Cardiac MRI: A preferred method for assessing myocardial ischemia and infarct burden
Images
During the past 20 years, cardiac MRI (CMR) has earned growing acceptance as a modality for assessing myocardial ischemia, viability, and function. Accurately assessing ischemic risk and identifying hemodynamically significant coronary artery disease amenable to revascularization can be challenging tasks with non-invasive imaging modalities. Here, we aim to discuss how cardiac MRI achieves both with a single study without exposure to ionizing radiation.
Indications for CMR for ischemic heart disease (IHD)
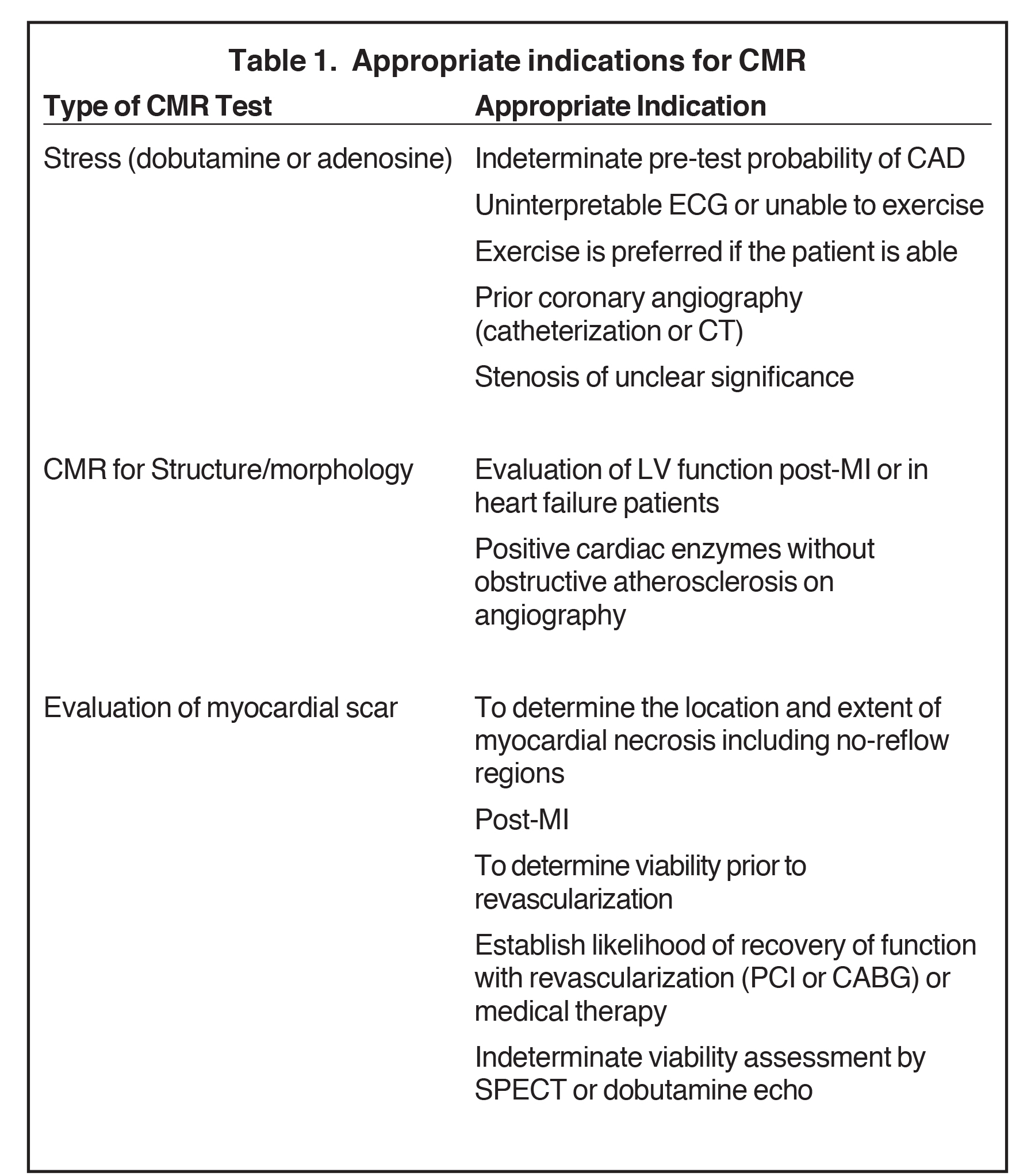
Table 1 lists the appropriate indications for the use of CMR as reported in the 2006 ACCF/ACR/SCCT/SCMR/ASNC/NASCI/SCAI/SIR appropriateness criteria for cardiac computed tomography and cardiac magnetic resonance imaging.
