Imaging the patient with hearing loss
Images
Having trouble hearing? You’re not the only one. Over 15% of adults over the age of 18 report some degree of hearing loss. The prevalence of reported hearing loss increases among men and with increasing age.1 Presbycusis, or age-related hearing loss, is the most common cause of acquired hearing loss, with noise, otitis and cerumen impaction representing other common causes. For many forms of hearing loss, im aging is a critical step in evaluation. This article reviews the pertinent anatomy of hearing, clinical workup of acquired hearing loss, and the radiologic appearance of common entities that cause acquired hearing loss.
Normal auditory pathway
The auditory pathway is divided into the outer, middle, and inner ear and the central auditory pathway. The outer ear is composed of the auricle and external auditory canal (EAC). The tympanic membrane (TM) is located at the end of the EAC and separates the EAC from the middle ear. The middle ear contains ossicles, nerves and muscles (stapedius and tensor tympani). The inner ear consists of the cochlea, vestibule and semicircular canals encased in the bony otic capsule. The air-filled middle ear and fluid-filled inner ear are connected by two membrane-covered openings -- the oval and round windows. The central auditory pathway consists of a complex network of neurons and nuclei that connect the cochlea to the auditory cortex.
Sound waves from the external environment are collected by the EAC. They go on to impact upon the TM, causing it to vibrate. The malleus attaches to the TM at one end and articulates with the incus. The incus then articulates with the stapes, and the footplate of the stapes is attached to the oval window. Vibrations of the tympanic membrane are transmitted along these ossicles to the oval window.
The inner ear is a fluid-filled structure encased in bone, which restricts the movement of the fluid within. It is only at the membranes of the oval and round windows that an exception is made. As sound vibrations travel through the stapes and push the oval window into the inner ear, fluid waves are created in the inner ear that travel through the cochlea to dissipate at the round window. As the oval window bulges in, the round window bulges out. The motion of fluid in the cochlea stimulates hair cells in the Organ of Corti which, in turn, generate action potentials that are transmitted via the cochlear nerve through the central auditory pathway to the auditory cortex.
Types of hearing loss
Hearing loss can be classified as conductive hearing loss (CHL), sensorineural hearing loss (SNHL), or mixed hearing loss (MHL). Disruption of the transmission of sound waves from the outside environment to the cochlea will result in CHL. Common pathologies that produce CHL include otitis, TM rupture, cholesteatoma and trauma. Dysfunction of the cochlea or disruption of neural impulses in the central auditory pathway will result in SNHL. This can be seen with presbycusis, internal auditory canal (IAC) and cerebellopontine angle (CPA) tumors, labyrinthitis and CNS pathology. Mixed hearing loss occurs when components of both sensorineural and conductive hearing loss are present. Otospongiosis is a common pathology within this category.
Clinical evaluation
Initial evaluation of hearing loss often occurs in a primary care setting where a history, physical examination, questionnaires and the Whispered-Voice test are common and effective methods of screening for hearing loss. Tuning fork exams may also be utilized.
Once hearing loss is suspected, the patient should be referred for pure tone audiometry, the current gold standard in hearing evaluation. In pure tone audiometry, tones of varying frequencies and intensities are generated by an audiometer. The tones are presented to the patient via headphones when testing air conduction, and via bone vibrators when testing bone conduction. Generally, tones ranging from 250 to 8000 Hz are tested, as they cover the human speech spectrum of 500 to 4000 Hz.
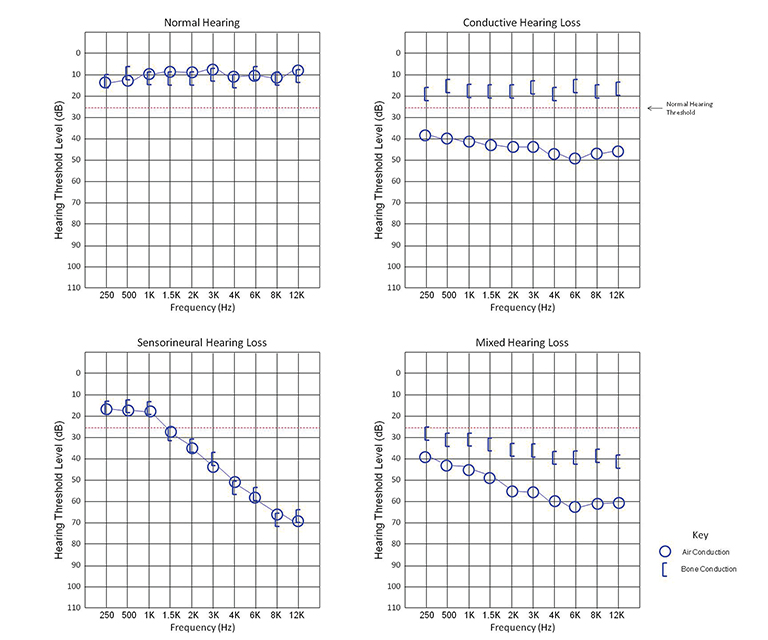
Each ear is tested separately. The intensity of the tone is decreased until an intensity threshold is reached where the patient can no longer hear the tone. Hearing loss is classified by this threshold as mild (26-40 dB), moderate (41-55 dB), moderately severe (56-70 dB), severe (71-90 dB), and profound (>90 dB). The sound intensity thresholds of several frequencies are tested to determine if the hearing loss is limited to a specific frequency range. Data is plotted on a chart termed an audiogram.
Pure CHL will present as an increased hearing threshold when testing for air conduction and a normal threshold for bone conduction. The difference between the air and bone conduction thresholds is termed the air-bone gap. In SNHL, both air and bone conduction thresholds will be elevated with a negligible air-bone gap. In MHL, both thresholds will be elevated, but an air-bone gap will be present (Figure 1).2-4
After audiometry, imaging studies may be performed to further evaluate the hearing loss. If CHL is present, a CT scan of the temporal bones is often the test of choice as it provides a superior evaluation of the bony sound conduction pathway. CT may also be the preferred imaging study in cases of MHL as it can accurately evaluate for otospongiosis. If SNHL is present, MRI is usually the preferred study as it better assesses the vestibulocochlear nerve and CNS.4 In some cases, CT and MRI will have complementary roles in the workup of hearing loss. MRI has become increasingly popular in recent years as there is appropriate and growing concern over excess radiation from diagnostic imaging.
Conductive hearing loss
Conductive hearing loss results from disruption of the transmission of sound waves from the outer environment to the inner ear. Pathologies that cause CHL will likely be evident on CT. If a patient presents with CHL and no abnormality is seen on temporal bone CT, reevaluation of the images is warranted.
