MR elastography of liver disease: State of the art
Images
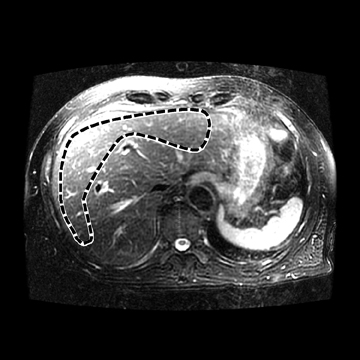
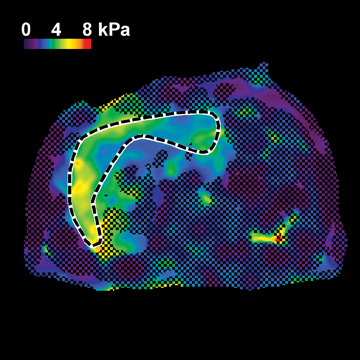

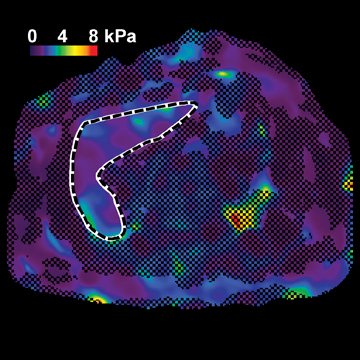
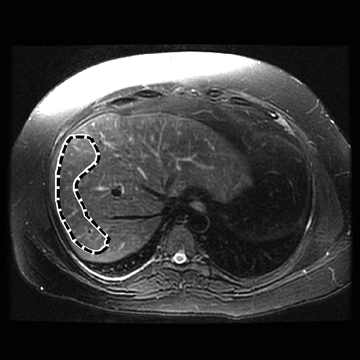



Hepatic fibrosis is a common result of different chronic liver diseases caused by viral infections, alcohol abuse, nonalcoholic fatty liver disease (NAFLD), autoimmune disease, and metabolic/genetic disorders. Hepatic fibrosis is a dynamic process which can, in some cases, be reversed with effective treatment.1,2 Otherwise, it can progress to more advanced stages, including cirrhosis, where liver function is impaired and severe complications, such as variceal bleeding, ascites, portal hypertension, hepatocellular carcinoma, and death can occur.3 In 2009, chronic liver disease and cirrhosis were responsible for 143,000 hospitalizations, 11,000 in-hospital deaths (7.7% death rate), and $6.7 billion in hospital charges in toto.4 Accurately detecting and staging fibrosis are important in the treatment of chronic liver disease and monitoring its effects on the liver. Chronic viral hepatitis C (HCV) infection is a significant cause of hepatic disease and its treatment may depend on the presence of substantial fibrosis.5,6
Liver biopsy is the current reference method for detection and staging of fibrosis. However, it is an invasive method and can cause undesired complications, including bleeding (1.7%)7 and death (0.01-1%).7-9 Liver biopsy accuracy is affected by sampling errors, which can lead to misclassified fibrosis stage by 1 category in 20% to 33% of patients10,11, and has high intra- and interoperator inconsistency, with a Kappa coefficient of 0.62-0.89, where 1.0 means perfect agreement.11,12 Furthermore, this approach is not well accepted by patients or physicians.13
Conventional noninvasive imaging techniques, such as ultrasound, computed tomography (CT), and magnetic resonance imaging (MRI), can visualize the morphologic changes of liver arising from fibrosis and cirrhosis. However, the sensitivity and accuracy of these modalities are low for detecting earlier stages of fibrosis and thus are not suitable for staging liver fibrosis over its entire spectrum.14 During the last several years, new noninvasive methods of assessing liver fibrosis have been developed and investigated; these include serum tests, diffusion-weighted MR imaging (DWI), contrast-enhanced MR imaging (CE-MRI), ultrasound-based transient elastography (UTE), and MR elastography (MRE). Accumulated data show that the AUROC (area under receiver operating characteristic curve) values are between 77% and 91% for serum tests,15-20 88% for DWI,21, and 83% for CE-MRI22 to detect the presence of liver fibrosis arising from different disease causes. As one of the 2 existing methods of measuring liver stiffness, UTE has higher AUROC values (0.79-0.98),23-25 yet it is less accurate for detecting early fibrosis stages, with a high technical failure rate in certain patient subsets (ascites, narrow intercostal space, obesity).26-28 The other method of measuring liver stiffness, MR elastography (MRE) is the most accurate (AUROC = 92%-100%) method to date for detecting and staging liver fibrosis, especially in the early stages of fibrosis because of its ability to measure cross-sectional areas of hepatic parenchyma, and using a technique that is not affected by ascites and obesity.3,21,26,27,29-31 In one study, MRE even detected necroinflammation before the onset of fibrosis in patients with nonalcoholic fatty liver disease (NAFLD).32 The detection of early stage fibrosis is of clinical significance as treatment of liver disease during this stage is thought to be more effective than treatment during later stages of fibrosis. Given MRE’s technical advantages and high diagnostic accuracy, this article reviews the application of MR elastography to clinical questions involving HCV, NAFLD, and methotrexate (MTX) exposure.
