Imaging of hydrocephalus
Images
This article is accredited for one SA-CME credit. Visit appliedradiology.org/SAM2 for full SA-CME information.
Hydrocephalus is a common and important process, a mechanical complication of many different pathologic conditions and a disease process itself. The morphologic features are easily recognizable, but the pathophysiology remains incompletely understood. Current treatments are limited and invasive: cerebrospinal fluid (CSF) diversion via catheter or endoscopic third ventriculostomy (ETV). Further research and improved understanding of the pathophysiology is necessary to expand and refine treatment options and optimize patient outcomes.
Hydrocephalus can be acute or chronic and has a myriad of causes, with different etiologies primarily affecting children, young and middle age adults, and the elderly. For this reason, the scientific community has yet to produce a universally accepted definition. The most commonly accepted definition of hydrocephalus is “…active distention of the ventricular system of the brain resulting from inadequate passage of CSF from its point of origin within the cerebral ventricles to its point of absorption into the systemic circulation.1 This definition has limitations, but is simple and supports the idea that hydrocephalus is an active process, produced by obstruction along the course of CSF flow, that can be evaluated with morphologic or physiologic imaging.
CSF Flow dynamics and hydrocephalus
The traditional bulk flow model of CSF posits that CSF is secreted by choroid plexus epithelium in the ventricles, flows through the ventricular system, enters the spinal and cerebral subarachnoid spaces, and is absorbed by the arachnoid granulations into the superior sagittal sinus, to enter the systemic venous system. We now know that brain interstitial fluid is the source of 30-40% of CSF2 and 20-40% is absorbed by cranial and spinal nerve sheaths and at the cribriform plate rather than arachnoid granulations.2,3
Hemodynamic factors also contribute to this complex picture. The Monro-Kellie doctrine states that changes in intracranial blood, CSF, or parenchymal volume require reciprocal changes in one or both of the other factors. Beyond a certain point of compliance, compensation fails and there is an exponential rise in intracranial pressure (ICP). The impact of blood flow is substantially greater than secretion and accumulation of CSF. In the acute setting of trauma, hemorrhage, or stroke, CSF drainage can reduce ICP, but accumulation in the form of hydrocephalus is rarely a primary cause of increased ICP. In the chronic setting, arterial systolic pressure waves that are normally dissipated in the subarachnoid spaces affect the brain based upon age-dependent changes in brain compliance, likely contributing to idiopathic hydrocephalus in infants, idiopathic intracranial hypertension in young adults, and idiopathic normal pressure hydrocephalus in the elderly.4,5,6
The discovery of aquaporins (AQP), water channels expressed in the central nervous systems adds an additional layer of complexity to the process. AQP-4 channels expressed in the ependymal cells and subependymal astrocytic endfeet lining the ventricles facilitate bidirectional transport of water between the interstitial and CSF spaces.7 Water and solutes flow and diffuse between the ventricles and parenchyma depending upon regional concentration and hydrostatic pressure gradients. Iliff and colleagues hypothesize that a “glymphatic” system facilitates directional CSF flow through the brain serving as a type of lymphatic system to clear metabolites and waste.8 CSF flows from perivascular spaces (PVS) surrounding end arterioles, into the interstitial space, and out through the PVS surrounding the venules. AQP-4 channel expression is key to the directionality of the process, and disordered expression with aging, cerebrovascular disease, stroke and traumatic brain injury impairs parenchymal waste removal, contributing to neurodegeneration.
Classification of hydrocephalus
Walter Dandy first described the basic mechanism and classification of hydrocephalus as obstructive or non-obstructive in 1913. Despite advances in understanding of the underlying process, current classification systems still rely upon Dandy’s classification scheme.9
Nearly all hydrocephalus is due to CSF obstruction at some point between the ventricles and the systemic venous circulation. The point of obstruction in non-communicating hydrocephalus may lie within the ventricles or connecting channels or outlet foramen (non-communicating intraventricular hydrocephalus) or at the level of the arachnoid granulations (non-communicating extra-ventricular hydrocephalus). Normal pressure hydrocephalus, low pressure hydrocephalus, and CSF overproduction by choroid plexus hyperplasia or tumor are the three causes of communicating hydrocephalus (Table 1).