Outer ear pathologies
Anything in the outer ear that prevents sound waves from reaching the tympanic membrane will produce CHL. Common causes that are clinically evident and typically do not require imaging are cerumen impaction, foreign bodies or fluid/pus, as in the case of otitis externa.
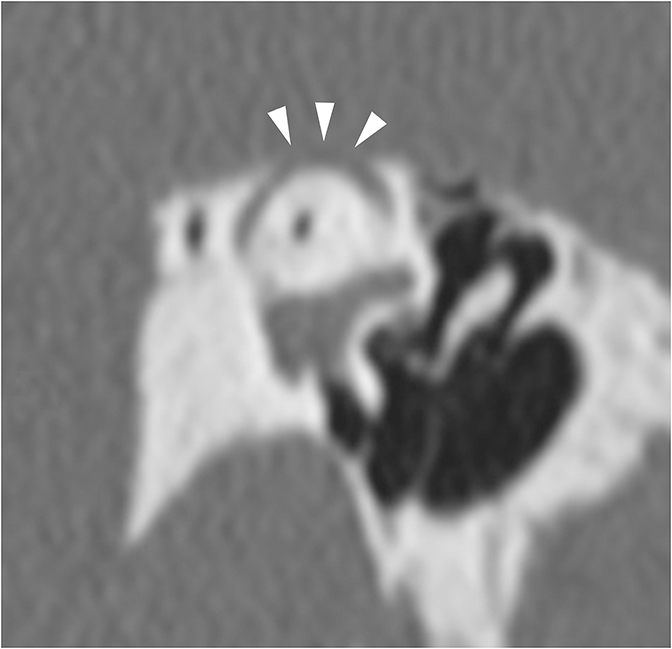
The bone that forms the wall of the inner two-thirds of the EAC may become hyperplastic and narrow or obstruct the EAC. This can be seen with both exostoses and osteomas. Exostoses are broad-based, circumferential bony lesions that are usually bilateral and associated with repeated cold water exposure (surfer’s ear). Osteomas are usually unilateral and pedunculated. The clinical and radiologic appearances of these lesions are usually distinctive.
Two pathologies which arise from the abnormal accumulation of keratinizing squamous epithelium in the EAC are keratosis obturans (KO) and cholesteatomas of the EAC (CEAC). In KO, keratin squames are shed circumferentially from the EAC epithelium, resulting in a soft tissue mass that has a layered appearance on pathology. In contrast, CEAC is a soft tissue mass containing keratin squames arranged in a random pattern. There is frequently overlap between the clinical presentation and imaging appearance of these two lesions.
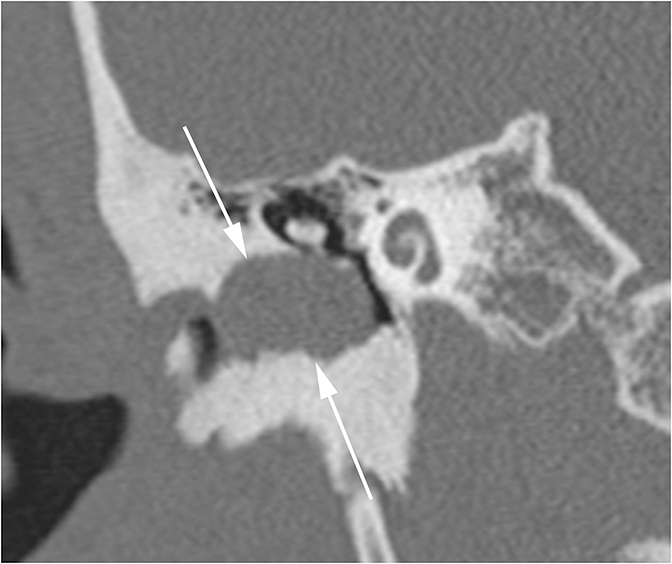
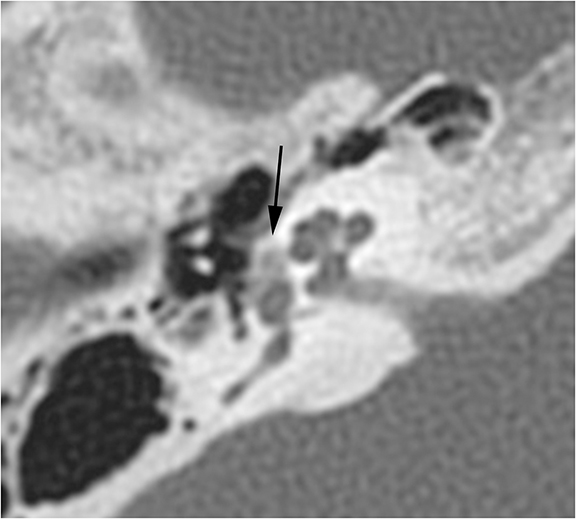
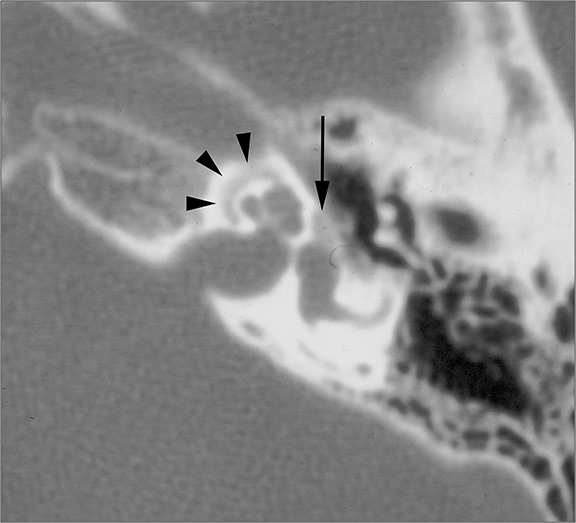
Clinically, patients with KO present under the age of 40 with acute pain, usually bilateral, and a history of bronchiectasis or sinusitis. On CT, KO will appear as a soft tissue mass which widens the EAC with smooth bony wall remodeling (Figure 2A). Treatment entails removing the keratin plug and routine cleaning of the EAC to prevent recurrence.
