MR in the small bowel: Applications, techniques, and imaging findings
Images


With recent advances in magnetic resonance imaging (MRI) software and hardware, and increasing expertise among technologists and radiologists, MR techniques have come to the forefront of imaging patients with known or suspected bowel pathology in both emergent and outpatient settings. MRI provides a number of advantages over more conventional techniques for imaging the small bowel. Superior tissue contrast and lack of ionizing radiation are its major advantages over computed tomography (CT) and contrast-enhanced fluoroscopy. Furthermore, with its dynamic sequences, MRI permits assessment of functional information and improved visualization of the entire bowel.1, 2 Lastly, MR imaging may also be used for patients with contraindications to contrast-enhanced CT imaging, including those who are pregnant and those with allergies or contraindications to iodinated contrast.
MRI techniques available to evaluate the small bowel include MR enterography (MRE) and MR enteroclysis. In MR enteroclysis, enteric contrast is administered directly via a nasoenteric tube, providing superior distention of small-bowel loops compared to oral ingestion in MR enterography. However, MR enteroclysis remains of limited availability, perhaps due in part to patient discomfort, and at least partially because such distention may not be necessary to evaluate for a number of pathologic processes that may affect the small bowel. As a result, this review will focus primarily on MR enterography.
MRI applications in small-bowel imaging
The general limitations of MR imaging include limited availability, variable image quality, and higher cost, lower spatial resolution, and longer acquisition times than CT. In recent years, however, a variety of advanced MR techniques have garnered great support and recognition from the radiology community in evaluating small bowel pathology. For example, current recommendations from the American College of Radiology (ACR), rate MRE with a high-priority score of 8-9 for initial evaluation of Crohn’s disease in patients with nonacute presentations, as well as for surveillance and acute exacerbation of known Crohn’s disease, in both adults and children/young adults.2, 3 Of note, MR techniques also score well in situations of acute abdominal pain in pregnant females (appropriateness score of 7).4 Lastly, it’s worth noting that MR enteroclysis has a high priority score of 8 in evaluating suspected intermittent or low-grade small bowel obstruction, on par with CT of the abdomen and pelvis with contrast, which also scores 8.5
Crohn’s disease
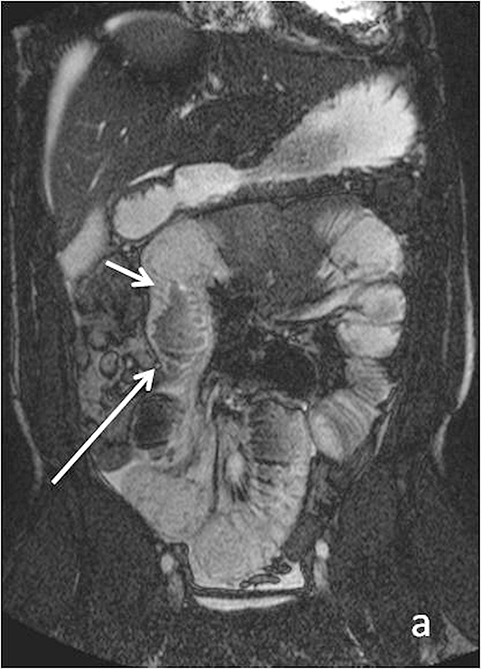
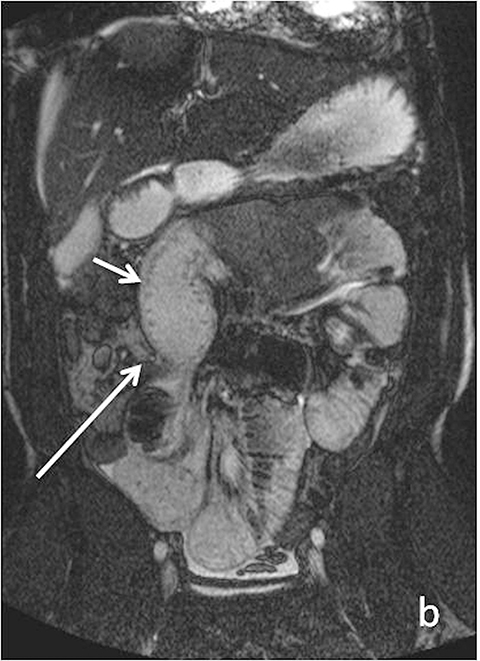
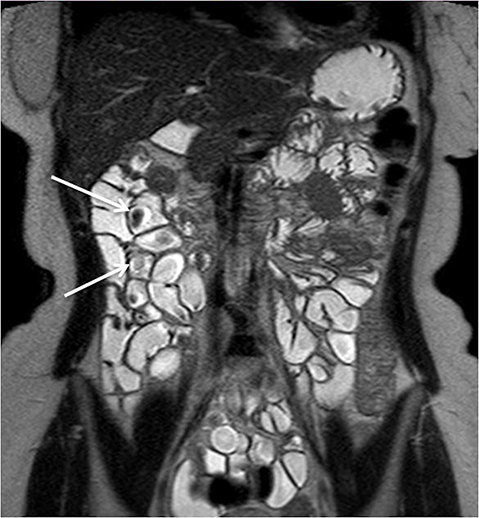
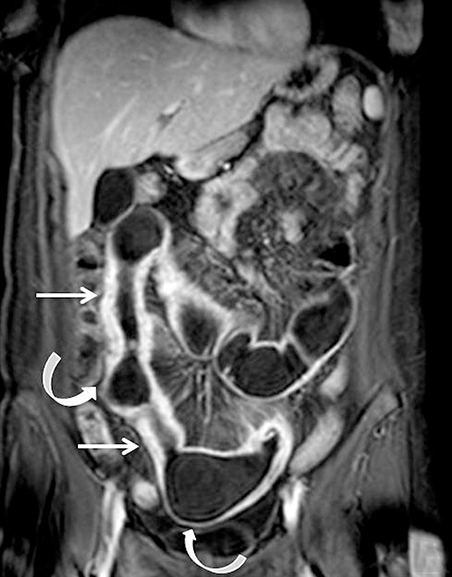
