Detection and diagnosis of gastric carcinoma with multidetector and 3D computed tomography
Images








Dr. Bean is an Instructor of Radiology, Dr. Horton is an Associate Professor of Radiology, and Dr. Fishman is a Professor of Radiology and Oncology, and the Director of Diagnostic Imaging and Body CT, Johns Hopkins University School of Medicine, Baltimore, MD. Dr. Fishman is also a member of the editorial board of this journal.
The development of multidetector computed tomography (MDCT) has provided improved visualization and evaluation of the stomach. Due to their ability to provide excellent mucosal detail, endoscopy and upper gastrointestinal imaging (UGI) series have traditionally been considered the primary modalities for the detection of gastric carcinoma. Previously, this exquisite detail was unmatched by conventional single-slice computed tomography (CT). With advancements in computer and scanner technology, as well as development of 3-dimensional (3D) software programs, MDCT now allows improved detection and staging of stomach cancer. In addition, evaluation of intramural, extraluminal, and metastatic disease is not possible with endoscopy or UGI, but is readily performed with the cross-sectional capabilities of MDCT. This article illustrates the usefulness of MDCT and 3D CT for the evaluation of gastric carcinoma.
Technique
Scan protocol
Thorough examination of the stomach requires optimal gastric distension and properly timed intravenous (IV) contrast administration. The stomach wall enhances significantly with contrast administration, and this enhancement pattern can be altered by underlying pathology. Therefore, IV contrast administration is essential to detect disease. With the faster scanning capabilities of MDCT, this enhancement can be optimally timed to detect subtle mural abnormalities. In addition, the stomach must be adequately distended with proper oral contrast. Underdistension of the stomach can cause lesions to be missed or can produce pseudolesions, which can lead to over- or misdiagnosis. Gastric distension can be accomplished using positive oral contrast agents (diluted barium sulfate or iodinated solution), neutral substances (water), or negative contrast material (air).
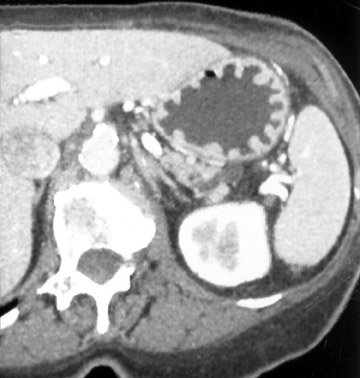
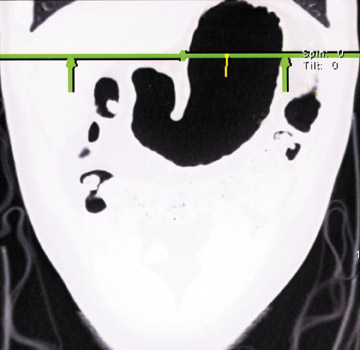
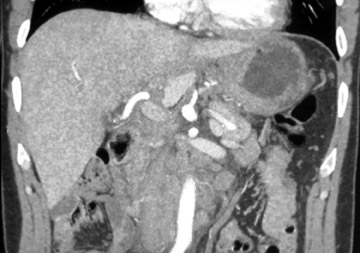
Water has emerged as an optimal oral contrast agent and several papers have shown its usefulness in delineating gastric disease. 1,2 Water is well-tolerated, inexpensive, results in good distension, and does not interfere with 3D manipulation of data sets. The relatively low attenuation of water (0 to 10 HU) produces excellent contrast differentiation between the enhancing wall of the stomach and the intraluminal water (Figure 1). Subtle enhancement differences of mural pathology may be masked by positive contrast agents in the adjacent lumen. The gastric wall may enhance up to 120 HU, which can be silhouetted by the positive oral contrast agent. 3 In addition, inadequate mixing of positive contrast agents and retained gastric products has led to pseudotumors.
At our institution, the patient is given 750 mL of water approximately 15 to 30 minutes before the study. Immediately prior to scanning, an additional 250 mL of water is given. Nonionic contrast material (120 mL of Omnipaque 350, GE Healthcare, Princeton, NJ) is routinely injected at a rate of 3 mL/sec. Arterial-phase images are acquired 25 seconds after the beginning of contrast injection. Venous-phase images are obtained 50 seconds after the start of the injection.