Patients with CEAC are typically over 40 and present with unilateral chronic, dull ear pain. CEAC will often demonstrate underlying bony necrosis or sequestration, a finding that is absent in KO. CT demonstrates a soft tissue mass in the EAC with focal bony erosion (Figure 2B), in contrast to the smooth widening seen in KO. Treatment involves surgical debridement.5
Middle ear pathologies
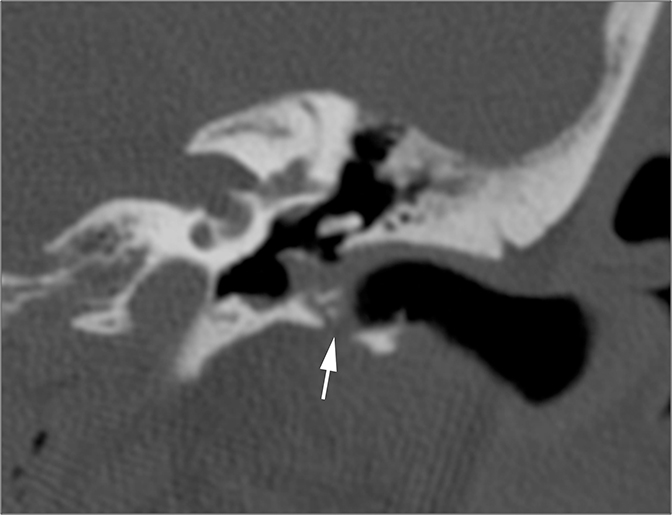
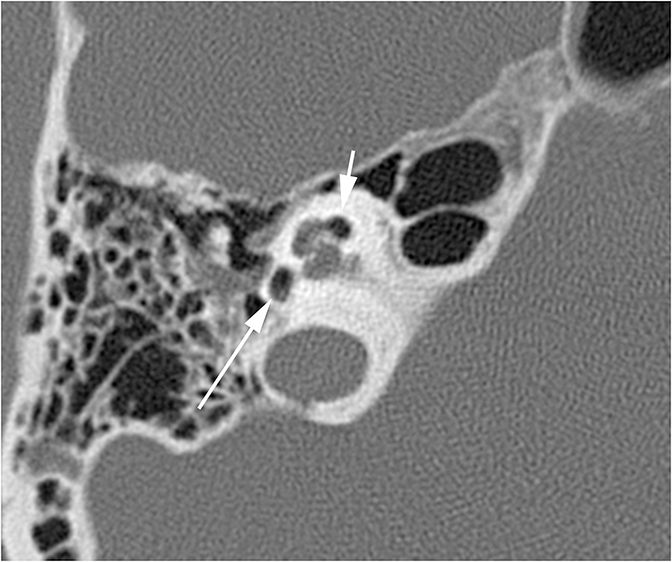
Conductive hearing loss originating from the middle ear can occur from traumatic injury to the ossicular chain or dampening of sound vibrations from soft tissue or fluid in the middle ear cavity. Traumatic CHL usually presents immediately after the trauma and can manifest itself as any number of injuries. CT scans should be carefully inspected for ossicular dislocations. The incus is the ossicle most susceptible to disruption as it has less ligamentous support than the malleus and stapes. Incudostapedial joint separation is the most common ossicular injury and is seen as widening of the joint on CT. Malleoincudal dislocation is seen as separation of the head of the malleus from the body of the incus, disrupting the normal “ice cream cone (Figure 3).” If posttraumatic hemotympanum is the cause of CHL, symptoms should improve as the hemorrhage resolves. Opacification of the middle ear cavity will be seen on CT scan.6
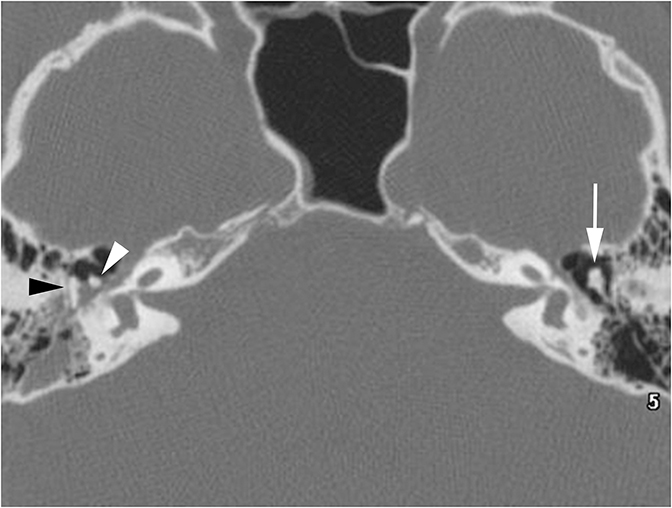
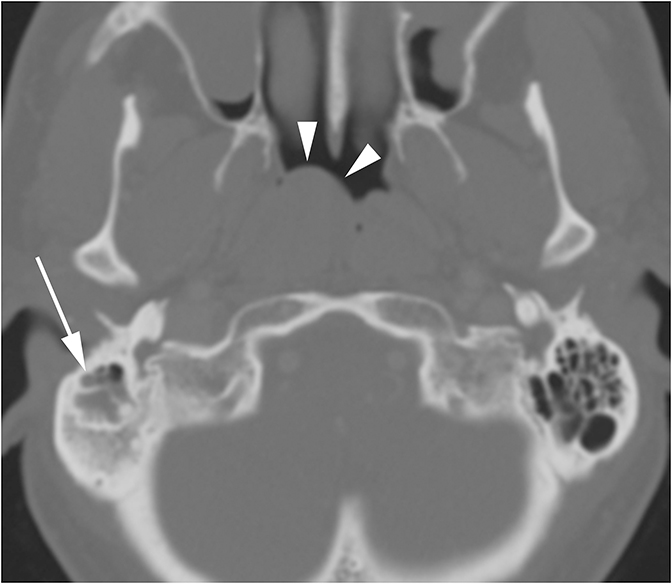
Middle ear effusions and acute otitis media (OM) will dampen vibrations of the TM and ossicles producing CHL. These processes are differentiated clinically and can easily be seen as opacification of the middle ear cavity on CT. When a unilateral middle ear or mastoid effusion is discovered in an adult, the nasopharynx should always be inspected for a nasopharyngeal mass (potentially a carcinoma) obstructing the eustachian tube (Figure 4).