Evaluating Crohn’s disease is one of the most common indications for MRI of the small bowel. MR techniques are uniquely suited for imaging this disease, as its early age of onset and its waxing and waning nature require repeat imaging to detect complications or assess response to treatment, sparing patients potential exposure to high lifetime doses of radiation.6,7 Although many advocate for use of CT enterography as the initial study of choice to establish baseline, especially in the setting of acute abdominal pain, and this is a common practice in our own experience, MR enterography plays a major role in serial follow up and in assessing treatment response. As mentioned previously, one of the major advantages of MR enterography over CT is its ability to provide functional information about the small bowel via multiphasic dynamic sequences, which can confirm the presence of bowel stricturing, thus avoiding misinterpretation with underdistention, a common pitfall at CT imaging (Figure 1). MR techniques also allow superb evaluation of the perianal region, a frequent site of complications and fistula formation in patients with Crohn’s disease. Lastly, MRE may more accurately depict submucosal pathology given its superior soft tissue contrast, thus improving ability to distinguish between active inflammation and chronic fibrotic disease, as compared to CT enterography.8
Small bowel masses
Another application of MRE is detection and surveillance of small-bowel polyps, especially in patients with polyposis syndromes, who may require chronic surveillance. Although MR enteroclysis has been found to perform better in detecting small enteric lesions given greater luminal distention than MRE, in at least one study of 20 patients with polyposis syndromes, MRE performed similarly to capsule endoscopy in detection of larger polyps (>15mm in size), which are more clinically relevant.9 Furthermore, MR was found to be more accurate for determination of the exact size and localization of enteric polyps than capsule endoscopy.9 In addition, capsule endoscopy has limited ability to evaluate submucosal lesions, or to fully characterize the invasive nature of a small bowel tumor, while MRI can delineate lesion extent as well as localize potential regional (lymph node or mesenteric) or even distal metastases (such as to the liver). A recent study aimed at prospectively evaluating the ability of MRE to detect small bowel tumors demonstrated sensitivity and specificity of 96% on a per-patient basis, but lower sensitivity of 70% on a per-lesion basis.10
Emerging applications of MR enterography
New applications for MRE have come to the forefront. For example, a number of authors in the radiology literature have described MRI in characterizing Celiac disease.1 Imaging findings such as reversal of the jejunoileal fold pattern, which have classically been described on conventional barium fluoroscopic studies, can be well visualized at MRE. Associated findings, such as small bowel intussusception, mesenteric lymphadenopathy, and possible associated Celiac disease-related lymphoma, are also readily identifiable. MR enterography may also aid in evaluation of small bowel obstruction, especially in patients in whom it is desirable to avoid radiation exposure (children/young adults or pregnant patients).