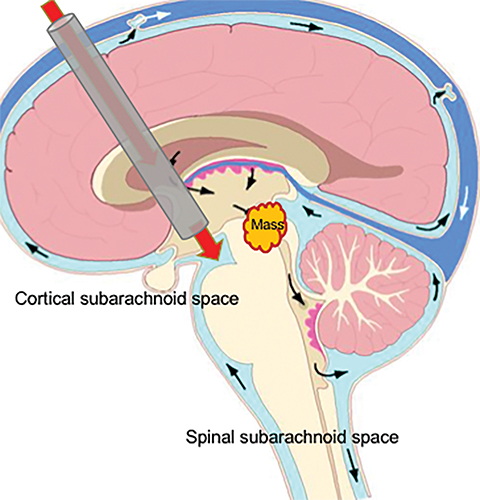
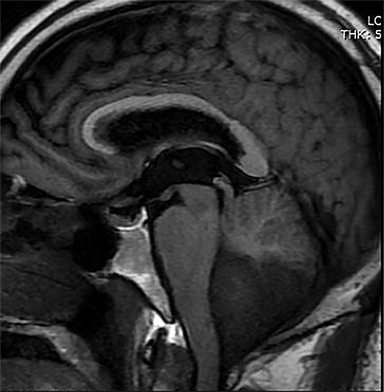
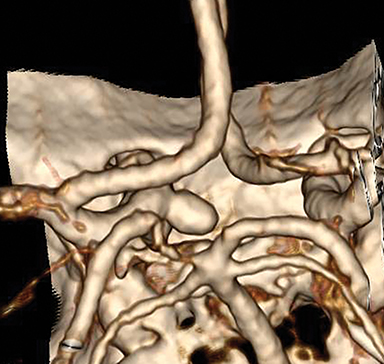
Classification is important as it informs optimal treatment. Hydrocephalus caused by obstruction of the ventricular system at a point between the third ventricle and cortical subarachnoid space can be treated with a ventricular shunt or by endoscopic third ventriculostomy (ETV). This procedure bypasses the obstruction, decompressing the obstructed ventricles by creating a direct connection with the cortical subarachnoid space in the interpeduncular and prepontine cisterns via fenestration of the third ventricular floor (Figure 1). ETV success varies with age and hydrocephalus etiology. The overall complication rate is 2-15%. Most complications are immediate and minor; in the long term, ETV confers a greater survival advantage than shunting.10