Acquired cholesteatomas are common in the middle ear. There are several different theories that attempt to explain how cholesteatomas are formed, but one widely accepted mechanism involves a combination of the retraction and proliferation theories. TM retraction pockets develop due to chronic eustachian tube dysfunction and decreased middle ear pressure, often secondary to chronic OM. In a small percentage of retraction pockets, the self-cleansing mechanism of the TM may become disturbed, allowing debris to accumulate and induce an inflammatory response. This inflammation alters normal keratinocyte proliferation to promote proliferation of epithelial cones. Within the cones is disordered keratinization and desquamation which results in the accumulation of keratinized cells, or microcholesteatomas. These microcholesteatomas gradually grow and fuse with others over time, forming a clinically evident cholesteatoma.7
Middle ear cholesteatomas are classified based on their site of origin on the TM. Pars flaccida cholesteatomas are the most common and begin along the pars flaccida, grow into the epitympanum in Prussak’s space (attic cholesteatomas), lateral to the ossicular chain. Pars tensa cholesteatomas begin along the pars tensa and grow medial into the facial recess and sinus tympani (sinus cholesteatomas). Pars tensa cholesteatomas often grow medial to the ossicular chain, displacing it laterally. Both types tend to grow posteriorly into the aditus ad antrum and the mastoid antrum. It may be impossible to differentiate pars flaccida and pars tensa cholesteatomas once they reach advanced stages.8
Cholesteatomas are usually diagnosed clinically. Patients have a history of chronic OM and may present with discharge, hearing loss, and a pearly white mass behind the diseased TM. The role of imaging is to evaluate the extent of the cholesteatoma. CT will demonstrate nonspecific soft tissue density in the middle ear, the size and location of which will be determined by the type and extent of the cholesteatoma (Figure 5A-B). The radiographic feature that may distinguish cholesteatomas from other lesions is bone erosion. The scutum, ossicles, and tegmen tympani are common sites for erosion.
MRI can provide complimentary information on the lesion and aid with diagnosis in equivocal cases. The improved tissue contrast better demonstrates involvement of the facial nerve and evaluates intracranial extension. Unlike many soft tissue lesions in the middle ear, including post-surgical granulation tissue, cholesteatomas demonstrate high signal intensity (restricted diffusion) on non-echo-planar DWI sequences (Figure 5C) and do not enhance.8,9
Recurrent inflammation from chronic OM can also lead to tympanosclerosis. The chronic inflammation leads to deposition of hyalinized collagen that can calcify, often associated with calcification of the ossicular ligaments and tendons. CT will demonstrate focal or linear soft tissue deposits within the middle ear, with or without calcification (Figure 6). When extensive enough, the deposits can appear like an ill-defined mass.
An extensive array of tumors can occur in the middle ear cavity which can cause CHL by interfering with normal ossicular movement. Paragangiomas (specifically glomus tympanicum), congenital cholesteatomas, and schwannomas are the most common. All three appear as well-circumscribed lesions on CT and MRI and may be difficult to differentiate. Paragangliomas and schwannomas demonstrate avid enhancement whereas congenital cholesteatomas will not enhance, a key distinguishing feature. Glomus tympanicum is classically seen along the cochlear promontory and facial schwannomas will occur along the course of the facial nerve.8
Uncommon benign tumors include hemangiomas and middle ear adenomas. These will generally demonstrate well-circumscribed margins and may be difficult to differentiate from congenital cholesteatomas. A unique feature of facial hemangioma is its tendency to produce honeycomb-like calcification (Figure 7). Malignant tumors of the middle ear are rare and include rhabdomyosarcomas, squamous cell carcinomas, and metastases. These lesions typically have more aggressive features, including ill-defined margins and bone erosion.8
Inner ear pathologies
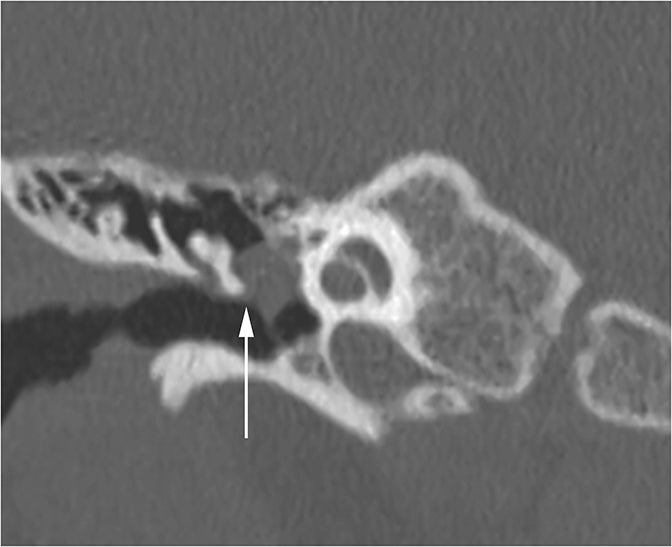
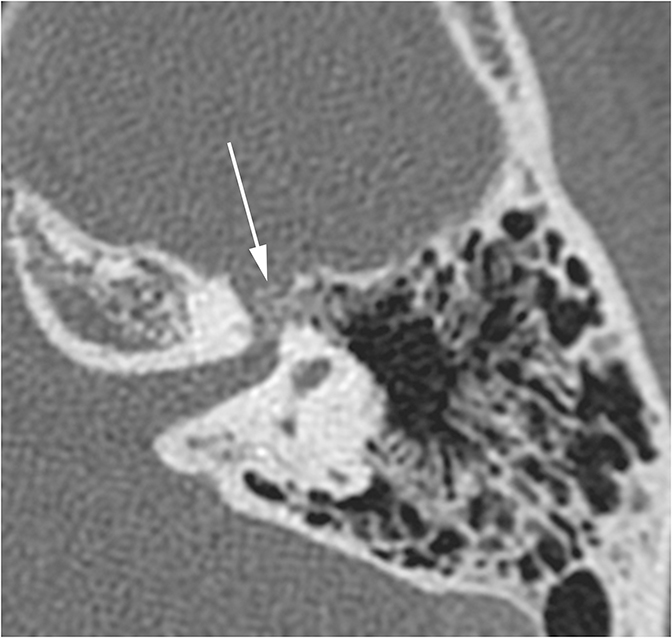
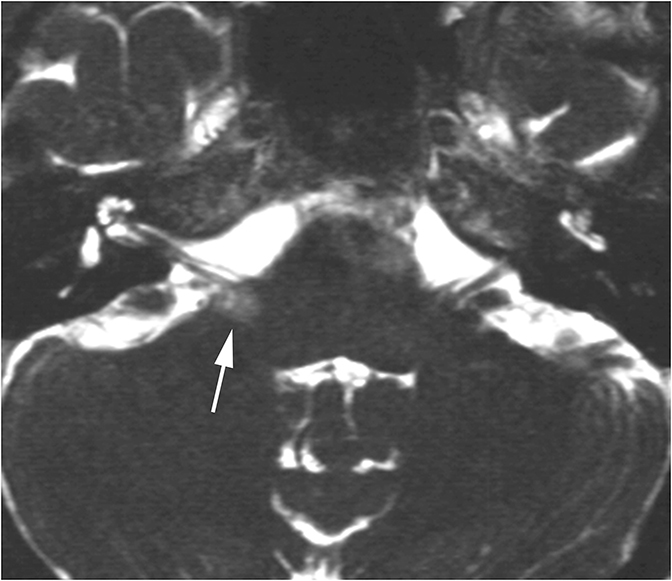
The vast majority of lesions involving the inner ear will result in SNHL or MHL. One inner ear entity has gained recognition as a cause of CHL—namely superior semicircular canal dehiscence (SSCD). In normal hearing, fluid waves travel from the oval window through the cochlea towards the round window. The bony otic capsule prohibits other fluid motion. In SSCD, there is a defect in the temporal bone overlying the superior semicircular canal (SSC), allowing the membrane covering the SSC to bulge into the middle cranial fossa. This creates a “third window” towards which fluid waves travel. Fluid waves are diverted away from the cochlea to the “third window” resulting in CHL. Additionally, when patients with SSCD are subjected to loud sounds, the conflicting fluid motion travelling through the SSC may induce vertigo or nystagmus, an entity known as Tullio Phenomenon.10 CT of SSCD demonstrates absence of the bone overlying the SSC (Figure 8).