MRE techniques
Oral contrast agents
MR enterography typically requires administration of a high volume of oral contrast material, usually on the order of 1500 to 2000 mL, to be ingested during the 2 hours preceding the examination. A number of enteric contrast agents may be used for MR imaging. These can be broadly classified into 3 major types: negative, positive, and “biphasic” contrast.11 The majority of commercially available enteric contrasts are biphasic. Biphasic contrasts demonstrate different signal intensities on T1- and T2-weighted imaging. Specifically, on T1-weighted imaging these agents demonstrate low signal intensity, thus allowing for improved contrast between bowel lumen and bowel wall, and improving detection of pathologic wall hyper-enhancement. On T2-weighted imaging, these contrast agents are high in signal intensity, allowing for delineation between the lumen and the dark bowel wall. There are a variety of commercially available enteric agents, with the preferred agent in our institution being barium sulfate (VoLumen; E-Z-Em/Bracco, Lake Success, NY). The intrinsic advantage of VoLumen is that it contains sorbitol, and thus acts as an osmotic agent, promoting luminal water retention1.
Negative contrast agents have low signal intensity on T1- and T2-weighted imaging, and include superparamagnetic particles. On T2-weighted images this provides the benefit of improved visualization of mural inflammatory high signal. This also allows for increased recognition and detection of interloop abscesses.2 An example of a negative contrast agent is ferumoxsil oral suspension.
Unlike negative oral suspensions, positive enteric contrast agents are paramagnetic substances, which produce high signal intensity on both T1- and T2-weighted sequences. Although this may have the advantage of demonstrating bowel wall thickening on T1-weighted images, the drawback is potential masking of mural enhancement abnormalities.
Spasmolytics
Spasmolytics may be useful in MRE to reduce motion artifact related to bowel peristalsis, and therefore improve lesion detection. Motion-related artifacts are most problematic for fast gradient echo sequencing obtained after intravenous contrast administration, where they can reduce lesion detection. Decreased peristalsis is also helpful for optimization of images obtained by half-Fourier acquisition single-shot turbo spin-echo technique, which may otherwise be fraught with intraluminal flow artifacts. Glucagon is a commonly used spasmolytic agent.12
