Coming to a radiology department near you: The latest and greatest in technology
Images





It may seem like just yesterday, but it’s already been more than three months since Richard L. Baron, MD, looked out on a theater full of radiologists at the 102nd scientific meeting of the Radiological Society of North America (RSNA).
There, the current President of the RSNA called upon his audience to look beyond their own imaging realm to provide value in a healthcare era of declining reimbursements by striving to work collaboratively with other providers, to stay focused on patients, and to continue helping to drive innovation.
Dr. Baron recommended that radiologists consider taking a page out of their referring physicians’ book and sub-specialize in specific anatomical areas, such as abdominal, neuro or musculoskeletal images. He also implored them to develop a better understanding of the clinical answers that patients and referrers are seeking. The ultimate goal for radiologists, he said, should be to provide more meaningful consultations and better solutions.
New technology can go a long way toward helping radiologists achieve those goals. As always the RSNA 2016 exhibit hall was flush with new technology aimed at improving medical imaging and its surrounding workflows.Picking up where we left off in January, here’s a sampling of major modality offerings:
Breast Imaging/Breast Biopsy
Hologic introduced its Affirm prone breast biopsy system—reportedly the first dedicated prone biopsy system that is upgradable to allow for 3D image-guided breast biopsies. In addition to the 3D biopsy capability, Affirm also delivers 360-degree access to the breast. For the Affirm upright biopsy system, Hologic launched the Affirm lateral arm biopsy accessory. This versatile, easy-to-use device enables needle access parallel to the detector, allowing physicians to target challenging lesions confidently and successfully biopsy thinly compressed breasts.
The company also previewed the Brevera breast biopsy system with CorLumina imaging technology, which is currently pending FDA clearance. According to Hologic, it is the first and only system that offers clinicians breast biopsy and real-time verification, designed to streamline workflow, improve procedure speed and have a positive impact on the patient experience.
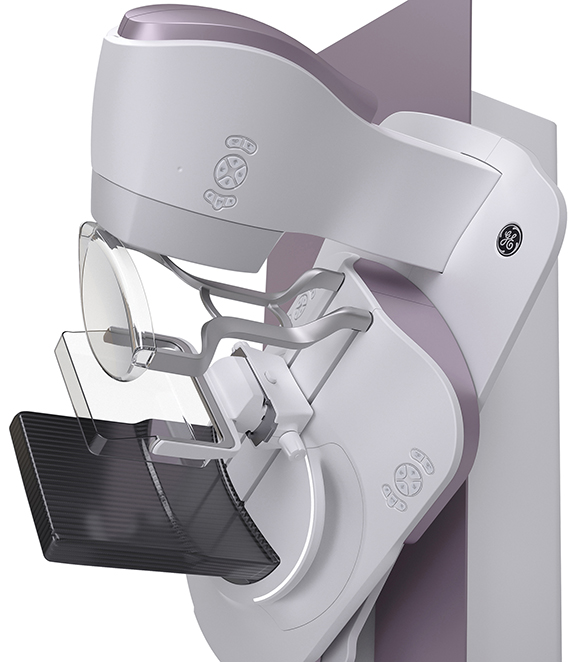
Senographe Pristina from GE Healthcare features a 510(k)-pending self-compression tool that enables patients to adjust the amount of pressure and breast compression with the help of a technologist. On the Bucky, the corners are rounded to reduce patient discomfort. Instead of women tensing their pectoral muscles while grabbing the conventional handgrips, which can make it hard to acquire pristine images, they can lean comfortably on armrests, relaxing the muscles to simplify positioning, compression and image acquisition.
Computed Tomography
A new premium CT and model-based iterative reconstruction (MBIR) were highlighted in Toshiba America Medical Systems’ RSNA booth. Aquilion ONE/GENESIS Edition also introduces PUREViSION Optics, PUREViSION Detector and the recently FDA-cleared FIRST MBIR. The system boasts reconstruction times of up to 80 images per second and FIRST reconstruction achieved in as little as three minutes per volume (320 images). The system is small, light and consumes less power, fitting into a footprint of only 204 square feet (19 square meters) with a short bore, flared design and 30-degree tilting gantry. PUREViSION Optics with FIRST body mode reportedly improves high contrast spatial resolution up to 129 percent (2.29 times), while reducing radiation dose up to 85.3 percent compared to filter-back projection reconstruction on the company’s Aquilion ONE ViSION Edition. FIRST can be integrated into scan protocols adapting to the individual patient and clinical task at hand, requiring as little as three minutes for volumetric reconstructions.