Sensorineural hearing loss
Sensorineural hearing loss occurs from damage to the cochlea or disruption of the transmission of neural impulses from the cochlea to the auditory cortex. Presbycusis and noise-induced hearing loss are among the most common causes of SNHL, however imaging is neither indicated nor helpful as these demonstrate characteristic patient histories, audiograms, and no positive radiologic findings. If trauma is the known cause of SNHL, CT would be the appropriate initial study to evaluate the bony labyrinth. If the cause of SNHL is unknown, MRI is typically preferred to evaluate the central auditory pathway.
Inner ear pathologies
Temporal bone fractures that involve the otic capsule produce SNHL by disrupting cochlear function. While fractures are classically described anatomically as longitudinal, transverse, and oblique, more clinically relevant information is relayed by describing them as involving or sparing the otic capsule (Figure 9).
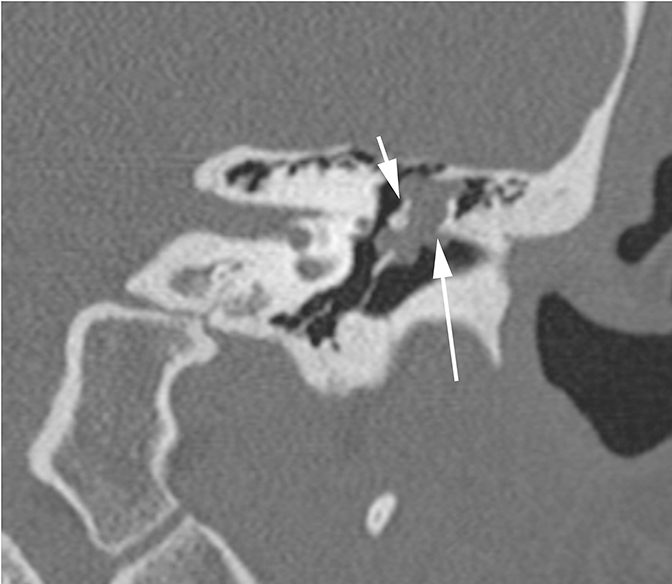
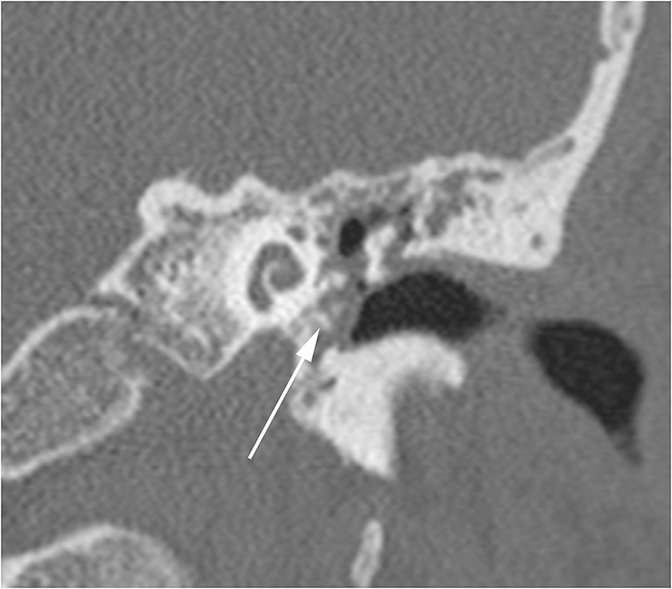
Two sequelae of trauma that can produce SNHL are perilymphatic fistula and intralabyrinthine hemorrhage. A perilymphatic fistula is an abnormal communication between the inner ear and adjacent spaces. The most common scenario is communication with the middle ear secondary to rupture of the oval or round window.6 CT will demonstrate air within the normally fluid-filled inner ear (Figure 10). Intralabyrinthine hemorrhage may be secondary to trauma or may be spontaneous and MRI is the preferred evaluation. T1 hyperintensity within the bony labyrinth on precontrast images is highly suggestive of the presence of subacute blood products (Figure 11). T2-weighted images may demonstrate signal that is brighter or darker than the surrounding inner ear fluid depending on the age of the hemorrhage. Contrast enhancement may be seen.11
On contrast-enhanced MRI obtained for SNHL, the radiologist should evaluate for enhancement within the labyrinth. A number of pathologies can present with this finding, including labyrinthitis, ischemia, and intralabyrinthine schwannomas. Vertigo is typically a prominent presenting symptom along with SNHL. Labyrinthitis refers to inflammation of the inner ear and may be secondary to viral, bacterial, or autoimmune causes. Enhancement is classically faint and involves the entire labyrinth. The enhancement can persist for months after resolution of symptoms.
Inner ear ischemia can produce enhancement that has a similar appearance to labyrinthitis. The cochlea and vestibule are supplied by a peripheral branch of the anterior inferior cerebellar artery. Ischemia of the labyrinth may be secondary to thromboembolic disease, vasculitis, or radiation-induced ischemia.12,13
In contrast to labyrinthitis and ischemia, enhancement in intralabyrinthine schwannomas tends to be more focal and intense. A corresponding hypointensity may be seen in the normally bright fluid on T2-weighted images. The location of an intralabyrinthine schwannoma is used to classify it as a vestibular, cochlear, or vestibulocochlear. There may be associated extension into the IAC or middle ear cavity.