Intravenous contrast
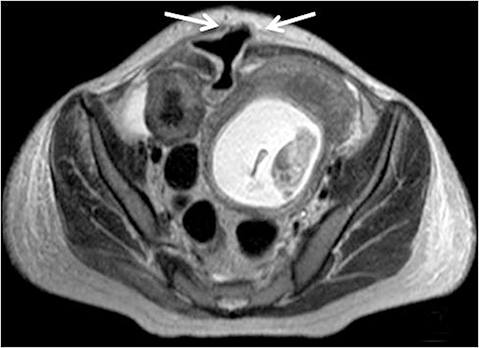
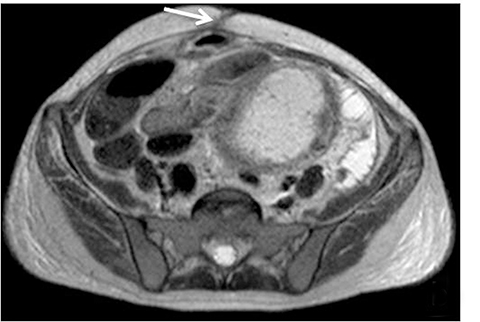
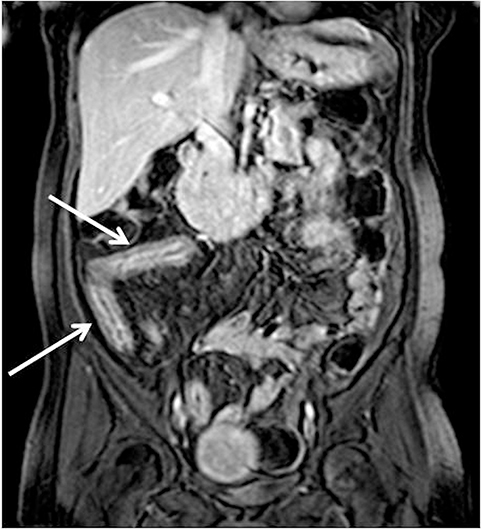
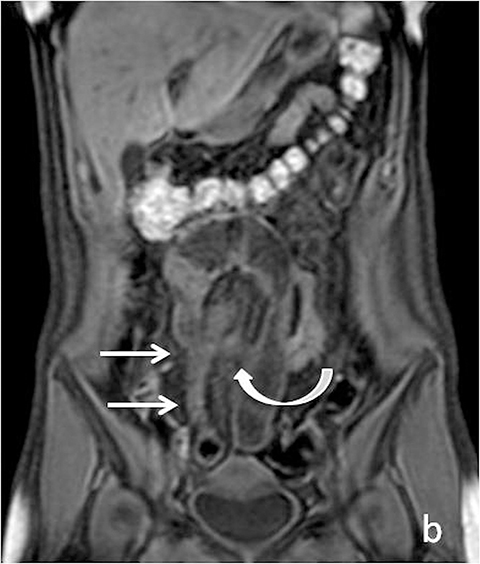
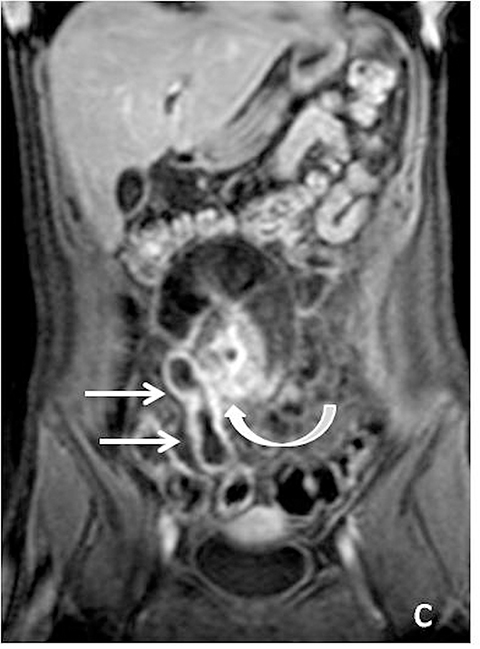
With administration of IV contrast, pathologic findings such as hyperenhancement of the bowel wall or enhancing mural/endoluminal mass lesions can be made more conspicuous.13 Intravenous contrast is not absolutely crucial to localizing small bowel pathology, as the excellent soft-tissue contrast resolution of MRI may be sufficient to delineate the small bowel abnormality. This may be especially useful in patients with contraindications to gadolinium-based contrast agents, such as patients with severe renal failure or those who are pregnant (Figure 2). However, in patients with Crohn’s disease, although regions of bowel wall thickening, stricturing, and mural edema may be detected without gadolinium chelates, some of the earliest signs of active disease such as mucosal hyperenhancement may be missed. Complications in Crohn’s disease, including abscess or fistula formation, are also made more conspicuous on post-gadolinium imaging.
Sequences
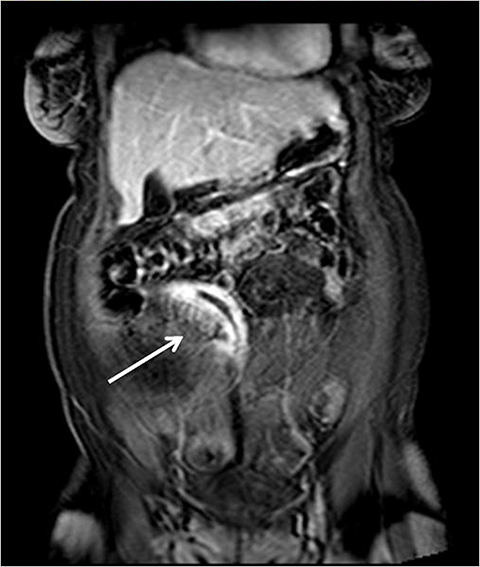
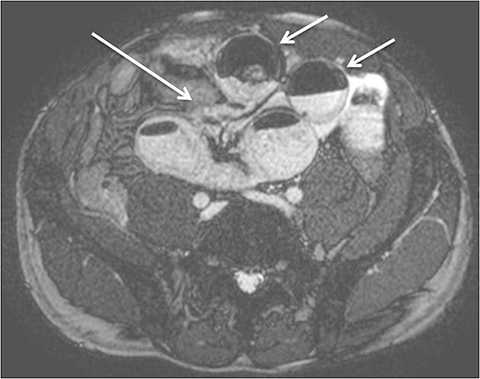
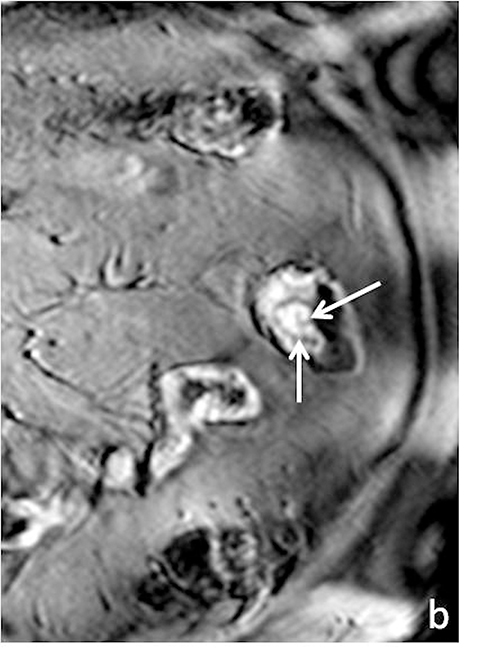
Although there is no single universal protocol for optimal imaging of small bowel, it is generally accepted that T2-weighted and multi-phasic contrast-enhanced gradient-echo sequences are particularly useful. Specifically, we find single-shot half-Fourier T2-weighted turbo spin-echo sequences (SSFSE, SSTSE, or HASTE, depending on vendor) to be helpful, especially with biphasic oral contrast, as they provide superb spatial resolution, good T2-weighting, and fast scan times.14 These sequences can be used to obtain an overview of the entire bowel and to localize pathology.15 One drawback of these sequences, however, as mentioned previously can be dark signal voids in fluid-filled bowel due to