According to the 2012 American Heart Association guidelines,1 stress CMR can be useful for patients for initial diagnosis with an intermediate-to-high pretest probability of obstructive IHD who have an uninterpretable ECG and at least moderate physical functioning or no disabling comorbidity (class II Level of Evidence: B). Stress CMR is at the same level of recommendations as pharmacologic stress echocardiography or SPECT. In particular, CMR is recommended as the initial test for IHD diagnosis in patients with resting ECG abnormalities which prevent accurate interpretation of ECG changes during stress (IB), in patients with a non-conclusive exercise ECG (IB), in patients with known IHD and deterioration in symptoms if the site and extent of ischemia would influence clinical decision making (IB), and for re-assessment in patients with stable IHD in the presence of recurrent or new symptoms once instability has been ruled out (IC).1
Stress imaging
Principle of stress imaging
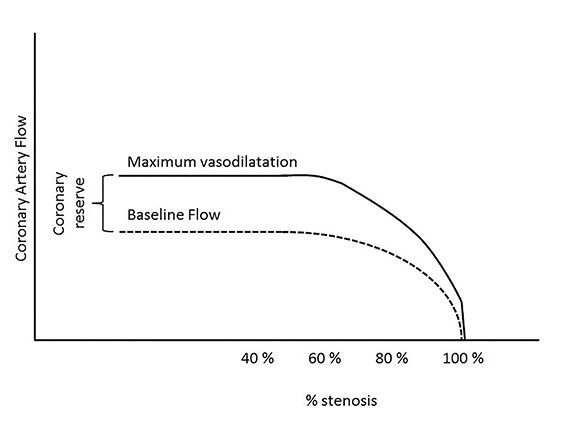
Epicardial arteries and resistance arterioles comprise the coronary blood supply. In the absence of epicardial coronary stenosis, an increased oxygen myocardial demand can be achieved by an increase in coronary flow due to arterial vasodilatation, commonly referred to as “coronary reserve,” which can increase up to four times the basal blood flow. This is shown in Figure 1. In the presence of an epicardial stenosis, as the coronaries are already dilated to maintain constant coronary flow, it is unable to augment further in the event of increased myocardial demand.2The resulting oxygen demand and supply mismatch leads to myocardial ischemia at rest if blood flow is insufficient at basal conditions and ischemia on stress if an increased oxygen demand exceeds oxygen supply.
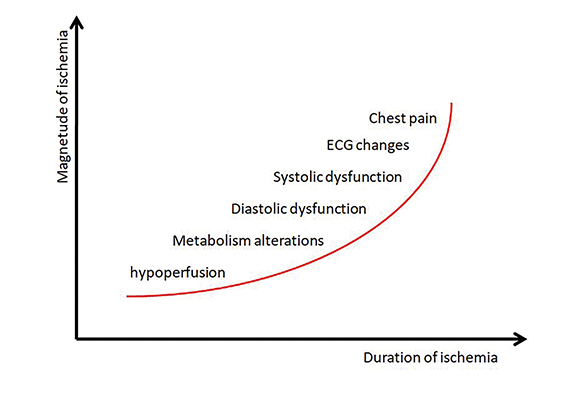
Myocardial ischemia involves a typical sequence of events termed the “ischemic cascade,” shown in Figure 2. The cascade begins with metabolic alterations, followed first by diastolic then by systolic dysfunction. This leads to electrocardiographic changes and subsequent precordial pain.3
Myocardial ischemia can be identified by metabolic alterations detected with PET imaging, perfusion defect reversibility detected by CMR with adenosine/Lexisan, dipyridamole or dobutamine, regional wall motion abnormalities seen by echocardiography and CMR, or ECG changes detected by standard stress test treadmill.
Stress CMR agents
The most commonly used pharmacological agents for stress testing include: adenosine infusion (or regadenoson, which is administered as a bolus), dipyridamole, and dobutamine.
Coronary vasodilators—Adenosine, regadenosine, and dipyrimadole are coronary vasodilators used for assessment of myocardial perfusion with CMR. They act via different mechanisms in the adenosine pathway, with adenosine directly binding to multiple types of adenosine receptors, regadenoson selectively binding to A2 receptors, and dipyrimidole blocking the cellular re-uptake of adenosine, to result in coronary vasodilatation and subsequent increase in coronary flow. In the presence of a significant coronary stenosis, these agents induce a relative hypoperfusion in the territory of the stenosis by a steal phenomenon, typically without inducing true ischemia. Contraindications to adenosine include second- or third-degree AV block, severe bronchospasm, or use of dipyridamole (which blocks the cellular uptake of adenosine, increasing the concentration of adenosine at its receptors).