Hepatic MR elastography (MRE)
MR elastography is a technology for characterizing the biomechanical properties (eg, stiffness) of tissues in vivo. It uses MR phase-contrast techniques to acquire images of wave propagation in tissue, which are produced by an external source of mechanical vibration. The images of wave propagation are interpreted and processed by inversion algorithms, which results in a tissue stiffness map called an elastogram from which the tissue stiffness is measured.33 MRE exams of normal livers have visibly shorter wavelengths in the wave images and lower liver stiffness values in the elastograms, which means normal livers are soft. Fibrotic and cirrhotic livers have longer wavelengths and higher liver stiffness values, which means diseased livers are hard.3,21,26,27,29-31 This is consistent with the experience of clinicians whose palpations with the fingers are used to subjectively examine and feel the stiffness of the liver. Details of the MRE technique and scan parameters can be found in the literature.3,30,31 A typical MRE procedure is summarized here.
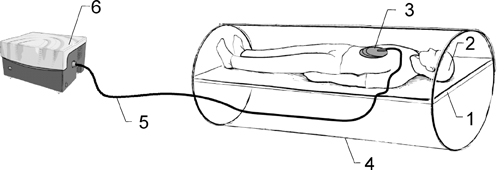
As shown in Figure 1, a patient lies on the scanner table in the supine position and a drum-like acoustic passive driver is positioned against the body wall, close to the liver, and secured by an elastic belt (not shown in the figure for a clear view) wrapped around the patient’s body. The passive driver is connected to an acoustic speaker system located outside the scanner room via a flexible polyvinylchloride (PVC) tube, which provides a programmed external mechanical vibration. The acoustic speaker produces vibrations at audible frequencies (typically 40Hz-80Hz), which are transmitted to the passive driver through the tube. A gradient-echo, MRE imaging sequence is used to acquire images of wave propagation within the liver. Subsequently, elastograms are calculated using a direct inversion of the differential equations describing the wave propagation.34 In addition to the liver, MRE has been adapted for use in other organs like the brain, breast, muscle, heart, kidney, spleen, and lung; promising results have been observed for these applications.35-47
Diagnostic accuracy of MRE in detecting fibrosis
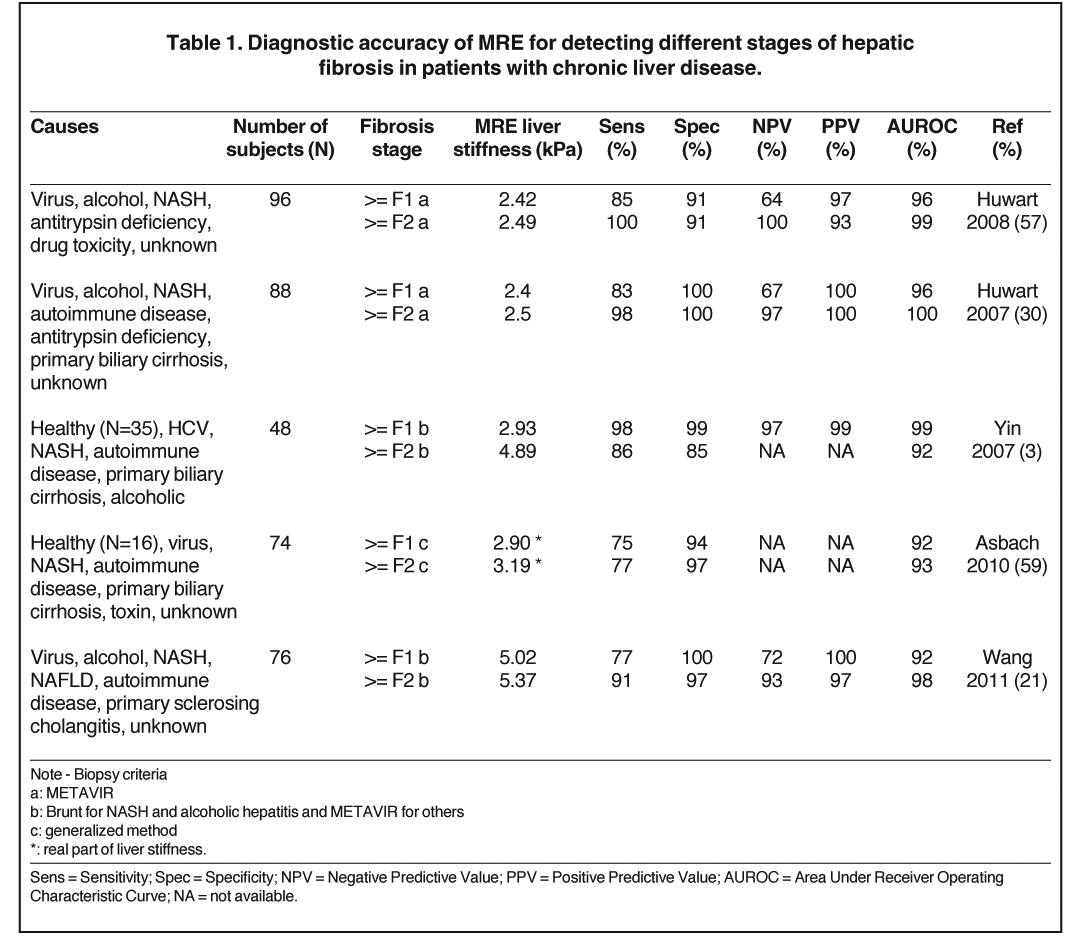
Hepatic fibrosis is an accumulation of the extracellular matrix that results from hepatic stellate cell transdifferentiation to myofibroblasts trigged by necroinflammation of hepatocytes. The necroinflammation is a wound-healing response to liver-cell injury that may be due to different causes of liver disease.48 Accumulated data have shown that liver stiffness measured by MRE is highly correlated and increases with fibrosis stage identified by liver histology.3,21,26,27,29-31 In some cases, an early increase of liver stiffness can be found even before the onset of fibrosis due to liver cell injury.32,49-53 One possible explanation of the strong relationship between liver disease and liver stiffness could be that early liver cell injury leads to changes in the extracellular matrix that increase the stiffness of hepatic tissue, which through a process known as mechanotransduction promotes the activation of stellate cells and the subsequent development of fibrosis,32,49,50 which in