Labyrinthitis ossificans is a fibroosseous reaction of the inner ear that most commonly occurs after infectious labyrinthitis, but can be seen with any prior inner ear insult. This should be investigated as a cause of SNHL that develops weeks to months after inner ear disease. Early on, fibrosis of the labyrinth occurs which can appear as hypointense areas on T2-weighted MRI that may enhance. As the disease progresses, it eventually calcifies and can be appreciated on CT (Figure 12). Cochlear implantation may be used to correct the SNHL in non-calcified states.13
Central auditory pathway pathologies (aka retrocochlear pathology)
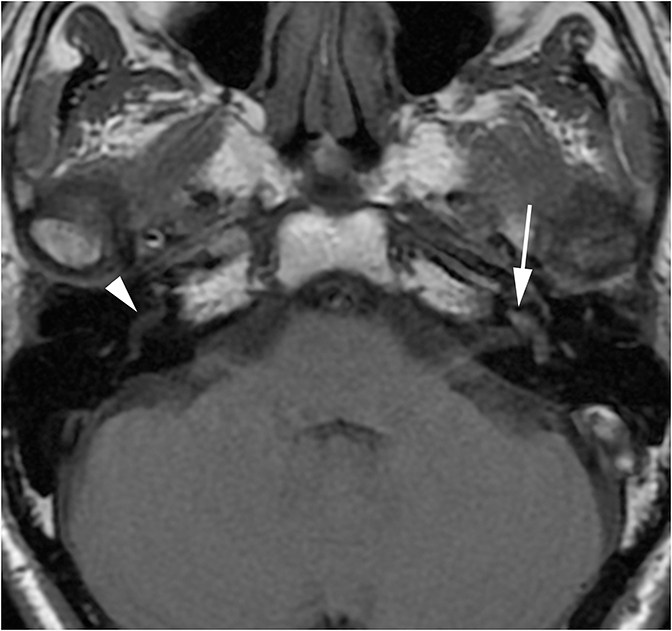
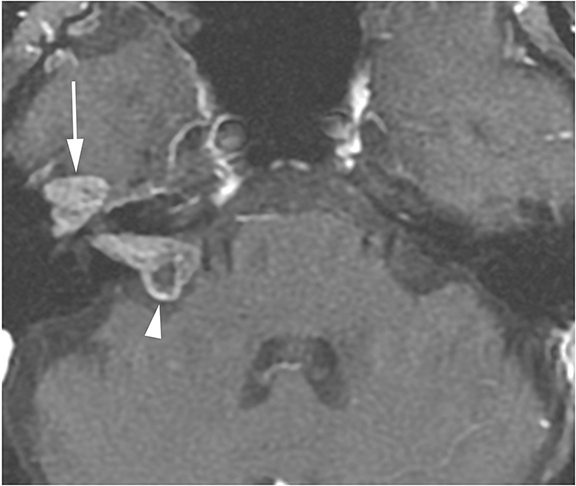
Lesions in the CPA-IAC can compress the cochlear nerve as it travels from the brainstem to the cochlea resulting in SNHL. Vestibular schwannomas are by far the most common, representing approximately 85% of CPA-IAC lesions.14 Schwannomas are benign tumors of Schwann cells of the vestibular nerve sheath. On T2-weighted MRI, they appear as hypointense, round, well-defined lesions which stand out against the background of hyperintense CSF. Vestibular schwannomas enhance homogenously when small (Figure 13A), but may develop cystic, non-enhancing areas when they grow larger. Smooth widening of the IAC may be seen with large schwannomas. Bilateral vestibular schwannomas are seen in neurofibromatosis type 2.15
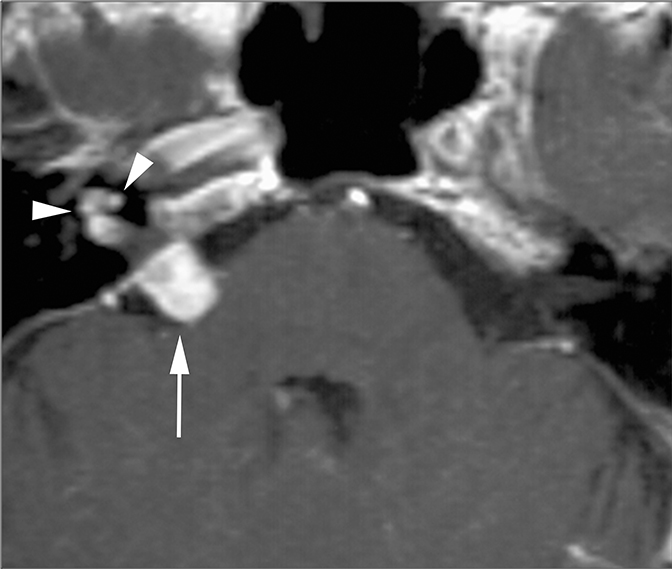
The facial and cochlear nerves also traverse the IAC. Though far less common, schwannomas of these nerves may appear similar to vestibular schwannomas. Facial schwannomas may extend onto the labyrinthine portion of the facial nerve and demonstrate associated enhancement and enlargement of the facial canal (Figure 13B). Cochlear schwannomas can demonstrate transmodiolar extension into the cochlea and present as a focal intracochlear mass with enhancement (Figure 13C).15
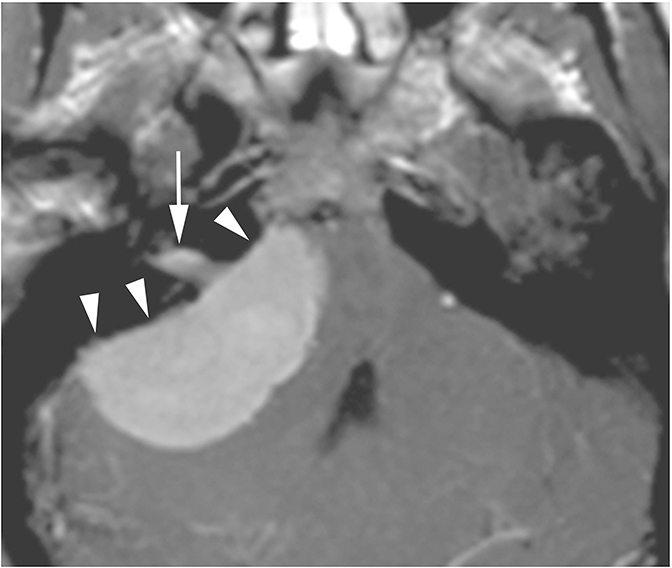