Dobutamine—Dobutamine is a sympathomimetic drug with a short half-life (2 min) that mainly acts on β1 cardiac receptors resulting in positive inotropic and chronotropic effects. It has a lesser mechanism of action on the β2 receptors with slight vasodilatory effect.4 Beta blockers are held 24 to 48 hours before the test as they counteract the effects of dobutamine.
Stress CMR technique
Continuous medical surveillance (blood pressure, ECG and SaO2 monitoring) is required during the performance of the examination.
Coronary vasodilators—adenosine, diyprimadole—The following is a commonly used protocol for stress-perfusion CMR, previously described by Klem et al. Standard balanced steady state free precession (bSSFP) breath-hold cine sequences at rest in the standard views (apical, mid, and basal short-axis views; 4, 2, and 3 chamber views) are first acquired. Stress is induced with an adenosine infusion (140 μg/kg/min; total duration: 6 mins) or after dipyridamole slow injection (0.56-0.84 mg/kg). Immediately after the intravenous injection of a contrast agent (Gadolinium: 0.1 mmol/kg), MR first-pass perfusion scanning on stress is performed using identical views and a T1 weighted sequence such as magnetization-prepared fast gradient-recalled echo (GRE) with non-selective saturation recovery preparation pulse or a balanced steady state free precession (bSSFP) sequence. Generally 3 views (apical, mid and basal short axis) can be acquired every heartbeat, with 6 views every 2 heart beats, allowing a dynamic study of the perfusion.
Standard CMR cine sequences are then performed to evaluate for wall motion abnormality. LGE sequences are then performed 10-15 mins following the initial gadolinium administration.
Stress-perfusion images may be obtained prior to rest, given their relative importance to the study and fact that rest imaging is typically unnecessary following a normal stress evaluation. If stress CMR is performed initially, adenosine is advantageous given its short half-life, as no reversal agent is necessary. With both regadenoson and dipyrimadole, reversal with aminophylline may be performed.
Dobutamine—The standard protocol begins with the acquisition of standard CINE SSFP sequences (apical, mid and basal short axis and 2, 3, 4 chamber long axis view) at rest. Dobutamine is then infused intravenously and the dosage is increased every 3 mins (10, 20, 30, 40, and 50 mg/kg per minute). A low level stress (5-10 μg/kg/minute of dobutamine) is used to assess viability whereas higher dose (up to 50 μg/kg/min) allows one to reach the theoretical maximal frequency to detect ischemia at stress. The infusion is stopped when 85% of the maximal heart rate is achieved, new or worsening wall motion abnormality develops in 1 or greater LV segments, BP increases to greater than 240/20mmHg or SBP drops >40mmHg, or if the patient develops adverse symptoms or persistent arrhythmias. At every 3 min stage, cine sequences with conventional views are repeated. Atropine (up to 2 mg) can be utilized to augment the heart rate response, although this is contraindicated in patients with narrow angle glaucoma, myasthenia gravis, obstructive uropathy, and obstructive gastrointestinal disorders. Contraindications to dobutamine include severe hypertension (>/=220/120mmHg), unstable angina, severe aortic stenosis (gradient >50mmHg or AVA <1cm2), uncontrolled atrial fibrillation, HCM, and CHF.
Perfusion imaging may be performed with dobutamine; however, it is complicated by the resultant heart rate increase not seen with adenosine and dipyrimadole.
Perfusion CMR
In clinical practice, myocardial perfusion is assessed with CMR by evaluation of contrast enhancement during the first pass of a contrast agent bolus through the cardiac chambers and the myocardium using gadolinium and the stress agent (adenosine/Regadenoson, dipyridamole, or dobutamine).