peristalsis, especially if spasmolytics are not used (Figure 3). To counteract the problem of bowel signal voids, balanced steady-state free precession sequences (FIESTA, balanced FFE, depending on the vendor) can be useful adjuncts as they have very rapid acquisition times, and also can be used to create cine loops which can depict bowel motility.15 Lastly, we obtain coronal T1-weighted dynamic, 3D fat-suppressed gradient echo sequences with and without gadolinium, to evaluate for regions of inflammation, peri-enteric complications, and possible regions of bowel stricture. Image subtraction sets may be created for each of the post-contrast phases (eg, arterial, venous or equilibrium), with areas of enhancement better delineated by the high signal remaining after the digital subtraction of the pre-contrast sequence from a post-contrast sequence. Subtraction images are useful for identifying equivocal enhancement, or for further characterizing known regions of abnormal enhancement.
Imaging findings
Crohn’s disease
Crohn’s disease is a chronic granulomatous inflammatory disease of the gastrointestinal tract that can involve any portion of the tract but is most frequently encountered in the distal small bowel. It is characterized by ulcerations, which may be superficial (aphthous ulcers which are confined to the mucosa) or deep (break through the mucosa), involving all layers of the bowel wall, resulting in penetrating and fistulizing manifestation of the entity. Segmental distribution with classic “skip lesions” is typical.15, 16
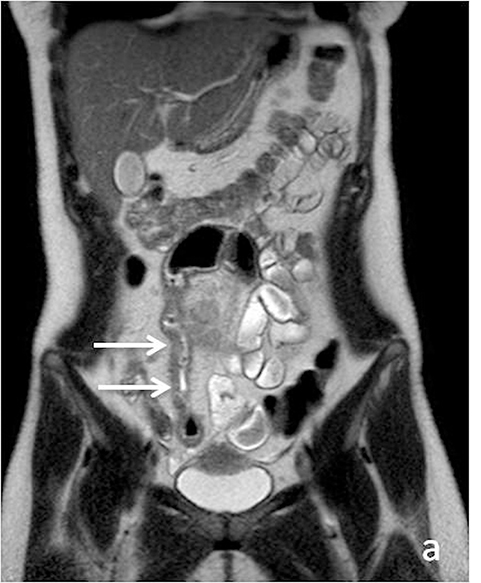
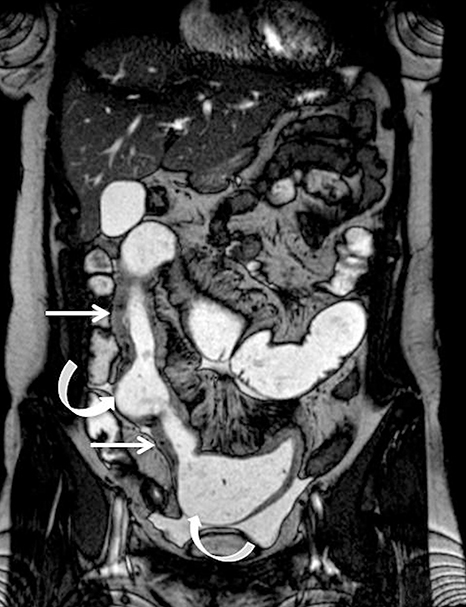
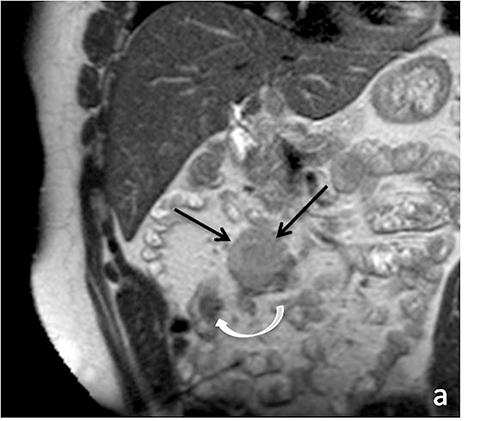
Identifying active inflammation is imperative for guiding clinical management algorithms for Crohn’s disease. While active disease may be treated medically with systemic steroids or immunomodulators, chronic disease may require surgery.17 Therefore, discerning active flare from chronic fibrostenotic disease is essential. Subtle mucosal hyperenhancement can be one of the earliest signs of active Crohn’s flare (Figure 4). In more severe active inflammation, small bowel may demonstrate classic “stratified” appearance, as a result of submucosal edema. Serosal hyperenhancement may also be seen.