turn further increases liver stiffness. The persistent elevation in stiffness of the mechanical environment is then believed to accelerate fibrosis progression to more advanced stages. To date, the data have shown that, in chronic liver diseases with different causes, MRE has high diagnostic accuracy (AUROC = 92%-100%) for detecting and staging hepatic fibrosis, as seen in Table 1. Within our institution, we use a liver stiffness value of 2.93 kPa (kilopascals) as the threshold for detecting nonfibrotic liver tissue, where an abnormal liver stiffness is > 2.93 kPa. The diagnostic accuracy, sensitivity, specificity, positive- and negative-predictive values of this cut-off value are no lower than 97%.3
Several groups have shown that hepatic MRE has excellent reproducibility and inter-operator consistency. The coefficient of variation between hepatic stiffness measurements conducted on different days among the same individuals ranged between 7% and 12%,54-56 while the intra-class correlation coefficient for hepatic stiffness measured by 2 different operators was 97% to 99% (100% means exactly the same reading).57,58
Chronic hepatitis C virus
Approximately 150,000 new cases of chronic liver disease were diagnosed in the U.S. adult population each year from 1999 through 2001, with two-thirds of individuals affected by hepatitis C.60 Determining the presence and degree of liver fibrosis is important for planning treatment in patients with chronic hepatitis C viral infection.5,6 Serum tests are routinely performed to assess liver damage due to HCV, but they have low specificity for disease severity. Therefore, liver biopsy is the only reference method to confirm and stage fibrosis in HCV patients. Utilizing multiple parameters from serum tests and clinical risk factors in a model could improve diagnostic accuracy. One study using a model that included 4 readily available characteristics to predict the presence of cirrhosis in HCV patients (platelet count, presence of spider nevi, aspartate transaminase (AST), and gender) resulted in an AUROC of 0.94 for the training set and 0.93 for the validation patient group.61 However, a model using combined data from serum tests and clinical information needs further validation because of the different patient demographics between the training patient group and the tested patients. For example, the AUROC value of such a model dropped from 0.84 in a training patient group to 0.77 in a validation patient group for detecting significant fibrosis (stages F2-F4) in HCV patients.16 A combined data review from 4 studies showed that ultrasound-based transient elastography (UTE) had an AUROC of 0.83 to detect significant fibrosis (F2-F4) in HCV-related patients, but was less accurate for earlier stages of fibrosis.62
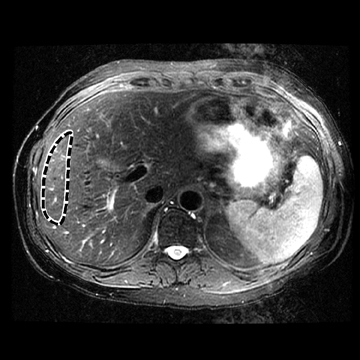
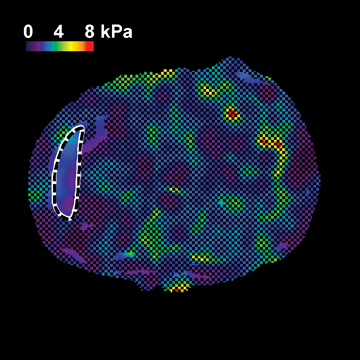
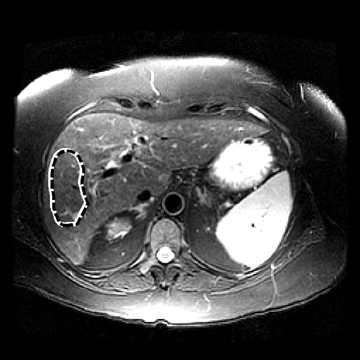
In one MRE study of subjects with HCV (METAVIR F2–F4) and healthy volunteers, the mean liver stiffness was significantly greater in patients with HCV than in healthy volunteers, and the correlation between liver stiffness and fibrosis stage, confirmed by liver biopsy, was 0.89.54 MRE scan examples of HCV patients with biopsy-proven fibrosis stages 0 and 3 as shown in Figure 2.