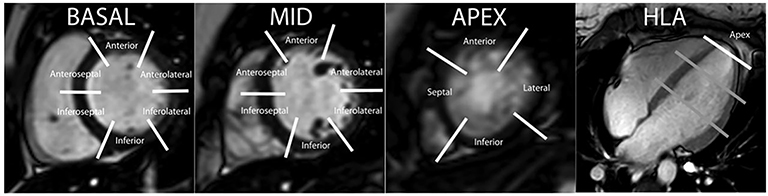
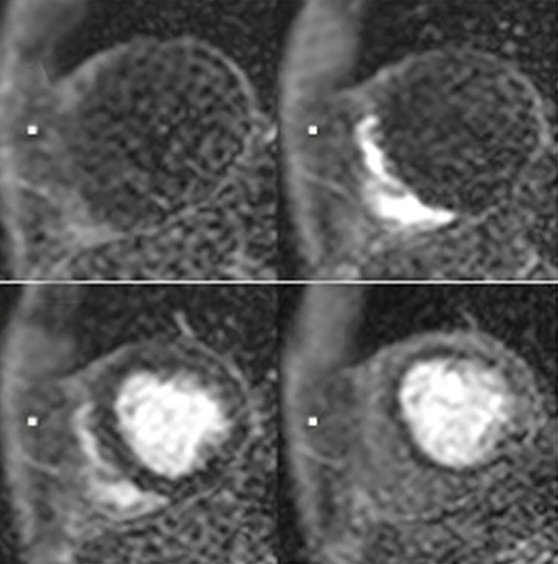
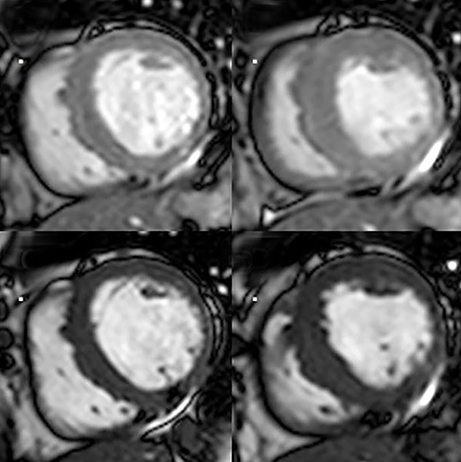
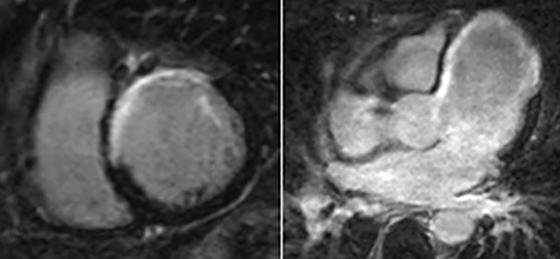
Perfusion CMR images are analyzed using the AHA 17 segment model (shown in Figure 3). The presence of segmental myocardial hypoenhancement reflects a relative hypoperfusion in comparison to other territories. Examples of first pass CMR perfusion images are shown in Figure 4. Short axis cine sequences at rest and stress are shown in Figure 5.
Interpretation of perfusion sequences can be limited by the presence of Gibbs ringing artifact at the blood-myocardial interface (mimicking a subendocardial hypoenhancement by creating a dark rim artifact due to sub-pixel shifting). However, the artifact is transient and typically occurs at the arrival of contrast in the left ventricular cavity, before its arrival into the myocardium, and disappears after 8 to 10 frames.5
Abnormalities in wall motion may be seen using either pharmacologic method; however, dobutamine has demonstrated greater sensitivity than adenosine.6 Myocardial contraction is analyzed using the AHA 17 segment model as in perfusion CMR.7
On dobutamine stress CMR, viability is defined by improvement of wall motion contractility in at least 2 contiguous segments at low dose (5 or 10 μg/kg per min). Ischemia is defined by an induction of wall motion abnormality in at least 2 contiguous segments during stress or if a biphasic response (improvement at low dose followed by decreasing contractility at higher dose) was demonstrated in areas with resting wall motion abnormalities. The test is negative if >85% of the theoretical maximal heart rate is reached with no induced wall motion abnormality. The test is non diagnostic if >85% of the theoretical maximal heart rate is not reached and no induced wall motion abnormalities are seen. The test is positive if the presence of ischemia is detected. Wall motion abnormalities detected by adenosine/dipyrimadole typically indicate severe stenoses and have been seen to be predictive of subsequent clinical events.8
Late gadolinium enhancement (LGE)
LGE is a well-documented technique that allows detection of myocardial scar with gadolinium, which shortens the T1 relaxation time. The addition of this sequence to CINE SSFP and perfusion CMR provides valuable information regarding myocardial viability as well as expected recovery following revascularization.
After the administration of the extracellular contrast agent (gadolinium chelates are typically used), infarcted or fibrotic tissue demonstrates increased signal on T1-weighted sequences due to reduced clearance and increased volume of distribution of the contrast agent in these regions.9 Ten to 30 minutes after intravenous injection of gadolinium (0.10-0.20 mmol/kg), gradient-echo inversion recovery (FLASH/PSIR) sequences are performed. The inversion time is set with a Look-Locker sequence at the point of optimal myocardial ing, allowing a better quantification of the infarcted bright zone.