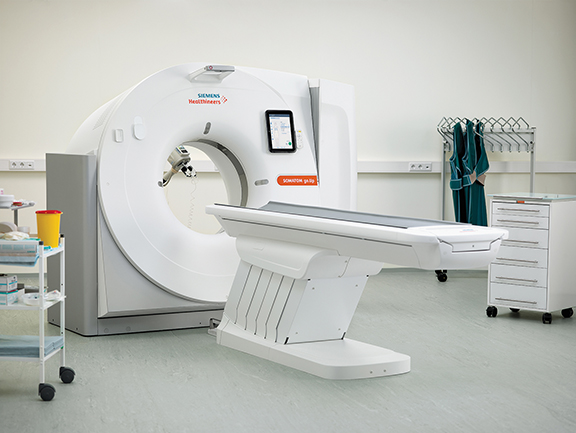
Siemens Healthineers launched a new CT platform, the SOMATOM go., featuring the SOMATOM go.Now, a 32-slice system, and SOMATOM go.Up equipped with a wider detector that provides up to 64 slices. Automated, standardized workflows along with multi-year service packages are designed to help users run their business successfully, the company said. Developed with input from more than 500 radiologists, the platform enables all routine examinations to be controlled via a tablet for a mobile workflow. The standardized work steps and automated post-processing facilitates scanner operation and enables users to run the scan with just a few inputs. The mobile workflow provides an option for a single-room concept with a shielded niche for the radiology staff.
Revolution CT with Gemstone Spectral Imaging (GSI) Xtream was introduced by GE Healthcare. The system, pending FDA 510(k) clearance, is designed to be improve small lesion detection, tissue characterization and metal artifact reduction along with a simplified workflow to help clinicians make it part of their daily practice. According to the company, with the additional information GSI Xtream can provide, clinicians can have more confidence in diagnosing disease, can potentially avoid additional studies, and ultimately put patients on the right treatment path sooner.
Carestream has begun accepting orders and shipping the OnSight 3D Extremity System, the company’s cone beam CT solution for weight bearing and other types of extremity exams. The system is targeted to orthopedic and sports medicine practices, hospitals, imaging centers and urgent care facilities.
Digital Radiography (DR)
AGFA Healthcare introduced the 510(k)-pending DR 800 X-ray room with Dynamic Musica for radiography and fluoroscopy applications in supine and upright positions. The DR 800 can handle a full range of radiographic exams (skeletal, thorax, abdomen, weight bearing) and fluoroscopy exams (including barium studies, arthrograms, cystograms, myelography and catheter placement, etc.). It comes with MUSICA for multi-scale image processing including moving images, enhanced noise suppression and brightness control, and reduced veiling glare.
The company also expanded its DR retrofit portfolio with the DX-D 60, a DR detector that fits into existing radiography rooms and workflow. With the 17x17 inch panel, technologists can capture landscape or portrait images without having to rotate the plate. It is available with either gadolinium- (GOS) or cesium- (CsI) based scintillators.
A new DR tablet solution has emerged from Canon USA, Inc. and its wholly-owned subsidiary Virtual Imaging, Inc. The solution comprises a tablet and choice of a Canon wireless CXDI Digital Radiography System includes CXDI Control Software NE with its optional new scatter correction feature. The CXDI Control Software NE features an auto-detection mode that allows the CXDI detectors to automatically detect X-rays for exposure without the need for a typical X-ray generator interface and cables. An optional scatter correction for grid-free imaging was also introduced on the CXDI Control Software NE. Also new is the RadPRO SOLTUS 100 Mobile Digital X-ray System which, when used with a compatible Canon CXDI digital detector, provides short exposure times and fast imaging acquisition. Easily folded for transportation and storage, it can serve as an ideal solution for ambulatory services and surgery suites, as well as humanitarian organizations.