Nonalcoholic steatohepatitis
Nonalcoholic fatty liver disease is an increasingly prevalent clinical syndrome associated with obesity and type-2 diabetes mellitus, which is estimated to affect one-third of the general adult population in the USA.63-67 The spectrum of NAFLD ranges from simple steatosis to necro-inflammation and fibrosis. Simple steatosis is thought to have a benign long-term prognosis. In contrast, up to 25% of patients may progress from simple steatosis to nonalcoholic steatohepatitis (NASH), which is characterized by necro-inflammation and/or varying degrees of fibrosis.63,65,68 Studies to date estimate that early-stage NASH has a probability of 18% to 39% of progressing to more advanced stages of fibrosis and cirrhosis within 8 years from initial detection.69-73 It is anticipated that NASH-induced cirrhosis will also become the most common indication for liver transplantation in the future.65 Therefore, early liver biopsy has been suggested in all NAFLD patients to stratify the disease so that earlier interventions and more aggressive treatment can be applied to reduce overall mortality.73
Due to the invasive nature of liver biopsies, however, noninvasive methods have been evaluated for the diagnosis and serial assessment of NASH; these include novel serological and imaging tests. Serum markers for NAFLD have reasonably good accuracy (AUROC = 77-91%) in detecting advanced fibrosis,15-20 but are poor at diagnosing milder degrees of fibrosis and necro-inflammation. Plasma pentraxin 3 (PTX3) and cytokeratin 18 (CK18) fragments have shown improved accuracy for detecting NASH (AUROC = 75-83%), yet further validation of these approaches is still required.74-77 UTE had high accuracy (AUROC = 79-0.98%) for detecting fibrosis in NASH patients.23-25 However, UTE scans in patients with NAFLD showed unreliable measurements in 14% of patients due to obesity and decreased diagnostic accuracy.28,78 In addition, UTE was not sensitive in identifying steatohepatitis without fibrosis in patients with NAFLD.25 In contrast, an independent study using MRE detected a significant increase in liver stiffness due to necroinflammation prior to the onset of fibrosis in NASH patients compared to patients with simple steatosis. MRE had a sensitivity of 94% and a specificity of 73% using a threshold of 2.74 kPa to discern NASH from simple steatosis (AUROC = 0.93).32 Figure 3 demonstrates 3 MRE scans of fatty liver disease patients with simple steatosis alone, necroinflammation, but no fibrosis, and with fibrosis.
Obese patients and bariatric surgery
Bariatric surgery has been shown to be a very effective treatment for medically-complicated obesity. In turn, patients undergoing bariatric surgery who are also affected by NAFLD and NASH have experienced improvements in liver histology following significant weight loss. One study showed that of 116 patients who had necro-inflammatory activity on a presurgical liver biopsy, 108 had complete regression after the surgery. In 12 patients with fibrosis at the first biopsy, 10 had complete remission and 2 showed improvement. In all patients, the BMI decreased from 55.2 ± 8.3 to 30.5 ± 6.6 kg/m2.79
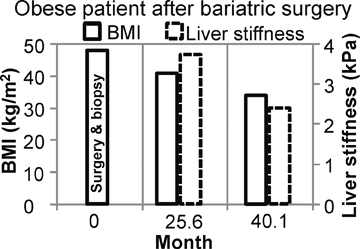
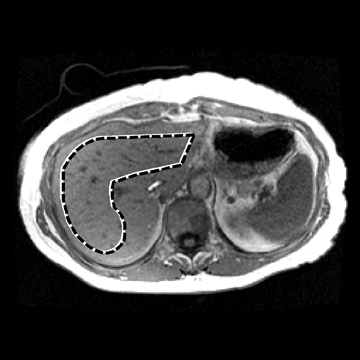
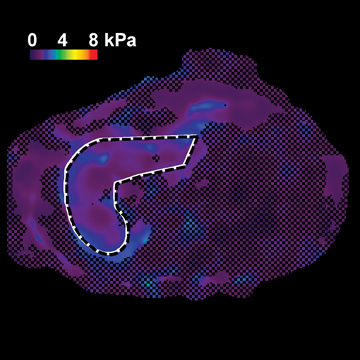
Figure 4 shows the potential use of follow-up MRE to evaluate NASH treatment with bariatric surgery in a 33-year-old obese female patient with a BMI of 47.2 kg. A liver biopsy done on the day of surgery found grade 1 of 3 necroinflammation and stage 3 of 4 fibrosis. No further liver biopsy was performed. A clinical liver MRI scan including MRE was performed 2 years later and a second exam 3 years later. On the first MRI/MRE study, the patient had a BMI of 40 kg/m2, a mean (± SD) liver stiffness of 3.67 ± 0.1 kPa (nonfibrotic value <2.93), with mild increased T2 signal in the periportal tracts suggestive of inflammation. On the second visit, the patient had a BMI of 33 kg/m2, mean (± SD) liver stiffness of 2.32 ± 0.33kPa (nonfibrotic value <2.93), and the increased T2 signal in the periportal tract was no longer evident. The decreased liver stiffness was consistent with improvement of this patient’s condition.