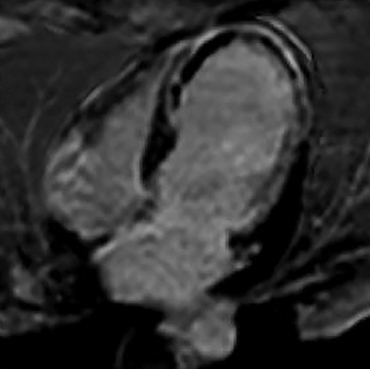
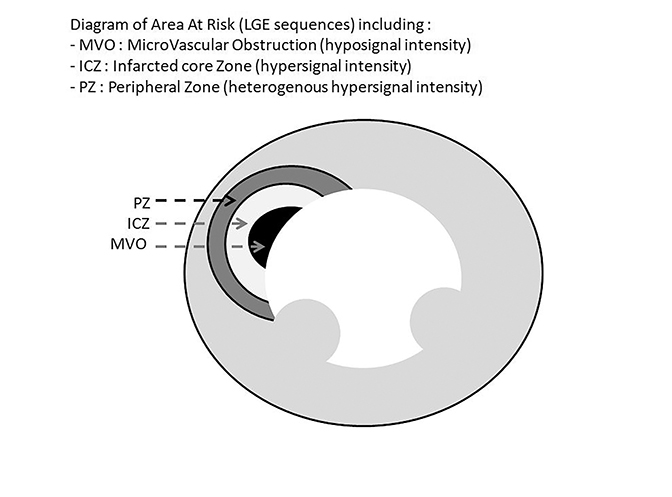
The infarct size defined by LGE hyperenhancement is a well-established strong predictor of outcomes after myocardial infarction (MI).10 Examples are shown in Figure 6. The transmural extent of hyperenhancement (<25%, 25-50%, 50-75%, >75%) predicts the functional improvement of the myocardium.11 In clinical practice, 50% transmurality of LGE is considered as a reasonable and practical cut-off to predict functional recovery after revascularization.12 LGE also detects microvascular obstruction (MVO) after acute MI, which is associated with poor clinical outcomes.13 MVO appears as an area of low signal intensity surrounded by enhancement and is described in Figure 7. After an acute MI, LGE is useful to assess size, extension, transmurality of the infarcted zone, and the presence of MVO. In case of a chronic MI, the sequence can assess viability for possible revascularization. This sequence is also useful for distinguishing between ischemic cardiomyopathy with the presence of segmental, subendocardial LGE or transmural LGE from a non-ischemic process, such as with myocarditis or Takotsubo cardiomyopathy in patients presenting with acute chest pain and elevated cardiac enzymes.14 Figure 8 describes the zones of delayed enhancement seen in an area at risk.
CMR vs PET and SPECT
Invasive coronary angiography is considered the gold standard for determining presence of coronary artery stenosis. When compared to coronary angiography, the diagnostic sensitivity and specificity of dobutamine cine-CMR are 83% and 86%, respectively.15 Adenosine stress perfusion CMR has a better sensitivity and specificity than with dipyridamole (90% and 81% versus 86% and 77%).16 Perfusion CMR has superior sensitivity and inferior specificity in comparison to SPECT.17 The diagnostic performance of CMR is similar to that of PET.18