For detailed imaging of the stomach and 3D reconstruction, we utilize our 16-slice MDCT scanner (Sensation 16, Siemens Medical Solutions, Malvern, PA) to obtain 0.75-mm slices, acquired at a rate of 32 slices per second. These thin slices acquired at a fast rate result in high-resolution data sets with minimal artifact. This dual-phase protocol produces angiographic-quality delineation of gastric arteries and veins as well as visualization of liver metastases.
3D imaging
Previously, data acquired could be evaluated only in the axial plane, which is inadequate for the complex anatomy of the stomach. Although MDCT still acquires data in the axial plane, data is acquired as a volume set rather than sequences of adjacent slices. For this reason, the data can be seamlessly manipulated with 3D software, allowing it to be viewed in multiple planes with the addition of depth perception, which allows for better understanding and evaluation of anatomy and pathology. In addition, gastric pathology can be easily visualized in relationship to adjacent structures. For the stomach, coronal or coronal oblique planes are often helpful for delineation of pathology. Referring physicians often like this view, as it simulates the traditional familiar UGI images.
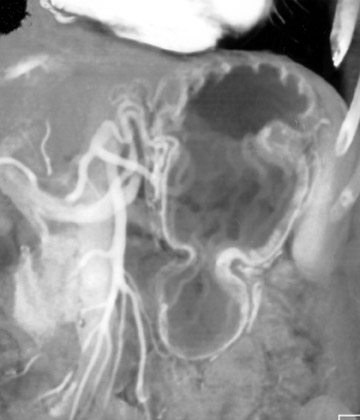
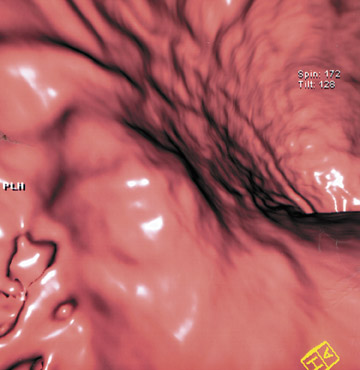
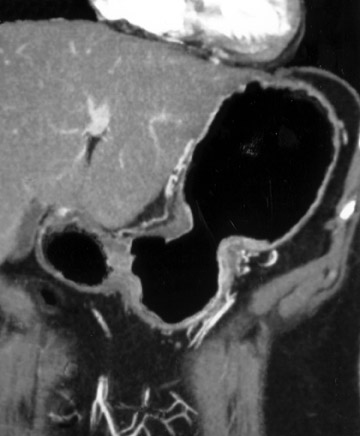
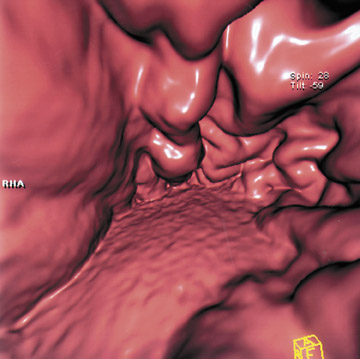
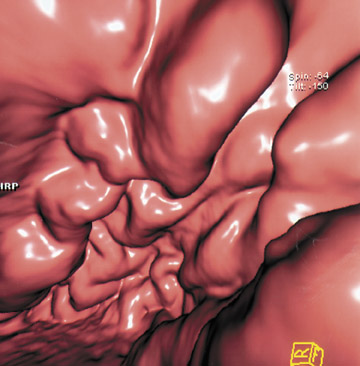
This 3D software allows manipulation of window width and level to accentuate desired structures, such as soft tissue, vessels, or even bone. CT angiography (CTA) can be performed using the same data set and can reveal effects of gastric pathology on adjacent vessels. This information can be valuable in determining a potential approach for surgery. The CT data set can also be windowed and manipulated to produce endoscopic images, giving a virtual gastroscopy, similar to virtual colonoscopy pictures. In this setting, the gastric mucosal folds are accentuated and intraluminal pathology is highlighted (Figures 2 and 3).
Gastric malignancy
Gastric adenocarcinoma
Adenocarcinoma accounts for approximately 95% of all malignant gastric neoplasms, typically occurring between the ages of 50 and 70 years with a male predominance. The distribution tends to be geographic, with the highest frequency in Japan, Chile, and Iceland.
Predisposing conditions include atrophic gastritis, pernicious anemia, gastric polyps, partial gastrectomy (such as Billroth II), and Ménétrier's disease. 4,5 This aggressive disease has an overall 5-year survival rate of <20%. 4 However, detection of early carcinoma offers the possibility of a cure.