Methotrexate use
Methotrexate (MTX) is the most frequently prescribed drug for rheumatoid arthritis (RA) because of its efficacy, low cost, and safety.80 Generally speaking, MTX is tolerated well by most patients, yet its potential for hepatotoxicity and liver fibrosis has remained an ongoing concern. It is uncertain whether the risk for liver fibrosis is increased with cumulative doses of MTX. In 1994, the American College of Rheumatology (ACR) developed guidelines for laboratory monitoring of liver enzyme tests, and suggested further evaluation, including liver biopsy, based on the frequency (5 of 9) of abnormal aspartate aminotransferase (AST) values during one year.81 A recent study using liver stiffness measurement by MRE as a surrogate measure of liver fibrosis demonstrated that neither the total MTX dose nor the duration of MTX treatment was associated with mean hepatic stiffness in 65 RA patients who underwent MRE scans (unpublished data). Figures 5A and 5B shows MRE scan examples of 2 RA patients receiving MTX for disease control, and show the potential of MRE to detect liver fibrosis related to drug toxicity.
These images show an MRI/MRE exam of a 70-year-old female RA patient with 24 years of MTX treatment; the total MTX dose was 18,468 mg, BMI = 24.1 kg/m2. This patient had mean (± SD) hepatic stiffness measured as 2.07 ± 0.30 kPa (nonfibrotic value < 2.93), and had 0/4 abnormal AST tests done during 2 years prior to the MRI/MRE exam. This patient continued to use MTX for the treatment of RA.
Figures 5C and 5D show an MRI/MRE exam of a 66-year-old male RA patient with 4 years of MTX use; the total MTX dose was 3680 mg; his BMI = 37.3 kg/m2. The mean (± SD) hepatic stiffness measured on the elastogram was 3.13 ± 0.40 kPa (nonfibrotic value <2.93). A liver biopsy was ordered due to the abnormal liver stiffness and the biopsy revealed mild fatty change with mild portal fibrosis. This patient had 0/10 abnormal AST tests done during the 2 years prior to the MRI/MRE exam. Liver fibrosis would not have been suspected or found without the MRE scan.
Comparison with ultrasound-based transient elastography
Both MRE and UTE noninvasively measure liver stiffness, but MRE can measure liver stiffness more accurately because it captures 2-dimensional cross-sectional images; UTE may have sampling errors due to its 1-dimensional nature. UTE also has technical limitations when measuring liver stiffness in obese patients and those with ascites or narrow intercostal spaces.26-28 UTE scans in 14% of patients with NAFLD showed unreliable measurements and decreased diagnostic accuracy due to obesity.28,78 Recently, extra-large probes have been developed to address the technical limitations of UTE for imaging obese patients, but these still require validation. MRE is not limited by such factors, yet the approach also has its own technical limitations, including claustrophobia and other typical contraindications for MRI, such as magnetically susceptible implants. Low liver signal related to increased hepatic iron from hemochromatosis or advanced chronic liver disease is also problematic.3,57 However, continued developments in MRE technology indicate that patients with low liver signal can potentially undergo liver stiffness assessment using short-echo time and spin-echo MRE sequences.82
In characterizing liver tumors, MRE is the only technique capable of noninvasively measuring tumor stiffness because it reports any masses along with cross-sectional stiffness information about the liver. Tumor assessment is currently not possible with UTE because its fixed acoustic window cannot specifically target tumors, which can occur at any location in the liver.
Conclusions
Based on growing evidence in the scientific literature, quantitative elasticity imaging is being increasingly recognized by the hepatology community as a useful tool for noninvasively assessing and diagnosing chronic liver disease. MR elastography has moved from the laboratory to the clinic and is increasingly available as an FDA-approved option for MRI systems. Over the past 4 years, > 1700 patients have undergone hepatic MRE exams as a part of clinical practice at the authors’ institution. Similar experience is accumulating at many other centers around the world. For many patients, MRE is emerging as an effective, more comfortable, and less expensive diagnostic alternative to biopsy for assessing hepatic fibrosis.
References
- Friedman SL, Bansal MB. Reversal of hepatic fibrosis -- fact or fantasy? Hepatology. 2006;43:S82-88.
- Albanis E, Friedman SL. Antifibrotic agents for liver disease. American Journal of Transplantation. 2006;6:12-19.
- Yin M, Talwalkar JA, Glaser KJ, et al. Assessment of hepatic fibrosis with magnetic resonance elastography. Clin Gastroenterol Hepatol. 2007;5:1207-1213, e1202.
- AHRQ. Healthcare Costs Utilization Project online datebase. In:http://hcup.ahrq.gov/HCUPNet.asp:U.S. Department of Health & Human Services, 2009.
- Friedman SL. Liver fibrosis - from bench to bedside. Journal of Hepatology. 2003;38:38-53.
- Strader DB, Wright T, Thomas DL, Seeff LB. Diagnosis, management, and treatment of hepatitis C. Hepatology. 2004;39:1147-1171.