The primary treatment of hydrocephalus caused by obstruction beyond the level of the fourth ventricular outlet is ventricular shunting. The most common complications of shunting are obstruction and infection. Infection rate is approximately 5-9% per procedure, usually within three months of placement.6 Estimated thirty-year mortality related to shunt placement is 5-10%.11
Non-communicating intraventricular hydrocephalus
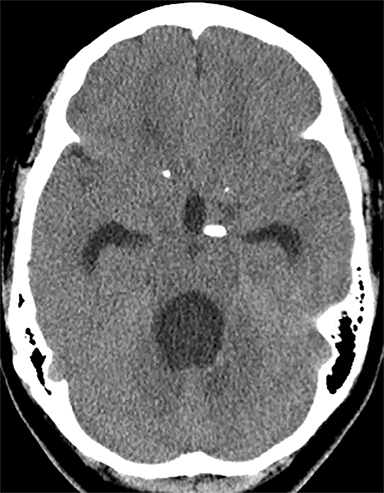
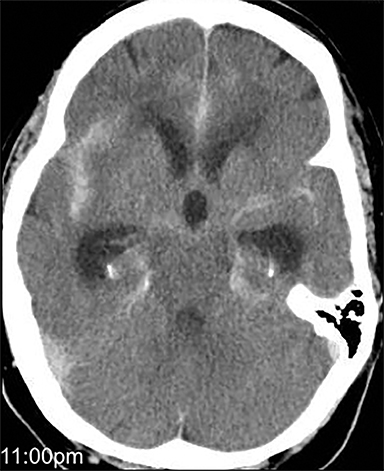
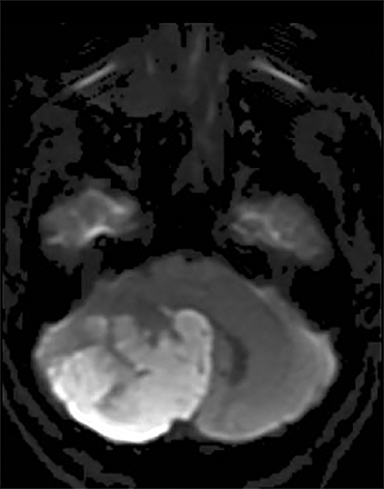
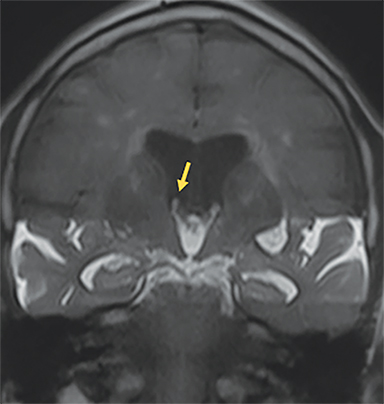
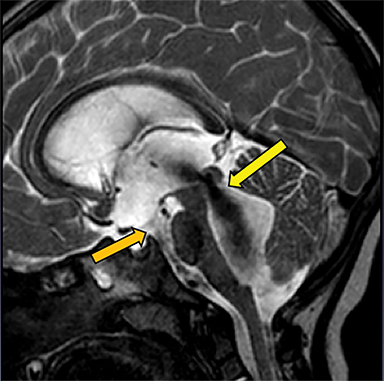
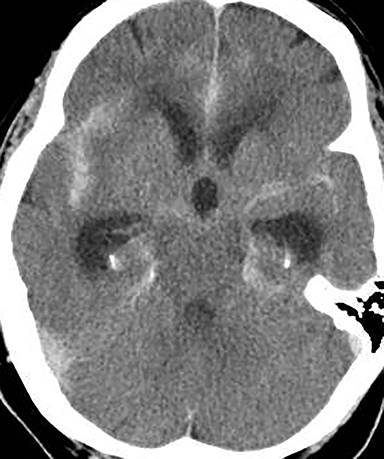
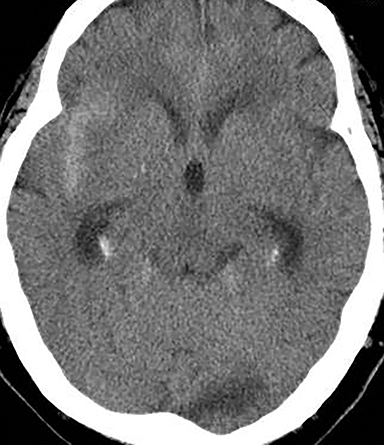
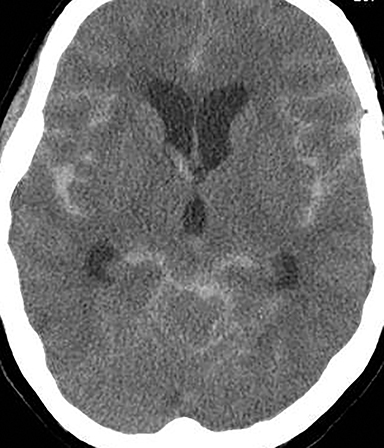
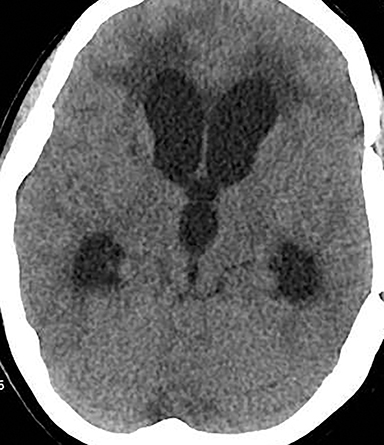
Obstruction of CSF flow within the ventricular system may be produced by neoplasms, intraventricular cysts or infections, webs or membranes, or aqueductal stenosis (Figure 2). More commonly, parenchymal masses, hemorrhage, infarct, and other processes that produce extrinsic compression upon the ventricles or connecting channels produce obstructive hydrocephalus (Figure 3).