Functional significance of coronary artery stenosis is best approximated by invasive pressure wire-derived fractional flow reserve (FFR). With FFR as reference, the sensitivity and specificity of adenosine stress CMR were 91% and 94%.19
Cardiac stress perfusion MRI has higher spatial resolution (2 mm) compared with SPECT (10 mm) and PET (5 mm), allowing greater sensitivity for detecting ischemic defects. In addition, there are no attenuation artifacts with CMR, conferring it a higher sensitivity in detecting diffuse balanced ischemia when compared to SPECT.20
CMR vs dobutamine echocardiography
Using invasive coronary angiography as a reference, dobutamine stress CMR has been reported to have higher sensitivity and specificity compared to dobutamine echocardiography (86.2% and 85.7% vs 74.3% and 69.8%).21 A more recent meta-analysis further supported this relationship with a sensitivity of 89% with CMR compared to 84% with stress echocardiography for excluding hemodynamically significant CAD determined by FFR.22
Feasibility and safety
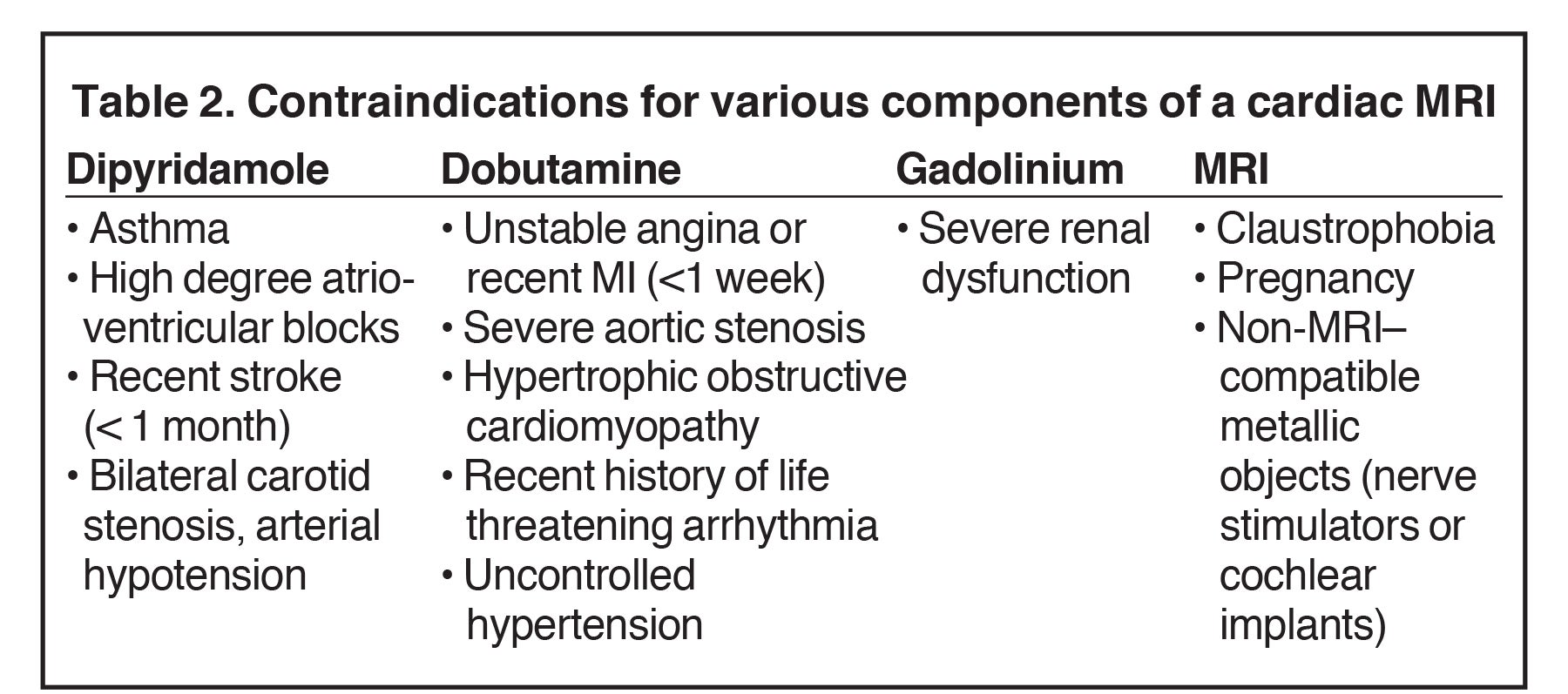
The safety of dobutamine CMR is comparable to dobutamine echocardiography with about 1 severe adverse event over 1000 scans.23 Perfusion stress MRI also has a good safety profile and greater feasibility than dobutamine stress MR, at approximately 100%. Excluding patients with contraindications to cardiac MRI, the main limitations to stress CMR include difficulty obtaining an acceptable ECG signal and patient claustrophobia.
Contraindications.
Contraindications are shown in Table 2.