Meningiomas are the second most common lesion occurring at the CPA-IAC and can compress the cochlear nerve. These tumors demonstrate a broad-based dural attachment +/- a dural “tail” (Figure 13D). Meningiomas typically remain cisternal, however extension into the IAC or rarely IAC tumor without a cisternal component may occur, mimicking a schwannoma. Metastasis to the CPA-IAC is uncommon, but can mimic a meningioma or schwannoma.15
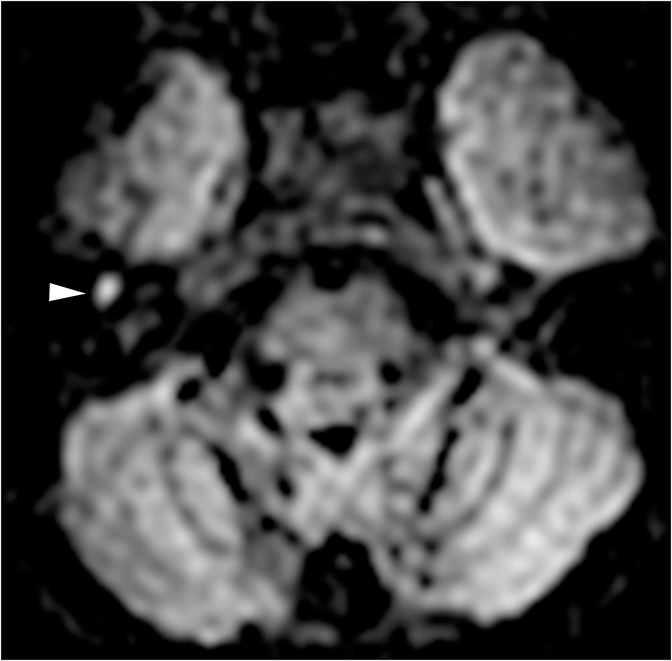
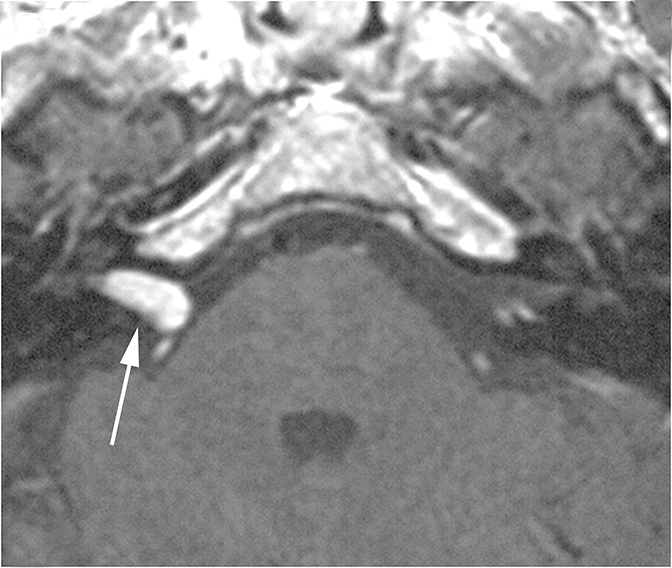
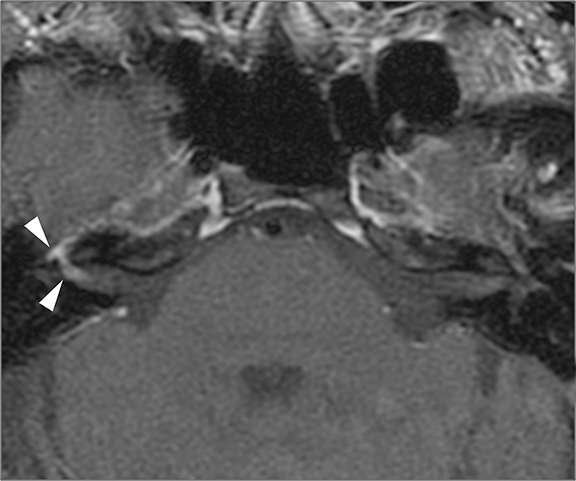
Infection or inflammation of the cochlear nerve may produce SNHL. Findings of cochlear neuritis include enhancement of the nerve on contrast-enhanced MRI without evidence of a focal mass. A small schwannoma may be difficult to distinguish radiographically from neuritis. Ramsay-Hunt syndrome describes simultaneous vestibulocochlear and facial neuritis secondary to varicella zoster infection. Patients present with painful herpetic lesions around the auricle, vertigo, tinnitus, CN VII palsies, and SNHL. Intense enhancement of the facial and vestibulocochlear nerves can be seen in the IAC (Figure 14).15
Any one of the myriad of intra-axial CNS pathologies has the potential to cause SNHL if it affects the central auditory pathway, whether it is ischemic, traumatic, neoplastic, or demyelinating. Lesions affecting the cochlear nerve or cochlear nuclei in the medulla will produce ipsilateral SNHL (Figure 15). More proximal to the cochlear nuclei, however, some ascending fibers decussate while others stay ipsilateral. This results in transmission of input from both ears to the auditory cortex in both superior temporal gyri. Therefore lesions affecting the auditory pathway more proximal to the cochlear nuclei may not produce SNHL or may produce contralateral SNHL.17
Mixed hearing loss
Otospongiosis affects the bone of the otic capsule and is a classic example of a cause of MHL. The etiology of the disease is still poorly understood, but it is associated with both genetic and environmental factors. In early stages, the normal bone is replaced with spongy, irregular bone. Most cases are diagnosed during this spongy phase, hence the name otospongiosis. Later in the disease progression, otospongiosis may recalcify and increase in density. This latter process is seen less frequently but has led to the widespread use of the term otosclerosis.