Using carbon nanotube technology, the work-in-progress DRX-Revolution Nano Mobile X-ray system from Carestream delivers a nonmotorized imaging solution weighing just 205 lbs. The compact design includes an articulating arm and rotation column and independent controls of the diaphragm without moving the tubehead. An expanded detector portfolio now includes the CARESTREAM DRX Plus and CARESTREAM DRX Core detectors that offer rapid image capture and achieve IPX Level 57 rating for liquid resistance to the IEC standard 60529. The DRX Core portfolio includes wireless gadolinium and cesium detectors in 35 × 43 cm and 43 × 43 cm sizes.
FUJIFILM Medical Systems USA, Inc., unveiled the FDR AQRO, a works-in-progress mobile DR system that brings dose saving features to an ultra-lightweight point-of-care mini mobile cart. It offers the same low-dose capabilities as the company’s FDR D-EVO II, ISS and noise reduction circuitry as well as Virtual Grid image processing software for grid-less imaging. An integrated touchscreen monitor and X-ray control panel articulates and swivels for viewing from virtually any; the system can be used for up to 12 hours on its lithium batteries.
Konica Minolta Healthcare Americas, Inc., introduced its next-generation DR wireless detector, AeroDR HD, which delivers 100-micron pixel size for a 3488 × 4256 pixel count the company says is four times higher than standard resolution. Measuring 14 × 17 inches and weighing 5.7 lbs., the detector’s new ergonomic design has a high drop, high load and high bend resistance in a protective waterproof enclosure. AeroDR HD boasts two pre-programmable modes for optimal flexibility: the very high resolution selection for detailed views of small areas such as extremities, and a wide dynamic range mode for larger areas. Using pixel binning, the high dynamic range mode aggregates the data to provide faster processing and more contrast for the anatomical areas that demand that type of image.
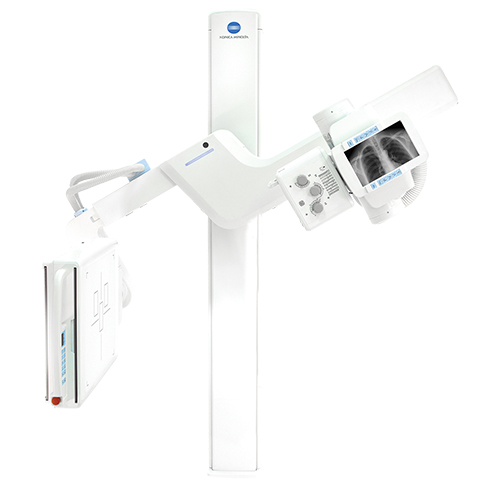
Also released by the company was a new digital U-arm, the KDR AU System, that includes pre-programmed positioning capability and floor-mounted installation in a small footprint. Using the company’s Ultra software, anatomy selections can be preprogrammed into the system so only minor adjustments are required once the patient enters the room. Multiple images may be preprogrammed as well, resulting in less demands and less stress for the patient throughout the exam. The floor-mounted system fits 8-foot ceilings, with a stand that lowers to less than 1 foot from the floor for easy positioning and efficient use of space. The technologist can accept and reject images right at the patient’s side and even add patients to the worklist on the fly.
