal cancer. J Nucl Med. 2006; 47:1241-1248.
- Kalff V, Duong C, Drummond EG, et al. Findings on 18F-FDG PET scans after neoadjuvant chemoradiation provides prognostic stratification in patients with locally advanced rectal carcinoma subsequently treated by radical surgery. J Nucl Med. 2006;47:14-22.
- Szyszko T,Al-Nahhas A, Canelo R, et al. Assessment of response to treatment of unresectable liver tumours with 90Y microspheres: Value of FDG PET versus computed tomography. Nucl Med Commun. 2007;28:15-20.
- Kawada K, Murakami K, Sato T, et al. Pro-spective study of positron emission tomography for evaluation of the activity of lapatinib, a dual inhibitor of the ErbB1 and ErbB2 tyrosine kinases, in patients with advanced tumors. Jpn J Clin Oncol. 2007;37:44-48.
- Zubeldia JM, Bednarczyk EM, Baker JG, Nabi HA. The economic impact of 18FDG positron emission tomography in the surgical management of colorectal cancer with hepatic metastases. Cancer Biother Radiopharm. 2005;20:450-456.
- Lejeune C, Bismuth MJ, Conroy T, et al. Use of a decision analysis model to assess the cost-effectiveness of 18F-FDG PET in the management of metachronous liver metastases of colorectal cancer. J Nucl Med. 2005;46:2020-2028.
- Ono K, Ochiai R, Yoshida T, et al. The detection rates and tumor clinical/pathological stages of whole-body FDG-PET cancer screening. Ann Nucl Med. 2007;21:65-72.
- Ide M. Cancer screening with FDG-PET. Q J Nucl Med Mol Imaging. 2006;50:23-27.
- Lee FT, Hall C, Rigopoulos A, et al. Immuno-PET of human colon xenograft-bearing BALB/c nude mice using 124I-CDR-grafted humanized A33 monoclonal antibody. J Nucl Med. 2001; 42:764-769. Comment in: J Nucl Med. 2001;42: 770-771.
- Mume E, Orlova A, Malmström PU, et al. Radiobromination of humanized anti-HER2 monoclonal antibody trastuzumab using N-succinimidyl 5-bromo-3-pyridinecarboxylate, a potential label for immunoPET. Nucl Med Biol. 2005;32:613-622.
- Robinson MK, Doss M, Shaller C, et al. Quan-titative immuno-positron emission tomography imaging of HER2-positive tumor xenografts with an iodine-124 labeled anti-HER2 diabody. Cancer Res. 2005;65:1471-1478.
- WuAM, Yazaki PJ, Tsai S, et al. High-resolutionmicroPET imaging of carcinoembryonic antigen-positive xenografts by using a copper-64- labeled engineered antibody fragment. Proc Natl Acad Sci U S A. 2000;97:8495-8500.
- Cai W, Olafsen T, Zhang X, et al. PET imaging of colorectal cancer in xenograft-bearing mice by use of an 18F-labeled T84.66 anti-carcinoembryonic antigen diabody. J Nuc Med. 2007;48: 304-310. Comment in: J Nucl Med. 2007;48:170-172.
- Griffiths GL, Chang CH, McBride WJ, et al. Reagents and methods for PET using bispecific antibody pretargeting and 68Ga-radiolabeled bivalent hapten-peptide-chelate conjugates. J Nucl Med. 2004;45:30-39.
- Sundaresan G, Yazaki PJ, Shively JE, et al. 124I-labeled engineered anti-CEA minibodies and diabodies allow high-contrast, antigen-specific small-animal PET imaging of xenografts in athymic mice. J Nucl Med. 2003;44:1962-1969. Comment in: J Nucl Med. 2003;44:1970-1972.
- Wang M, Gao M, Mock BH, et al. Synthesis of carbon-11 labeled fluorinated 2-arylbenzothiazoles as novel potential PET cancer imaging agents. Bioorg Med Chem. 2006;14:8599-8607.
- Grigsby PW, Malyapa RS, Higashikubo R, et al. Comparison of molecular markers of hypoxia and imaging with (60)Cu-ATSM in cancer of the uterine cervix. Mol Imaging Biol. 2007;9:278-283.
- Ritchie D, Mileshkin L, Wall D, et al. In vivo tracking of macrophage activated killer cells to sites of metastatic ovarian carcinoma. Cancer Immunol Immunother. 2007;56:155-163.
- Kuruppu D, Brownell AL, Zhu A, et al. Pos-itron emission tomography of herpes simplex virus 1 oncolysis. Cancer Res. 2007;67:3295-3300.
- Hung SC, Deng WP, Yang WK, et al. Mesenchymal stem cell targeting of microscopic tumors and tumor stroma development monitored by noninvasive in vivo positron emission tomography imaging. Clin Cancer Res. 2005;11:7749-7756. Erratum in: Clin Cancer Res. 2006;12(14 Pt 1):4364. Dosage error in text.
- Short S, Hoskin P, Wong W. Ovulation and increased FDG uptake on PET: Potential for a false-positive result. Clin Nucl Med. 2005;30:707.
- Subhas N, Patel PV, Pannu HK, et al. Imaging of pelvic malignancies with in-line FDG PET-CT: Case examples and common pitfalls of FDG PET. RadioGraphics. 2005;25:1031-1043.