Otospongiosis commonly begins at the fissula antefenestrum, a cleft of fibrocartilaginous tissue just anterior to the oval window. Fenestral otospongiosis describes disease affecting the oval window which produces CHL. Cochlear otospongiosis occurs in the otic capsule surrounding the cochlea. The cochlear form affects the organ of Corti through a mechanism that is not completely understood resulting in SNHL. Cochlear otospongiosis rarely presents without concurrent fenestral otospongiosis and the two entities are thought to be part of a continuum. The combined CHL of fenestral otospongiosis and SNHL of cochlear otospongiosis produce MHL clinically. 13
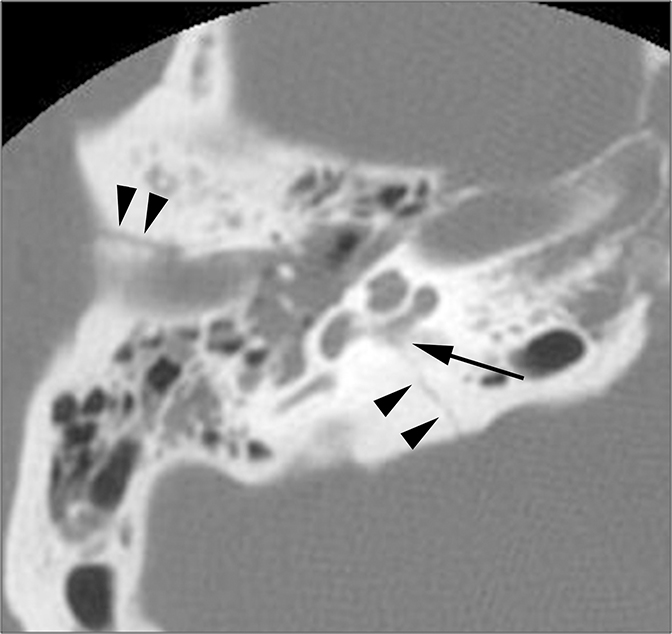
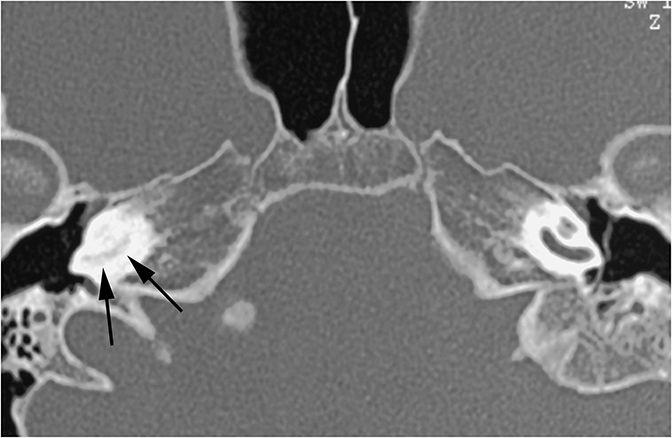
CT is the study of choice for evaluating suspected otospongiosis. The fenestral form will demonstrate a focal lytic plaque in the region of the fissula antefenestrum (Figure 16A). Thickening of the stapes footplate can sometimes be seen. The cochlear form presents with lytic plaques adjacent to or surrounding the cochlea (Figure 16B). If the plaque occurs parallel to the basal turn, it may appear as if the cochlea has a “fourth turn.”
Conclusion
Hearing loss is becoming increasingly common in today’s society. The causes are varied and proper workup and precise diagnosis helps patients. Evaluation begins with the clinician where patient history, physical exam, and audiometry can classify the hearing loss as conductive, sensorineural, or mixed. The type of hearing loss determines which imaging study will yield the most diagnostic information. Knowledge of the anatomy and common pathologies of the auditory pathway is required of radiologists to accurately interpret these studies and guide the treatment of hearing loss.
References
- Blackwell DL, Lucas JW, Clarke TC. Summary health statistics for U.S. adults: National health interview survey, 2012.Vital Health Stat. 2014;10:1-171.
- Bagai A, Thavendiranathan P, Detsky AS. Does this patient have hearing impairment? JAMA.2006;295(4):416–428.
- Walker JJ, Cleveland LM, Davis JL, Seales JS. Audiometry screening and interpretation. Am Fam Physician. 2013;87(1):41-47.
- Stachler RJ, Chandrasekhar SS, Archer SM, et al. Clinical practice guideline: sudden hearing loss. Otolaryngol Head Neck Surg. 2012;146(suppl):S1-S35.
- Persaud RA, Hajioff D, Thevasagayam MS, et al. Keratosis obturans and external ear canal cholesteatoma: how and why we should distinguish between these conditions. Clin Otolaryngol. 2004;29(6):577-581.
- Wang EY, Shatzkes D, Swartz JA. Temporal bone trauma. In: Swartz JD, Loevner LA. Imaging of the Temporal Bone. 4th ed. New York, NY: Thieme; 2009: 412-443.
- Sudhoff H , Tos M . Pathogenesis of attic cholesteatoma: clinical and immunohistochemical support for combination of retraction theory and proliferation theory. Am J Otol. 2000;21:786–792.
- Swartz JD. The middle ear and mastoid. In: Swartz JD, Loevner LA. Imaging of the Temporal Bone. 4th ed. New York, NY: Thieme; 2009: 58-246.
- Schwartz KM, Lane JI, Bolster BD Jr, Neff BA. The utility of diffusion-weighted imaging for cholesteatoma evaluation. AJNR Am J Neuroradiol. 2011;32:430-436.
- Rosowski JJ, Songer JE, Nakajima HH, et al. Clinical, experimental, and theoretical investigations of the effect of superior semicircular canal dehiscence on hearing mechanisms. Otol Neurotol. 2004;25:323-332.
- Hegarty JL, Patel S, Fischbein N, et al. The value of enhanced magnetic resonance imaging in the evaluation of endocochlear disease. Laryngoscope. 2002 ;112(1):8-17.
- Mafee MF. MR imaging of intralabyrinthine schwannoma, labyrinthitis and other labyrinthine pathology. Otolaryngol Clin North Am. 1995;28:407-430.
- Swartz JD, Mukherji SK. The Inner Ear and Otodystrophies. In: Swartz JD, Loevner LA. Imaging of the Temporal Bone. 4th ed. New York, NY:Thieme; 2009: 298-411.
- Hasso AN, Smith DS. The cerebellopontine angle. Semin Ultrasound CT MR. 1989;10:280–301.
- Glastonbury CM. The vestibulocochlear nerve, with an emphasis on the normal and disease internal auditory canal and cerebellopontine angle. In: Swartz JD, Loevner LA. Imaging of the Temporal Bone. 4th ed. New York, NY: Thieme; 2009:480-558.
- Armington WG, Harnsberger HR, Smoker WR, Osborn AG. Normal and diseased acoustic pathway: Evaluation with MR imaging. Radiology. 1988;167:509-515.
Citation
Chen D, Phillips CD.Imaging the patient with hearing loss. Appl Radiol. 2015; (4):20-25.
September 2, 2015