- Gilmore IT, Burroughs A, Murray-Lyon IM, et al. Indications, methods, and outcomes of percutaneous liver biopsy in England and Wales: An audit by the British Society of Gastroenterology and the Royal College of Physicians of London. Gut. 1995;36:437-441.
- McGill DB, Rakela J, Zinsmeister AR, Ott BJ. A 21-year experience with major hemorrhage after percutaneous liver biopsy. Gastroenterology. 1990;99:1396-1400.
- Cadranel JF, Rufat P, Degos F. Practices of liver biopsy in France: Results of a prospective nationwide survey. For the Group of Epidemiology of the French Association for the Study of the Liver (AFEF). Hepatology. 2000;32:477-481.
- Standish RA, Cholongitas E, Dhillon A, et al. An appraisal of the histopathological assessment of liver fibrosis. Gut. 2006;55:569-578.
- Regev A, Berho M, Jeffers LJ, et al. Sampling error and intraobserver variation in liver biopsy in patients with chronic HCV infection. Am J Gastroenterol. 2002;97:2614-2618.
- Kleiner DE, Brunt EM, Van Natta M, et al. Design and validation of a histological scoring system for nonalcoholic fatty liver disease. Hepatology. 2005;41:1313-1321.
- Sporea I, Popescu A, Sirli R. Why, who and how should perform liver biopsy in chronic liver diseases. World J Gastroenterol. 2008;14:3396-3402.
- Faria SC, Ganesan K, Mwangi I, et al. MR imaging of liver fibrosis: Current state of the art. Radiographics. 2009;29:1615-1635.
- Angulo P, Hui JM, Marchesini G, et al. The NAFLD fibrosis score: A noninvasive system that identifies liver fibrosis in patients with NAFLD. Hepatology. 2007;45:846-854.
- Sud A, Hui JM, Farrell GC, et al. Improved prediction of fibrosis in chronic hepatitis C using measures of insulin resistance in a probability index. Hepatology. 2004;39:1239-1247.
- Sakugawa H, Nakayoshi T, Kobashigawa K, et al. Clinical usefulness of biochemical markers of liver fibrosis in patients with nonalcoholic fatty liver disease. World J Gastroenterol. 2005;11:255-259.
- Mohamadnejad M, Montazeri G, Fazlollahi A, et al. Noninvasive markers of liver fibrosis and inflammation in chronic hepatitis B-virus related liver disease. Am J Gastroenterol. 2006;101:2537-2545.
- Zhou K, Lu LG. Assessment of fibrosis in chronic liver diseases. J Dig Dis. 2009;10:7-14.
- Guha IN, Parkes J, Roderick P, et al. Noninvasive markers of fibrosis in nonalcoholic fatty liver disease: Validating the European Liver Fibrosis Panel and exploring simple markers. Hepatology. 2008;47:455-460.
- Wang Y, Ganger DR, Levitsky J, et al. Assessment of chronic hepatitis and fibrosis: Comparison of MR elastography and diffusion-weighted imaging. AJR Am J Roentgenol. 2011;196:553-561.
- Friedrich-Rust M, Muller C, Winckler A, et al. Assessment of liver fibrosis and steatosis in PBC with FibroScan, MRI, MR-spectroscopy, and serum markers. J Clin Gastroenterol. 2010;44:58-65.
- Ziol M, Handra-Luca A, Kettaneh A, et al. Noninvasive assessment of liver fibrosis by measurement of stiffness in patients with chronic hepatitis C. Hepatology. 2005;41:48-54.
- Takemoto R, Nakamuta M, Aoyagi Y, et al. Validity of FibroScan values for predicting hepatic fibrosis stage in patients with chronic HCV infection. J Dig Dis. 2009;10:145-148.
- Lupsor M, Badea R, Stefanescu H, et al. Performance of unidimensional transient elastography in staging non-alcoholic steatohepatitis. J Gastrointestin Liver Dis. 2010;19:53-60.
- Talwalkar JA, Yin M, Fidler JL, et al. Magnetic resonance imaging of hepatic fibrosis: Emerging clinical applications. Hepatology. 2008;47:332-342.
- Bensamoun SF, Wang L, Robert L, et al. Measurement of liver stiffness with two imaging techniques: magnetic resonance elastography and ultrasound elastometry. J Magn Reson Imaging. 2008;28:1287-1292.
- Petta S, Di Marco V, Camma C, et al. Reliability of liver stiffness measurement in non-alcoholic fatty liver disease: The effects of body mass index. Aliment Pharmacol Ther. 2011;33:1350-1360.
- Huwart L, van Beers BE. MR elastography. Gastroenterol Clin Biol. 2008;32:68-72.
- Huwart L, Sempoux C, Salameh N, et al. Liver fibrosis: Noninvasive assessment with MR elastography versus aspartate aminotransferase-to-platelet ratio index. Radiology. 2007;245:458-466.
- Asbach P, Klatt D, Hamhaber U, et al. Assessment of liver viscoelasticity using multifrequency MR elastography. Magn Reson Med. 2008;60:373-379.