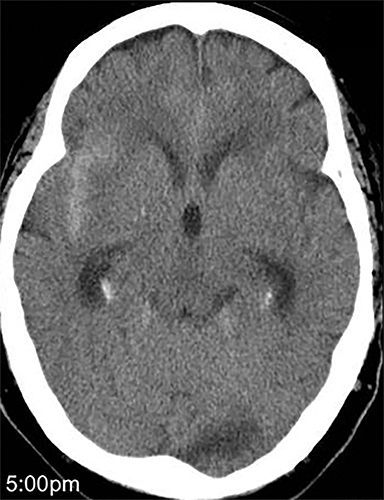
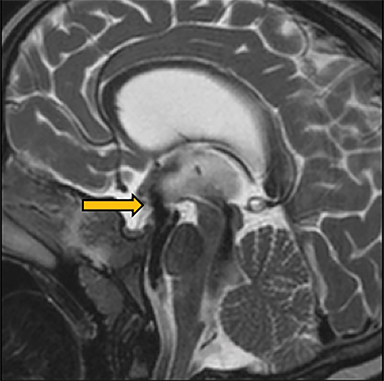
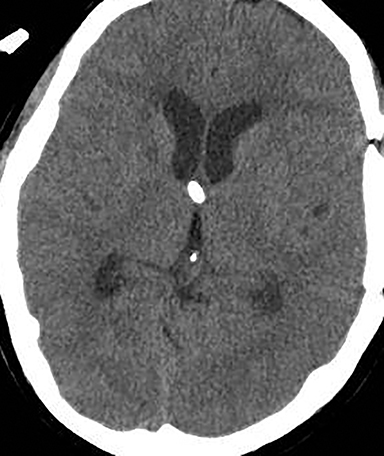
Acute obstructive hydrocephalus can progress rapidly, resulting in herniation and death within hours(Figure 4). The temporal horns have the greatest capacitance of the ventricular system, and enlargement is the earliest sign of hydrocephalus.12 Rounding of the frontal horns and bulging of the third ventricle floor are additional early findings. Periventricular interstitial edema is visible on CT, but better characterized on the MR FLAIR sequence. In acute obstruction, CSF secretion continues, and low density (CT) or high intensity (FLAIR) signal around the ventricles is thought to reflect some combination of CSF trapped within the surrounding brain parenchyma, unable to flow into the high-pressure ventricles, and outward, transependymal flow from the high-pressure ventricles into the parenchyma. With time the ventricles compensate: ventricular enlargement decreases the pressure gradient, the periventricular edema resolves, and signal abnormality on imaging normalizes.
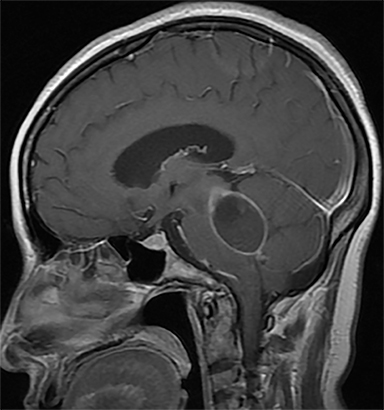
Treatment planning for patients with intraventricular obstruction benefits from high-resolution 3D MRI sequences that facilitate detection of small webs and membranes that may be occult on conventional sequences. Morphologic evaluation is optimized with 3D cisternographic imaging sequences (CISS-Siemens, FIESTA-GE) sequences. Morphology and physiology can be studied with T2-weighted 3D fast /turbo spin echo techniques SPACE-Siemens, CUBE-GE), which manifest signal changes associated with flowing CSF in patent channels, such as the cerebral aqueduct and a functioning third ventriculostomy. A relatively new technique, time-spatial labeling inversion pulse (TimeSLIP, Toshiba) tags CSF spins in a manner analogous to arterial spin labeling, allowing visualization of CSF movement over a short distance (Figure 5).13 This technique can be used to determine patency of CSF channels and ventriculostomies.
High-resolution imaging is vital for the pre-operative evaluation for ETV. The radiologist must report the presence of the Liliquist membrane and other thin membranes and webs in the interpeduncular and prepontine cisterns, as non-recognition may result in ETV failure. Dincer et al studied 134 cases of hydrocephalus with preoperative evaluation using conventional and high resolution sequences.14 They found that 20% of cases originally diagnosed as communicating hydrocephalus demonstrated small intraventricular webs on 3D CISS images. In 32% of cases, CISS images changed the treatment plan: an ETV was performed in patients originally planned for shunting.
In addition, the radiologist should report the location of the basilar artery in relation to the third ventricular floor to prevent inadvertent injury. The endoscopic surgical trajectory extends from the frontal horn, through the foramen of Monro to the third ventricle. The surgeon pierces the third ventricular floor, blind to subjacent structures. Post-operative evaluation of ventriculostomy patency and evaluation in the setting of recurrent symptoms also benefits from MR imaging. High resolution sequences such as SPACE, phase contrast MRI, or TimeSLIP demonstrate CSF flow or obstruction at the surgical site (Figure 6).