Interventional Radiology
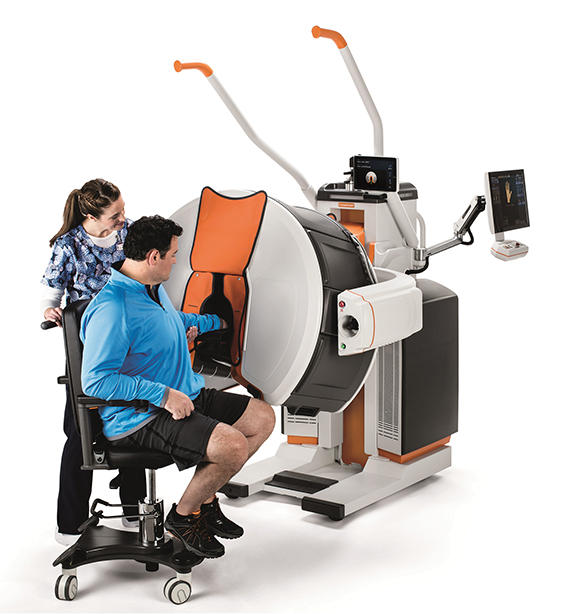
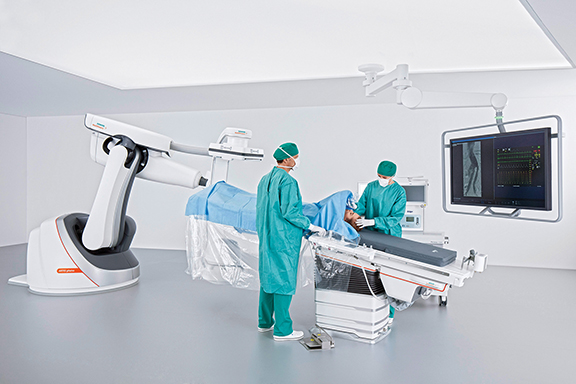
Siemens Healthineers unveiled the robot-supported ARTIS pheno angiography system, currently pending US FDA 510(k) clearance, developed for use in interventional radiology, minimally invasive surgery, and interventional cardiology. In addition to possessing a zen40HDR flat panel detector and GIGALIX X-ray tube for high image quality, the ARTIS pheno boasts new 2k recording technology capable of delivering 2D imaging resolution that is four times higher in all recording processes than prior Siemens Healthineers systems.
The system’s StructureScout feature can adapt and optimize imaging parameters to suit the X-rayed area, potentially resulting in less radiation dose than prior Siemens Healthineers systems. The ARTIS pheno also supports the treatment of multimorbid patients and can be fitted with a comprehensive range of optional software applications for complex cases. Large, sealed surfaces with fewer spaces and the lack of any moving parts on the ceiling and floor helps support infection control and prevention efforts, and also makes the system easier to install in an operating suite. An antimicrobial coating prevents bacteria and viruses from multiplying on the system. A new double-sliding C-arm on Toshiba Medical’s Infinix –I Sky+ will allow clinicians to position the detectors for 210 degrees of anatomical coverage on both the right and left side of the patient. The system’s C-arm can also be positioned laterally over or under the table for increased flexibility in long needle biopsies, aspirations and vertebroplasty procedures. The system also boasts a high-speed 3D rotation of 80 degrees per second, supports low contrast imaging from the head-end or either side of the patient, and easy head-end access with the system’s Access Halo 270-degree C-arm rotation.
Magnetic Resonance Imaging (MRI)
GE Healthcare announced a new magnet technology designed to consume 1% of liquid helium compared to conventional MRI systems. Systems with Freelium technology, a work-in-progress, would be less dependent on helium, easier to site without the need for extensive venting, and eco-friendly as a Freelium magnet would not need any refilling during transportation or throughout its lifetime. The company also believes the technology could help make MRI more accessible, particularly in regions where access to helium is limited, or where there is a lack of infrastructure.
The company also introduced several new SIGNA MRI systems—Architect, Premier (technology in development), Voyager and Artist—along with SIGNAWorks, GE Healthcare’s productivity platform of standard applications and advanced tools, such as HyperSense for hyper scanning.
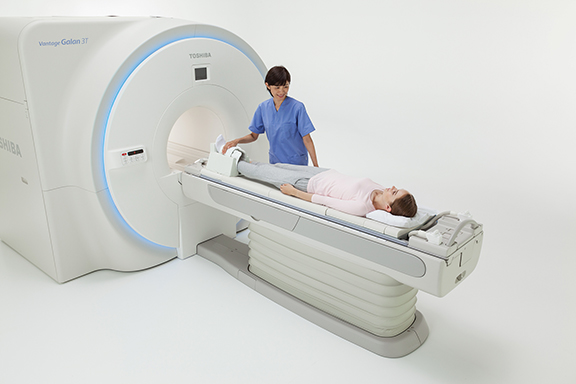
Toshiba America Medical Systems unveiled the Vantage Galan 3T premium MR solution, designed to increase signal-to-noise ratio (SNR) by up to 20%, allowing for faster sampling and higher resolution images. The system, which is pending 510(k) clearance, can also stack protocol sequences, which may enable a neuro exam using SAG T1, AX T2, AX T2 FLAIR, AX T2* and AX DWI/ADC to be performed in less than five minutes. Additionally, Pianissimo Zen technology reduces sound during acquisition by 99% (loudness level dB) and the 71 cm bore expands access to bariatric and claustrophobic patients.