Once uniform enhancement of all layers of small bowel is achieved, discerning active inflammatory bowel disease from chronic fibrostenotic disease may become more challenging. Searching for secondary signs of regional inflammation, such as increased mesenteric vascularity (“comb sign”) or perienteric fluid or enhancement can prove particularly fruitful in identifying regions of active inflammation (Figure 5).8 Wall thickening (>3mm) is also a nonspecific finding, and care must be taken not to confuse underdistention for thickening. Evaluation for wall enhancement or edema high signal on fat-suppressed T2-weighted images would indicate the presence of active disease. When reporting, it is critical to describe the number, length, and locations of the segments involved, as this can guide surgical planning. In at least one study, MR measurements of the length of abnormal segments of small bowel were found to be accurate compared with anatomic measurements.18
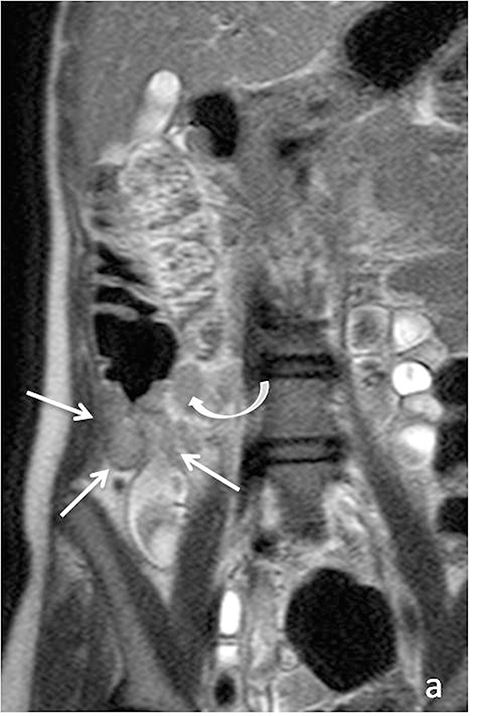
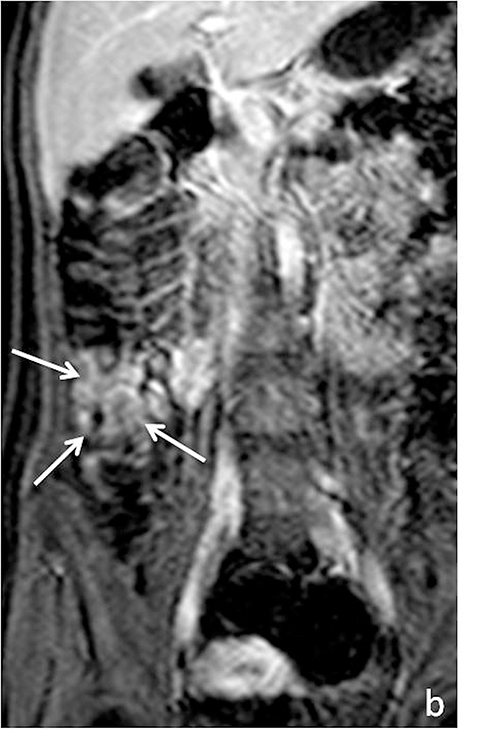
As on CT, evaluating for complications of Crohn’s disease, which may include fistulizing/penetrating disease and obstruction (Figure 6), is critical. A detailed search of multiplanar images available at MR is essential when assessing for fistulizing disease. Sensitivities for detecting complex internal fistulas on MR is reported to be around 80%,1,19-20 with T1-weighted post-gadolinium fat-suppressed imaging being the most helpful. Tethering of small bowel loops, forming a stellate appearance can be seen in the setting of complex internal bowel fistulas or inflammatory adhesions, and may result in small bowel obstruction. True fistulas and sinus tracts typically demonstrate internal fluid signal with peripheral enhancement, and thus can be differentiated from tracts of inflammation, which would show uniform enhancement. Adhesions also tend to be thinner and enhance later then fistulas (Figure 7).15
Perienteric and interloop abscesses may form, but may be difficult to detect on MR, and thorough investigation of bowel loops and the mesentery in all available planes should be undertaken. Negative enteric contrast may be especially beneficial in recognition of interloop abscesses, which may otherwise be masked by retracted, matted loops.