Staging
Some surgeons opt for laparotomy in every case of gastric carcinoma, with the outcome either being curative resection or palliative therapy. Other physicians rely on imaging modalities for staging and therapy planning. CT is the most widely used modality for staging of gastric adenocarcinoma. Reported accuracy for CT staging of gastric cancer varies due to differences in equipment and scanning protocols. In a study by Hori et al 6 using single-detector CT with IV contrast and water as oral contrast, examinations detected 95% of advanced gastric cancers, 93% of elevated carcinomas, and 18% of early depressed carcinomas. With MDCT allowing more narrow collimation and thinner, more closely spaced scanning, it is probable that this accuracy will improve. 7,8
In addition, though controversial, dual-phase imaging may be beneficial in detecting and staging stomach cancer. In studies by Hundt et al 9 and Mani et al, 10 dual-phase imaging was performed with water as an oral contrast agent. In a study of 40 patients, Hundt and colleagues 9 were able to detect 97.5% of cancers correctly with CT staging-79.4% accuracy compared with pathologic staging. Mani and coworkers studied 20 patients and concluded that earlier-phase CT was highly accurate in determining the depth of tumor invasion through the wall, correctly determining the depth in 17 of 20 patients. Later-phase images are useful in detecting metastatic spread, especially liver metastasis. Optimal imaging protocols for detection and staging are still under investigation.
CT presentation
Adenocarcinoma of the stomach has varying presentations. The purpose of CT is to detect gastric tumors, determine the stage, and analyze for metastasis.
Primary tumor
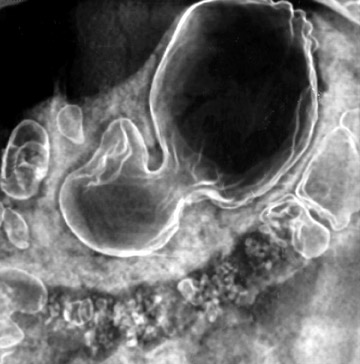
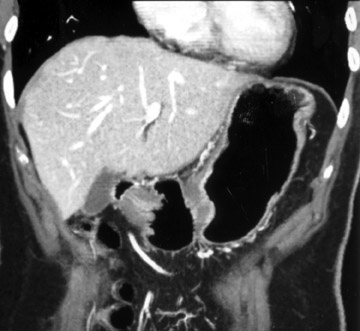
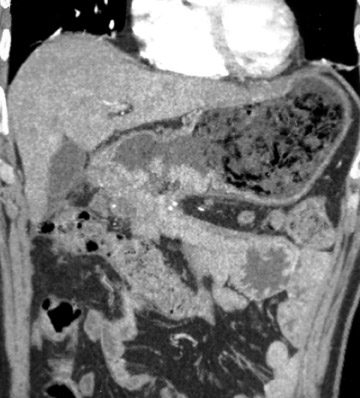
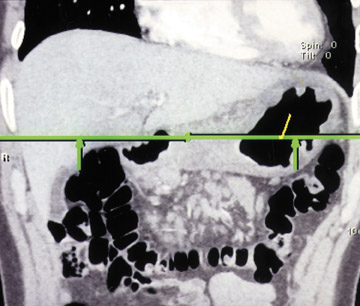
With optimal gastric distension, the wall of the stomach is very thin and its folds become essentially effaced. Normal gastric wall thickness, on average, measures 5 to 7 mm. Walls thicker than 8 to 10 mm are abnormal. On CT, gastric tumors may appear as a discrete mass with or without ulceration or may appear as an infiltrative lesion. These appearances were described in a paper by Ba-Ssalamah et al 4 that attempts to correlate tumor pathologic type and depth of wall invasion using MDCT (Figure 4). Infiltrating tumors can be focal, segmental, or diffuse. Diffusely infiltrating tumors can cause gastric rigidity, resulting in linitis plastica shown by limited distension after administration of oral contrast. The average wall thickness for this malignant neoplasm is 2 cm, with a range of 6 mm to 4 cm. 11 Though there have been significant technologic CT advances, the limitation of spatial resolution hampers distinction of gastric layers and, therefore, distinction of depth of tumor invasion.
Moss et al 12 developed a widely used staging system for gastric adenocarcinoma: Stage I disease-intraluminal mass invading gastric mucosa without deeper tumor extension; stage II disease-gastric wall thickness >1 cm, which is compatible with tumor spread into the muscularis propria; stage III disease-tumor invasion through the muscularis propria and serosa with or without local adenopathy; and stage IV disease-tumor invasion of adjacent organs or distant metastases.