Non-communicating extraventricular hydrocephalus
The term of “communicating” hydrocephalus is now controversial: the prevailing thought that nearly all hydrocephalus is obstructive (non-communicating) has limited the entities that fall under this heading to normal pressure hydrocephalus, low pressure hydrocephalus, and CSF overproduction. Pathologies that produce CSF obstruction between the fourth ventricular outlet and arachnoid granulations fall under the heading of “non-communicating extra-ventricular” hydrocephalus. Hemorrhage, infection, and leptomeningeal spread of tumor are the most common etiologies that result in this type of hydrocephalus.
Subarachnoid and Intraventricular hemorrhage
Post-hemorrhagic hydrocephalus may be the result of subarachnoid hemorrhage, intraventricular hemorrhage, or traumatic brain injury. Hydrocephalus in these cases increases morbidity and disability independent of the inciting event. Nearly all patients require surgical shunt placement emergently, and many require permanent ventricular catheters.15
Vasospasm, re-hemorrhage, and hydrocephalus are the most common complications of aneurysmal subarachnoid hemorrhage (aSAH). aSAH results in acute and/or chronic hydrocephalus in up to 37% of cases and worsens prognosis.15,16 Aneurysm rupture in the posterior circulation more frequently results in hydrocephalus than rupture in the anterior circulation (Figure 7).17
In the acute phase, blood obstructs the ventricles, outflow channels, basilar cisterns, and arachnoid granulations, impairing CSF flow and absorption. Hydrocephalus can develop within hours, requiring vigilant observation, liberal imaging, and rapid interpretation and communication of imaging findings. In the chronic phase, hemosiderin, fibrosis, and scarring obstructs CSF flow primarily over the convexities at the arachnoid granulations, but also within the basilar cisterns and outflow channels (Figure 8).
Intraventricular hemorrhage and traumatic brain injury produce hydrocephalus by similar mechanisms. Forty percent of patients with spontaneous intracranial hemorrhage have extension into the ventricular system, and of these, 51% to 89% will develop hydrocephalus.15
Extraventricular drainage catheters are used to lower intracranial pressure, but do not inhibit the circulation of blood products or clot production, and therefore do not reduce the incidence of post-hemorrhagic hydrocephalus. Trials of low dose intraventricular thrombolysis with recombinant tissue-type plasminogen activator (rtPA) have shown mixed results in preventing hydrocephalus and improving neurological outcomes.18,19,20
Infection and carcinomatosis
Hydrocephalus complicates bacterial meningitis in approximately 5% of cases, and is associated with substantially increased morbidity and mortality.21,22 In this case, exudates block the arachnoid granulations, and less commonly, the outlet foramen. High CSF protein levels may also contribute to decreased absorption at the arachnoid granulations.23
Lung and breast carcinoma and melanoma are the most common malignancies to produce leptomeningeal carcinomatosis. The incidence of carcinomatosis in patients with solid tumors is approximately 5%, and of these, 25% develop hydrocephalus.24,25,26 Survival at this point is limited, but prolonged by shunting.24 Carcinomatosis produces hydrocephalus in a similar manner as infection, obstructing CSF outflow at the level of the granulations.
Communicating hydrocephalus
True communicating hydrocephalus has no obstruction within the ventricular system, within the basilar cisterns, cortical subarachnoid space, or at the arachnoid granulations. Treatment depends upon etiology.
CSF Overproduction
CSF overproduction by a choroid plexus tumor or hyperplasia is exceedingly rare. In this case, the volume of CSF produced exceeds the absorption capacity at the arachnoid granulations, across aquaporin receptors in the ventricular walls, and via alternative drainage routes, such as spinal and cranial nerve sheaths.
Low pressure hydrocephalus
The syndrome of low pressure hydrocephalus (SILPAH) is also extremely rare. In this case, complete obstruction of the ventricular system results in disconnection from the cortical subarachnoid space (CSAS).27,28,29 CSF in the CSAS drains into the SSS, with resultant outward expansion of the brain parenchyma and enlargement of the ventricles. The intracranial pressure is low: CSF does not drain from the ventricular shunt. Treatment involves lowering the EVD setting to subzero pressure to force drainage, binding of the neck to increase sinus pressure, and eventual reconnection of the ventricular system with the CSAS via ETV.30
Normal pressure hydrocephalus
Normal pressure hydrocephalus (NPH) was first described by Hakim and Adams in 1965 as a syndrome of gait apraxia, dementia and urinary incontinence in a patient with imaging findings of enlarged ventricles and normal CSF pressure measured with lumbar puncture.31 Prevalence increases with age, reaching 6% in individuals over 80 years old.32 It is one of the few potentially treatable causes of dementia. Clinical diagnosis can be difficult, as symptoms overlap with other neurodegenerative diseases, primarily Alzheimer’s disease (AD).