As discussed above, chronic fibrostenotic disease may present as mural thickening without associated active inflammation, such as mucosal hyperenhancement or wall edema.1 Evaluating dynamic sequences for persistent regions of narrowing, with proximal dilatation can identify associated stenosis. The presence of submucosal fat can also represent sequelae of chronic inflammation, although this finding is nonspecific and can be seen in normal cohorts. Other stigmata of chronic Crohn’s inflammation may present in the forms of hypertrophy of the mesenteric fat and/or pseudosacculations, which is relative dilatation of the uninvolved antimesenteric wall, in the setting of a fore-shorted and fibrosed diseased mesenteric wall (Figure 8).16
Imaging mimics of IBD-related enteritis
A number of infectious, inflammatory, and treatment-related causes of small bowel inflammation can mimic Crohn’s disease on imaging. It is especially important to be mindful of these differential considerations at MR, as not all small-bowel thickening or inflammation on MRE is necessarily Crohn’s disease. Inflammation and potentially ischemia due to vasculitidies, such as systemic lupus erythematosus, polyarteritis nodosa, and Henoch-Schonlein purpura, may present as variable segments of wall thickening, potentially with submucosal edema, and when ischemia is present may exhibit decreased wall enhancement.21 Radiation-induced enteritis also has nonspecific imaging findings on MR depending on the time elapsed from treatment, but involvement of noncontiguous, geographically adjacent loops of small bowel at the targeted location of the radiation field should be sufficient to suggest the diagnosis. Finally, several typical and atypical infectious entities may affect the small bowel. Tuberculous involvement of small bowel has a predilection for involving the terminal ileum, with contiguous involvement of the ileocecal valve and the cecum/ascending colon, and can thus mimic Crohn’s disease given that distribution (Figure 9). Adjacent necrotic lymphadenopathy may be seen and may support tuberculosis as the diagnosis.21
Tumors of the small bowel
Superior soft-tissue contrast of MRI in conjunction with presence of enteric contrast for bowel distention has proven to be useful in evaluation for small bowel lesions (Figure 10). This is especially pertinent for patients with polyposis syndromes. Biphasic enteric contrast helps increase the conspicuity of mass lesions. Small bowel lesions demonstrate variable enhancement on post-gadolinium imaging. They are frequently also relatively lower in T2 signal intensity as compared to surrounding luminal fluid, but caution must be taken not to rely solely on these sequences, as luminal flow voids related to peristalsis my mask these lesions.
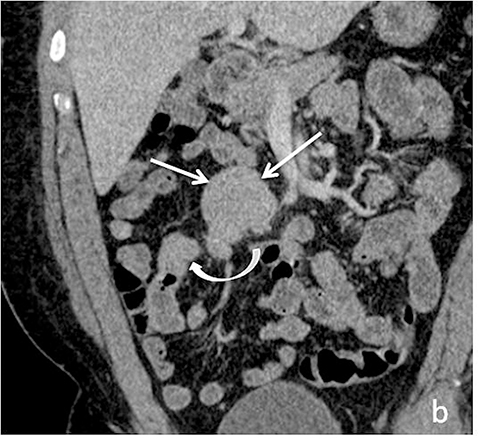
A variety of neoplastic and benign lesions can be found in the small bowel, each exhibiting variable imaging characteristics. Benign lesions such as lipomas can readily be characterized with MR given the hyperintense signal on T1-weighted imaging and loss of this high signal with fat-suppression techniques. Small bowel hemangiomas classically demonstrate high signal intensity on T2-weighted imaging. Adenomas, which are the most common asymptomatic benign tumor of the small bowel, can present as a pedunculated or sessile soft tissue mass, with moderate homogeneous enhancement (Figure 11).21 Malignant tumors such as primary adenocarcinomas and carcinoid tumors, may also be encountered at MRI.
Lymphomatous involvement of the small bowel is associated with nonspecific findings and can be confused for inflammatory or infectious enteric disease. At least one study attempted to use MRE as a means to suggest histologic subtypes of lymphoma based on morphologic imaging findings.22 For instance, luminal stricture was found to be associated with low-grade lymphoma, while propensity for aneurysmal dilatation of involved segment of small bowel had a higher association with celiac related non-Hodgkin’s lymphoma. Mesenteric involvement and infiltration were more characteristic of high-grade lymphoma.22
Conclusions
Over the last decade the use of MR techniques in evaluating small bowel pathology has expanded greatly, and this continues to evolve as more versatile uses are being recognized. At this time MRE plays a critical role in surveillance of patients with established Crohn’s disease and in follow up after medical treatment, sparing patients repeat radiation exposure, which is inherent in CT imaging. Additional applications of MRE include evaluation of small bowel benign and neoplastic lesions, and evaluation of non-inflammatory small bowel entities such as Celiac disease. Additional applications are sure to come as MR techniques continue to advance.