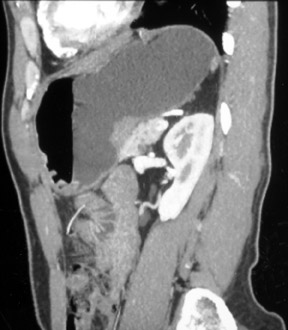
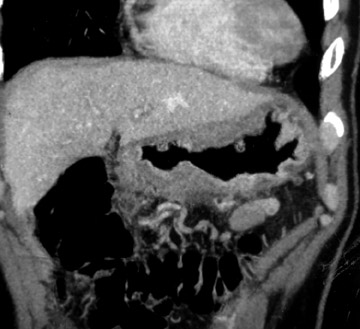
Stage I and stage II disease are considered limited and are treated with partial gastrectomy (Figure 5). Stages III and IV are considered locally advanced and are treated with chemotherapy. In advanced disease, surgery is reserved for palliative measures.
Local extension
Stage III disease indicates local disease composed of extragastric extension. This tumor spread can be subtle, manifested as slightly increased attenuation of the normally homogenous perigastric fat to complete obliteration of fat planes between organs with replacement by soft tissue due to tumor. This distinction can be difficult and can be mimicked by concurrent inflammatory conditions, such as pancreatitis. The probability of transmural extension increases in proportion to mural thickness of the stomach wall. Disease commonly spreads along ligaments and peritoneal reflections to extend to nearby organs.
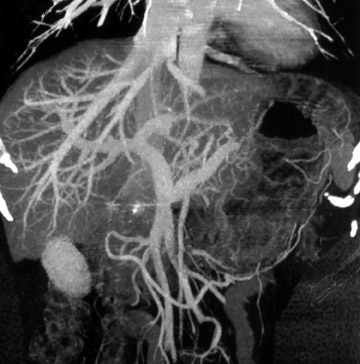
Common pathways of extension include the liver via the gastrohepatic ligament, the pancreas via the lesser sac, and the transverse colon via the gastrocolic ligament. Carcinoma of the cardia may extend to directly involve the distal esophagus, while tumor of the antrum may spread to the duodenum. Three-dimensional manipulation of the data sets may help with the detection of local spread (Figure 6).
Adenopathy
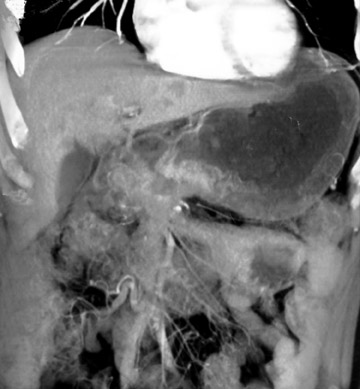
Lymph node involvement occurs in 74% to 88% of people with gastric malignancy because of the extensive lymphatic vessels of the stom-ach. 4 Lymphatic involvement increases relative to the size and depth of the primary lesion. Nodal malignancy decreases the median survival rate by 65%. 13 The best CT indicator of tumor extension to lymph nodes is enlargement. This spread commonly occurs in the region of the gastrohepatic ligament. Perigastric nodes are suspicious for nodal spread of disease when they measure >8 mm in diameter. Normal-size nodes may contain tumor, although enlarged ones do not. Multiple enlarged nodes are considered suspicious. The sensitivity for detection of nodal spread by CT ranges from 47% to 97%, primarily limited by inability to detect microscopic spread 14 (Figure 7).
Metastases
Gastric cancer frequently produces hematogenous metastases to the liver via gastric drainage through the portal vein. Other less frequent target organs that develop metastases include the lungs, adrenal glands, kidneys, bones, and brain. Peritoneal seeding may occur in advanced cases. Drop metastases to the ovaries are known as Krukenberg tumors. Multidetector CT can detect this spread of disease and allow for appropriate planning and treatment decisions.
Conclusion
Gastric adenocarcinoma is a deadly disease. Presentation varies from subtle mural abnormalities to widespread disease. Technologic advances, including MDCT and 3D capabilities, allow for improved detection and staging of stomach disease in conjunction with conventional imaging methods. With these improved imaging capabilities, MDCT may play an increasing role in evaluation and treatment planning.