Future development
Characterization of infarcted myocardial tissue—Area at risk
After an acute MI, infarcted myocardium can be better characterized by distinguishing between the microvascular obstruction zone, the infarction core necrosis, the peripheral infarction zone, and the ischemic zone with delineation of the myocyte area at risk.24
T2 relaxation time reflects the myocardial water content and has been shown to regions of edema in infarcted tissue.25 The area at risk, corresponding to myocyte edema, shows high signal intensity on T2-weighted black blood with fat saturation sequences but no LGE on delayed-enhancement images. The infarcted core necrosis also shows increased signal on T2 weighted images, although there is LGE on delayed-enhancement images. The LGE zone can be divided into a homogenously hyper-enhancing core reflecting non-viable myocardium surrounded by a rim of heterogeneous hyper-enhancement called the peripheral zone.24 T2 mapping can reduce the artifacts and loss of contrast seen with T2 weighted images through a quantitative method; however, it may underestimate the true area at risk.26 Precontrast (native) and postcontrast T1 mapping can be helpful in detecting interstitial edema in the setting of acute myocardial infarction as well as extracellular volume expansion with chronic myocardial infarction .27,28
CMR angiography
Although coronary MR angiography has made considerable improvements during the last two decades, it is still not widely applied per current guidelines.14,29 With the development of newer techniques (high-field strength, 32-channel cardiac coils, higher parallel imaging factors, and free breathing respiratory-gated sequences), coronary MR angiography approaches the diagnostic performance of 64-section CT.30 Moreover, unlike CT, MRA allows the assessment of the coronary lumen in a segment with heavily calcified plaque. There are a number of applications under investigation, including use of T1-weighted 3D black blood MR sequences to characterize coronary atherosclerotic plaque.31
Conclusion
CMR is a safe and proven modality for detecting ischemic heart disease. Within a single study, CMR is able to assess rest and stress regional contraction, and localize and evaluate regions of myocardial ischemia and viability, of importance given the prognostic impact for the patient. With performance that is comparable to PET/SPECT and at times favorable compared to stress echocardiography, it is now an essential tool for diagnosing ischemia and for determining the likelihood of success following myocardial revascularization.
References
- Fihn SD, Gardin JM, Abrams J, et al. 2012 ACCF/AHA/ACP/AATS/PCNA/SCAI/STS Guideline for the diagnosis and management of patients with stable ischemic heart disease: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines, and the American College of Physicians, American Association for Thoracic Surgery, Preventive Cardiovascular Nurses Association, Society for Cardiovascular Angiography and Interventions, and Society of Thoracic Surgeons. J Am Coll Cardiol. 2012; 60(24): e44-e164.
- Gould K L, Lipscomb K. Effects of coronary stenoses on coronary flow reserve and resistance. Am J Cardiol. 1974; 34(1): 48-55.
- Nesto RW, Kowalchuk GJ. The ischemic cascade: temporal sequence of hemodynamic, electrocardiographic and symptomatic expressions of ischemia. Am J Cardiol. 1987; 59(7): 23C-30C.
- Sonnenblick EH, Frishman WH, LeJemtel TH. Dobutamine: a new synthetic cardioactive sympathetic amine. N Engl J Med. 1979; 300(1):17-22.
- Motwani M, Maredia N, Fairbairn TA, et al. High-resolution versus standard-resolution cardiovascular MR myocardial perfusion imaging for the detection of coronary artery disease. Circ Cardiovasc Imaging. 2012; 5(3): 306-313.
- Paetsch I, Jahnke C, Wahl A, et al. Comparison of dobutamine stress magnetic resonance, adenosine stress magnetic resonance, and adenosine stress magnetic resonance perfusion. Circulation. 2004; 110(7): 835-842.
- Cerqueira MD, Weissman NJ, Dilsizian V, et al. Standardized myocardial segmentation and nomenclature for tomographic imaging of the heart. A statement for healthcare professionals from the Cardiac Imaging Committee of the Council on Clinical Cardiology of the American Heart Association. Int J Cardiovasc Imaging. 2002; 18(1): 539-542.
- Bodi V, Sanchis J, Lopez-Lereu MP, et al. Prognostic value of dipyridamole stress cardiovascular magnetic resonance imaging in patients with known or suspected coronary artery disease. J Coll Cardiol Am. 2007; 50(12): 1174-1179.
- Saeed M, Wagner S, Wendland MF, et al. Occlusive and reperfused myocardial infarcts: differentiation with Mn-DPDP--enhanced MR imaging. Radiology, 1989. 172(1): p. 59-64.
- Wu E, Ortiz J, Tejedor P, et al. Infarct size by contrast enhanced cardiac magnetic resonance is a stronger predictor of outcomes than left ventricular ejection fraction or end-systolic volume index: prospective cohort study. Heart. 2007; 94:730-736.
- Beek AM, Kuhl HP, Bondarenko O, et al. Delayed contrast-enhanced magnetic resonance imaging for the prediction of regional functional improvement after acute myocardial infarction. J Am Coll Cardiol. 2003; 42(5): 895-901.
- Kim RJ, Wu E, Rafael A, et al. The use of contrast-enhanced magnetic resonance imaging to identify reversible myocardial dysfunction. N Engl J Med. 2000; 343(20): 1445-1453.
- Bogaert J, Kalantzi M, Rademakers FE, et al. Determinants and impact of microvascular obstruction in successfully reperfused ST-segment elevation myocardial infarction. Assessment by magnetic resonance imaging. Eur Radiol. 2007; 17(10): 2572-2580.