References
- Masselli G, Gualdi G. MR imaging of the small bowel. Radiology. 2012;264(2):333-348.
- Fidler JL, Guimaraes L, Einstein DM. MR imaging of the small bowel. Radiographics. 2009;29(6):1811-1825.
- Kim DH, Carucci LR, Baker ME. Crohn’s Disease. ACR Appropriateness Criteria 2014. American College of Radiology Web site. http://www.acr.org/.
- Yaghmai V, Rosen MP, Lalani T, et al. ACR Appropriateness Criteria 2012. American College of Radiology Web site. http://www.acr.org/.
- Katz DS, Baker ME, Rosen MP, et al. ACR Appropriateness Criteria 2013. American College of Radiology Web site. http://www.acr.org/.
- Horsthuis K, Bipat S, Bennink RJ, et al.. Inflammatory bowel disease diagnosed with US, MR, scintigraphy, and CT: meta-analysis of prospective studies. Radiology. 2008 ;247(1):64-79.
- Schreyer AG, Seitz J, Feuerbach S, et al. Modern imaging using computer tomography and magnetic resonance imaging for inflammatory bowel disease (IBD) AU1. Inflamm Bowel Dis. 2004;10(1):45-54.
- Arif-Tiwari H, Kalb B, Chundru S, et al. The diagnostic role of magnetic resonance enterography in Crohn’s disease: An updated review of techniques, interpretation, and application. Appl Radiol. 2013;42(12):5-15.
- Caspari R, von Falkenhausen M, Krautmacher C, et al. Comparison of capsule endoscopy and magnetic resonance imaging for the detection of polyps of the small intestine in patients with familial adenomatous polyposis or with Peutz-Jeghers’ syndrome. Endoscopy. 2004;36(12):1054-1059.
- Amzallag-Bellenger E, Soyer P, Barbe C, et al. Prospective evaluation of magnetic resonance enterography for the detection of mesenteric small bowel tumours. Eur Radiol. 2013;23(7):1901-1910.
- Laghi A, Paolantonio P, Iafrate F, et al. MR of the small bowel with a biphasic oral contrast agent (polyethylene glycol): technical aspects and findings in patients affected by Crohn’s disease. Radiol Med. 2003;106(1-2):18-27.
- Grand DJ, Beland M, Harris A. Magnetic resonance enterography. Radiol Clin N Am. 2013;(51): 99-112.
- Pupillo VA, Di Cesare E, Frieri G, et al. Assessment of inflammatory activity in Crohn’s disease by means of dynamic contrast-enhanced MRI. Radiol Med. 2007;112(6):798-809.
- Lebedis CA, Penn DR, Broder JC, et al. Current applications of MRI in emergent gastrointestinal disease. Appl Radiol. 2012;41(11):9-15.
- Leyendecker JR, Bloomfeld RS, DiSantis DJ, et al. MR enterography in the management of patients with Crohn’s disease. Radiographics. 2009;29(6):1827-1846.
- Tolan DJ, Greenhalgh R, Zealley IA, et al. MR enterographic manifestations of small bowel Crohn’s disease. Radiographics. 2010;30(2):367-384.
- Lauenstein TC. MRI of inflammatory bowel disease. Appl. Radiol. 2008;37(7):19-24.
- Sinha R, Trivedi D, Murphy PD, et al. Small-intestinal length measurement on MR enterography: comparison with in vivo surgical measurement. AJR Am J Roentgenol. 2014;203(3):W274-9.
- Schmidt S, Chevallier P, Bessoud B, et al. Diagnostic performance of MRI for detection of intestinal fistulas in patients with complicated inflammatory bowel conditions. Eur Radiol. 2007;17(11):2957-2963.
- Fallis SA, Murphy P, Sinha R, et al. Magnetic resonance enterography in Crohn’s disease: a comparison with the findings at surgery. Colorectal Dis. 2013;15(10):1273-1280.
- Amzallag-Bellenger E, Oudjit A, Ruiz A, et al. Effectiveness of MR enterography for the assessment of small-bowel diseases beyond Crohn’s disease. Radiographics. 2012;32(5):1423-1444.
- Lohan DG, Alhajeri AN, Cronin CG, et al. MR enterography of small-bowel lymphoma: potential for suggestion of histologic subtype and the presence of underlying celiac disease. AJR Am J Roentgenol. 2008;190(2):287-293.
Citation
K M, C C, JA S.MR in the small bowel: Applications, techniques, and imaging findings. Appl Radiol. 2016; (6):24-20.
June 6, 2016