There is a heightened focus on cardiac MR imaging by manufacturers and clinicians, and Toshiba answered with a new technology intended to simplify cardiac exams. The Vantage Titan 1.5T / cS Edition with M-Power includes: the optional SUREVOI with CardioLine+ anatomy recognition technology to automatically determine the position of the heart and detect anatomical landmarks for accurate and reproducible positioning to complete alignment of 14 standard cardiac views with minimal operator interaction; the acquisition of multiple b-values in a single diffusion scan; Multi-echo T2 Mapping; and Vitrea Extend, an optional post-processing application that allows clinicians to complete post-processing at a separate workstation, freeing up the MR system for the next exam.
Taking on the need for speed in MRI, Siemens Healthineers introduced Compressed Sensing technology, which promises to shorten the long acquisition times. With Compressed Sensing, cardiac cine imaging can be performed in 16 seconds rather than the traditional four minutes, thanks to an algorithm that reduces the amount of data required. Using Compressed Sensing, only key data points are need to provide a diagnostic-quality MR image and data is calculated at the MRI scanner. Iterative reconstruction is used to deliver high-resolution, high-quality images with no information loss.
The company also announced the 510(k)-pending MAGNETOM Sempra, a 1.5T scanner featuring DotGO MRI imaging software that helps users standardize exams yet provides flexibility to adapt to the case and patient condition.
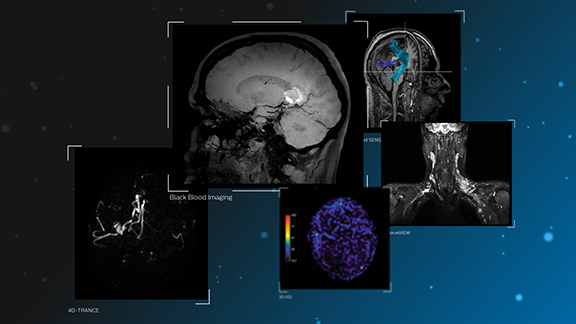
For the Ingenia family of digital MRI systems, Royal Philips unveiled new diagnostic applications aimed at neurodegenerative diseases. Utilizing Philips’ dStream digital broadband architecture, they include: Black Blood Imaging for 3D high-resolution brain images with reduction of the blood signal compared to a 3D T1w scan without MSDE pre-pulse; 4D-TRANCE for contrast-free dynamic (4D) imaging of the brain vascular anatomy; and MultiBand SENSE, which allows simultaneous acquisition of multiple slices in the brain in fMRI exams for accelerated neurofunctional scans. The apps will integrate with Philips’ IntelliSpace Portal, providing radiologists with access to the latest clinical capabilities covering their advanced visual analysis needs on a single platform.
Radiology Operations
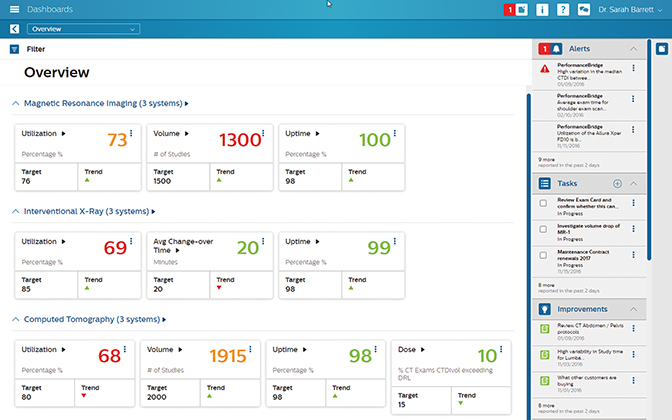
Philips introduced PerformanceBridge, a suite of performance-management software and services to assist radiology departments in enhancing productivity, improving the patient experience and delivering better value-based care. A secure, cloud-based or on-premise software solution, Philips’ PerformanceBridge gives clinicians access to near-real-time data on departmental performance through an interactive dashboard. Vendor agnostic, PerformanceBridge unifies information across different imaging modalities and existing information systems. Among its tools: imaging performance benchmarks, dose management through the company’s DoseWise Portal, knowledge sharing through NetForum, and services and equipment analytics.