- Chen J, Talwalkar JA, Yin M, et al. Early detection of nonalcoholic steatohepatitis in patients with nonalcoholic fatty liver disease by using MR elastography. Radiology. 2011;259:749-756.
- Muthupillai R, Lomas DJ, Rossman PJ, et al. Magnetic resonance elastography by direct visualization of propagating acoustic strain waves. Science. 1995;269:1854-1857.
- Manduca A, Oliphant TE, Dresner MA, et al. Magnetic resonance elastography: Non-invasive mapping of tissue elasticity. Med Image Anal. 2001; 5:237-254.
- Murphy MC, Huston J, Jack CR, et al. Decreased brain stiffness in Alzheimer’s disease determined by magnetic resonance elastography. J Magn Reson Imaging. 2011.
- Xu L, Lin Y, Xi ZN, et al. Magnetic resonance elastography of the human brain: A preliminary study. Acta Radiol. 2007;48:112-115.
- Siegmann KC, Xydeas T, Sinkus R, et al. Diagnostic value of MR elastography in addition to contrast-enhanced MR imaging of the breast-initial clinical results. Eur Radiol. 2009;20:318-325.
- Chen J, Glaser KJ, Stinson EG, et al. Noncontact driver system for MR elastography of the breast. Proc. Intl. Soc. Mag. Reson. Med. 2011:3490.
- Elgeti T, Beling M, Hamm B, et al. Cardiac magnetic resonance elastography: Toward the diagnosis of abnormal myocardial relaxation. Invest Radiol. 2010;45:782-787.
- Kolipaka A, McGee KP, Araoz PA, et al. MR elastography as a method for the assessment of myocardial stiffness: comparison with an established pressure-volume model in a left ventricular model of the heart. Magn Reson Med. 2009;62:135-140.
- Chen Q, Bensamoun S, Basford JR, et al. Identification and quantification of myofascial taut bands with magnetic resonance elastography. Arch Phys Med Rehabil. 2007;88:1658-1661.
- Bensamoun SF, Ringleb SI, Chen Q, et al. Thigh muscle stiffness assessed with magnetic resonance elastography in hyperthyroid patients before and after medical treatment. J Magn Reson Imaging. 2007;26:708-713.
- Rouviere O, Souchon R, Pagnoux G, et al. Magnetic resonance elastography of the kidneys: Feasibility and reproducibility in young healthy adults. J Magn Reson Imaging. 2011;34:880-886.
- Bensamoun SF, Robert L, Leclerc GE, et al. Stiffness imaging of the kidney and adjacent abdominal tissues measured simultaneously using magnetic resonance elastography. Clin Imaging. 2011;35:284-287.
- Mariappan YK, Glaser KJ, Hubmayr RD, et al. MR elastography of human lung parenchyma: Technical development, theoretical modeling and in vivo validation. J Magn Reson Imaging. 2011;33:1351-1361.
- Talwalkar JA, Yin M, Venkatesh S, et al. Feasibility of in vivo MR elastographic splenic stiffness measurements in the assessment of portal hypertension. AJR Am J Roentgenol. 2009;193:122-127.
- Nedredal GI, Yin M, McKenzie T, et al. Portal hypertension correlates with splenic stiffness as measured with MR elastography. J Magn Reson Imaging. 2011;34:79-87.
- Gressner OA, Weiskirchen R, Gressner AM. Evolving concepts of liver fibrogenesis provide new diagnostic and therapeutic options. Comp Hepatol. 2007;6:7.
- Wells RG. The role of matrix stiffness in hepatic stellate cell activation and liver fibrosis. J Clin Gastroenterol. 2005;39:S158-161.
- Wells RG. The role of matrix stiffness in regulating cell behavior. Hepatology. 2008;47:1394-1400.
- Li Z, Dranoff JA, Chan EP, et al. Transforming growth factor-beta and substrate stiffness regulate portal fibroblast activation in culture. Hepatology. 2007;46:1246-1256.
- Georges PC, Hui JJ, Gombos Z, et al. Increased stiffness of the rat liver precedes matrix deposition: Implications for fibrosis. Am J Physiol Gastrointest Liver Physiol. 2007;293:G1147-1154.
- Salameh N, Larrat B, Abarca-Quinones J, et al. Early detection of steatohepatitis in fatty rat liver by using MR elastography. Radiology. 2009;253:90-97.
- Shire NJ, Yin M, Chen J, et al. Test-retest repeatability of MR elastography for noninvasive liver fibrosis assessment in hepatitis C. J Magn Reson Imaging. 2011;34:947-955.
- Hines CD, Bley TA, Lindstrom MJ, Reeder SB. Repeatability of magnetic resonance elastography for quantification of hepatic stiffness. J Magn Reson Imaging. 2010;31:725-731.
- Hines CD, Lindstrom MJ, Varma AK, Reeder SB. Effects of postprandial state and mesenteric blood flow on the repeatability of MR elastography in asymptomatic subjects. J Magn Reson Imaging. 2011;33:239-244.
- Huwart L, Sempoux C, Vicaut E, et al. Magnetic resonance elastography for the noninvasive staging of liver fibrosis. Gastroenterology. 2008; 135:32-40.