- Roffi M, Patrono C, Collet J P, et al. 2015 ESC Guidelines for the management of acute coronary syndromes in patients presenting without persistent ST-segment elevation: Task Force for the Management of Acute Coronary Syndromes in Patients Presenting without Persistent ST-Segment Elevation of the European Society of Cardiology (ESC). Eur Heart J. 2016; 37(3): 267-315.
- Nandalur KR, Dwamena BA, Choudhri AF, et al. Diagnostic performance of stress cardiac magnetic resonance imaging in the detection of coronary artery disease: a meta-analysis. J Am Coll Cardiol. 2007; 50(14): 1343-1353.
- Hamon M, Fau G, Nee G, et al. C. Meta-analysis of the diagnostic performance of stress perfusion cardiovascular magnetic resonanace for detection of coronary artery disease . J Cardiovasc Magn Reson. 2010; 12: 29.
- Schwitter J, Wacker CM, Wilke N, et al. MR-IMPACT II: Magnetic resonance imaging for myocardial perfusion assessment in coronary artery disease trial: perfusion-cardiac magnetic resonance vs. single-photon emission computed tomography for the detection of coronary artery disease: a comparative multicentre, multivendor trial. Eur Heart J. 2013;34(10): 775-781.
- Schwitter J, Nanz D, Kneifel S, et al. Assessment of myocardial perfusion in coronary artery disease by magnetic resonance: a comparison with positron emission tomography and coronary angiography. Circulation. 2001; 103(18): 2230-2235.
- Watkins S, McGeoch R, Lyne J, et al. Validation of magnetic resonance myocardial perfusion imaging with fractional flow reserve for the detection of significant coronary heart disease. Circulation. 2009; 120(22): 2207-2213.
- Vogel-Claussen J, Skrok J, Dombroski D, et al., Comprehensive adenosine stress perfusion MRI defines the etiology of chest pain in the emergency room: Comparison with nuclear stress test. J Magn Reson Imaging. 2009; 30(4): 753-762.
- Nagel E, Lehmkuhl HB, Bocksch W, et al. Noninvasive diagnosis of ischemia-induced wall motion abnormalities with the use of high-dose dobutamine stress MRI: comparison with dobutamine stress echocardiography. Circulation. 1999; 99(6): 763-770.
- Takx RA, Blomberg BA, El Aidi H, et al. Diagnostic accuracy of stress myocardial perfusion imaging compared to invasive coronary angiography with fractional flow reserve meta-analysis. Circ Cardiovasc Imaging, 2015. 8(1).
- Mertes H, Sawada SG, Ryan T, et al. Symptoms, adverse effects, and complications associated with dobutamine stress echocardiography. Experience in 1118 patients. Circulation. 1993; 88(1):15-19.
- Ordovas K.G, Higgins CB. Delayed contrast enhancement on MR images of myocardium: past, present, future. Radiology. 2011; 261(2): 358-3574.
- Higgins CB, Herfkens R, Lipton MJ, et al. Nuclear magnetic resonance imaging of acute myocardial infarction in dogs: alterations in magnetic relaxation times. Am J Cardiol. 1983; 52(1): 184-148.
- Giri S, Chung YC, Merchant A, et al. T2 quantification for improved detection of myocardial edema. J Cardiovasc Magn Reson. 2009; 11: 56.
- Taylor AJ, Salerno M, Dharmakumar R, et al. T1 mapping: basic techniques and clinical applications. JACC Cardiovasc Imaging. 2016; 9(1): 67-81.
- Dall’Armellina E, Ferreira VM, Kharbanda RK, et al. Diagnostic value of precontrast T1 mapping in acute and chronic myocardial infarction. JACC Cardiovasc Imaging. 2013; 6(6): 739-742.
- Kohsaka S, Makaryus AN. Coronary angiography using noninvasive imaging techniques of cardiac CT and MRI. Curr Cardiol Rev. 2008; 4(4): 323-330.
- Ishida M, Sakuma H. Coronary MR angiography revealed: how to optimize image quality. Magn Reson Imaging Clin N Am. 2015; 23(1): 117-125.
- Makowski MR, Henningsson M, Spuentrup, et al. Characterization of coronary atherosclerosis by magnetic resonance imaging. Circulation. 2013; 128(11): 1244-1255.
Citation
FS DC, K M, J C, K O.Cardiac MRI: A preferred method for assessing myocardial ischemia and infarct burden. Appl Radiol. 2017; (11):21-29.
November 13, 2017