Ultrasound
A partnership between Mindray and Konica Minolta Healthcare America, Inc., has led to an expanded MSK ultrasound portfolio and the launch of the J5 Ultrasound System. Developed for use at the point of care, the J5 features a touchscreen and intuitive workflow with focused MSK exams making it well-suited for evaluations in sports medicine, orthopedics and rheumatology that require needle visualization for ultrasound-guided injections and biopsies. Users touch an MSK exam preset icon on the screen for frequently used functionality and one-touch optimization sharpens the image for a clear view to aid in diagnosis. The new system also includes built-in wireless communications and a two-plus hour battery.
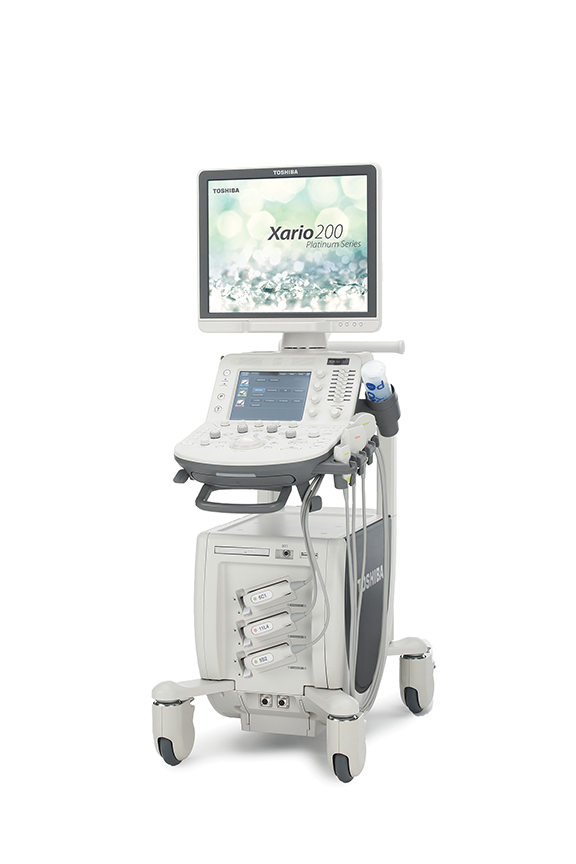
The new Xario 200 Platinum Series from Toshiba America Medical Systems delivers advanced imaging and high-end ultrasound technology in a small footprint. It includes technologies such as Precision Imaging, Differential Tissue Harmonics (D-THI) and Advanced Dynamic Flow (ADF). The new system comes with the added visualization benefits of Toshiba Medical’s Superb Micro-vascular Imaging (SMI), which provides excellent visualization of low velocity microvascular flow. The system also offers a customizable panel, touch command screen, and one-button optimization and clinical parameter setting with Toshiba Medical’s Quick Scan and Quick Start technologies. For ultrasound-guided interventions, CIVCO Medical Solutions introduced the Verza guidance system. A key feature of the Verza system is its direct transducer attachment in combination with VerzaLink, a proprietary locating feature designed onto the ultrasound transducer, eliminating the need for a custom bracket attachment and providing an ideal profile and position on the ultrasound probe. The system offers 14 instrument inserts, ranging in size from 25 gauge to 12 French, and a selection of five potential approach angles.
Citation
MB M.Coming to a radiology department near you: The latest and greatest in technology. Appl Radiol. 2017; (3):32-37.
March 4, 2017