- Motosugi U, Ichikawa T, Sano K, et al. Magnetic resonance elastography of the liver: Preliminary results and estimation of inter-rater reliability. Jpn J Radiol. 2011;28:623-627.
- Asbach P, Klatt D, Schlosser B, et al. Viscoelasticity-based staging of hepatic fibrosis with multifrequency MR elastography. Radiology. 2010;257:80-86.
- Bell BP, Manos MM, Zaman A, et al. The epidemiology of newly diagnosed chronic liver disease in Gastroenterology practices in the United States: Results from population-based surveillance. Am J Gastroenterol. 2008;103:2727-2736;quiz 2737.
- Kaul V, Friedenberg FK, Braitman LE, et al. Development and validation of a model to diagnose cirrhosis in patients with hepatitis C. Am J Gastroenterol. 2002;97:2623-2628.
- Shaheen AA, Wan AF, Myers RP. FibroTest and FibroScan for the prediction of hepatitis C-related fibrosis: A systematic review of diagnostic test accuracy. Am J Gastroenterol. 2007;102:2589-2600.
- Ehman RL. Science to practice: Can MR elastography be used to detect early steatohepatitis in fatty liver disease? Radiology. 2009;253:1-3.
- Adams LA, Lindor KD. Nonalcoholic fatty liver disease. Ann Epidemiol. 2007;17:863-869.
- Charlton M. Nonalcoholic fatty liver disease: A review of current understanding and future impact. Clin Gastroenterol Hepatol. 2004;2:1048-1058.
- Ludwig J, Viggiano TR, McGill DB, Oh BJ. Nonalcoholic steatohepatitis: Mayo Clinic experiences with a hitherto unnamed disease. Mayo Clin Proc. 1980;55:434-438.
- Browning JD, Szczepaniak LS, Dobbins R, et al. Prevalence of hepatic steatosis in an urban population in the United States: impact of ethnicity. Hepatology. 2004;40:1387-1395.
- Adams LA, Lymp JF, St Sauver J, et al. The natural history of nonalcoholic fatty liver disease: A population-based cohort study. Gastroenterology. 2005;129:113-121.
- Harrison SA, Torgerson S, Hayashi PH. The natural history of nonalcoholic fatty liver disease: A clinical histopathological study. Am J Gastroenterol. 2003;98:2042-2047.
- Powell EE, Cooksley WG, Hanson R, et al. The natural history of nonalcoholic steatohepatitis: A follow-up study of forty-two patients for up to 21 years. Hepatology. 1990;11:74-80.
- Fassio E, Alvarez E, Dominguez N, et al. Natural history of nonalcoholic steatohepatitis: A longitudinal study of repeat liver biopsies. Hepatology. 2004;40:820-826.
- Evans CD, Oien KA, MacSween RN, Mills PR. Non-alcoholic steatohepatitis: A common cause of progressive chronic liver injury? J Clin Pathol. 2002;55:689-692.
- Gaidos JK, Hillner BE, Sanyal AJ. A decision analysis study of the value of a liver biopsy in nonalcoholic steatohepatitis. Liver Int. 2008;28: 650-658.
- Feldstein AE, Wieckowska A, Lopez AR, et al. Cytokeratin-18 fragment levels as noninvasive biomarkers for nonalcoholic steatohepatitis: A multicenter validation study. Hepatology. 2009;50:1072-1078.
- Malik R, Chang M, Bhaskar K, et al. The clinical utility of biomarkers and the nonalcoholic steatohepatitis CRN liver biopsy scoring system in patients with nonalcoholic fatty liver disease. J Gastroenterol Hepatol. 2009;24: 564-568.
- Talwalkar JA. One step at a time: Identification and validation of biomarkers for nonalcoholic steatohepatitis. Hepatology. 2009;50:1000-1003.
- Yoneda M, Uchiyama T, Kato S, et al. Plasma Pentraxin3 is a novel marker for nonalcoholic steatohepatitis (NASH). BMC Gastroenterol. 2008; 8:53.
- Gaia S, Carenzi S, Barilli AL, et al. Reliability of transient elastography for the detection of fibrosis in non-alcoholic fatty liver disease and chronic viral hepatitis. J Hepatol. 2011;54:64-71.
- Weiner RA. Surgical treatment of non-alcoholic steatohepatitis and non-alcoholic fatty liver disease. Dig Dis. 2010;28:274-279.
- Mikuls TR, O’Dell J. The changing face of rheumatoid arthritis therapy: Results of serial surveys. Arthritis Rheum. 2000;43:464-465.
- Kremer JM, Alarcon GS, Lightfoot RW, Jr., et al. Methotrexate for rheumatoid arthritis. Suggested guidelines for monitoring liver toxicity. American College of Rheumatology. Arthritis Rheum. 1994; 37:316-328.
- Stanley DW, Yin M, Glaser KJ, Ehman RL. A protocol for assessing hepatic fibrosis in iron-overloaded liver tissue with MR elastography. Proc. Intl. Soc. Mag. Reson. Med. 2009;17:716.
Citation
MR elastography of liver disease: State of the art. Appl Radiol.
April 4, 2013