Since the original description, researchers have discovered numerous additional physiologic changes associated with this disease. In the majority of NPH cases there is resistance to CSF reabsorption at the arachnoid granulations33 with consequent reversal of flow into the periventricular parenchyma.34 Ventricular pressure is normal, but pulse pressure is increased 6-8 times normal, resulting in hyperdynamic flow through the cerebral aqueduct.35,36 PET and SPECT images show globally decreased cerebral blood flow.37
Diagnosis and treatment of NPH remains controversial, despite decades of study. Management decisions balance the risks associated with CSF diversion surgery in patients of this age, and often with multiple comorbidities, with the potential benefit of alleviating symptoms. Not all patients with NPH benefit from CSF diversion: late in the disease course ventricular dilation progresses to atrophy and damage is irreversible. For this reason, the primary goal of imaging research is to identify patients who will benefit from surgery.
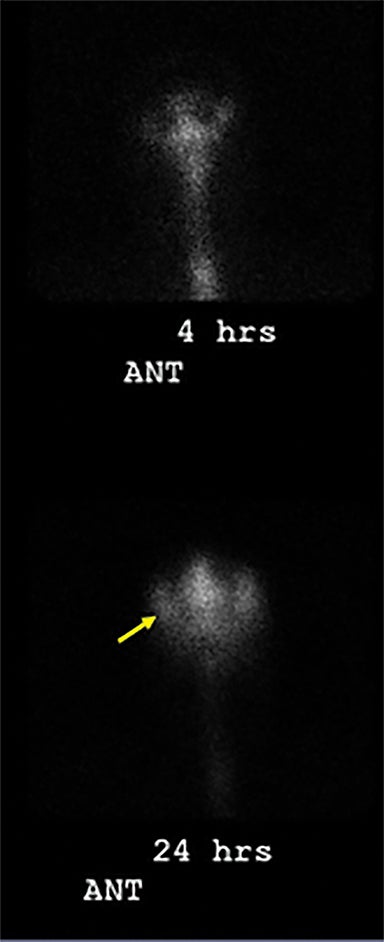
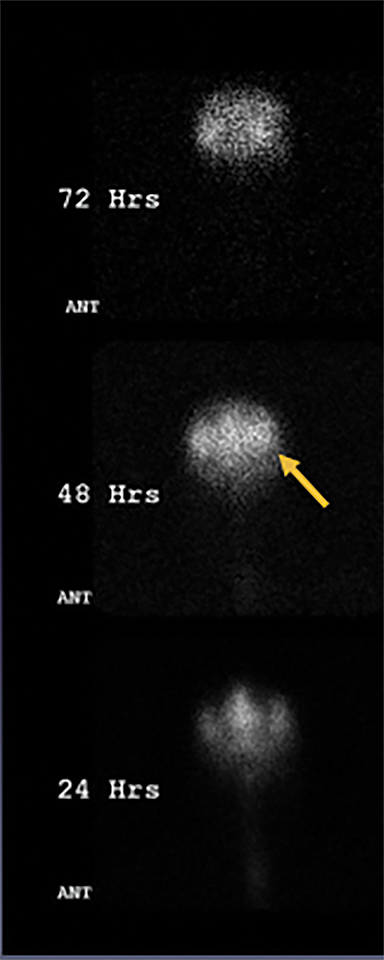
Nuclear cisternography, commonly utilized in the assessment of NPH and CSF flow disorders in the past, is less often performed today, given improved imaging techniques. This procedure entails injecting a radiopharmaceutical, typically Indium 111 DTPA, into the subarachnoid space by lumbar puncture and imaging flow at subsequent time points. Normally the radiotracer will reach the basilar cisterns in 1 to 3 hours and flow over the cerebral convexities within 12 to 24 hours. In the setting of NPH, the radiotracer flows preferentially into the ventricles, and remains on delayed imaging at 24 to 48 hours. This pattern suggests impairment of flow to the cortical subarachnoid space or resistance to absorption at the arachnoid granulations (Figure 9). No studies have shown benefit of nuclear cisternography in predicting shunt responsiveness.
Phase contrast MRI provides a quantitative measure of aqueductal stroke volume. Bradley et al found that elevated stroke volume, twice that of normal individuals, was associated with positive shunt response.38 Dixon et al found that phase contrast measurement of CSF flow rate did not inform shunt responsiveness, but did support diagnosis in clinically questionable cases.39 Many additional studies have produced mixed results regarding the utility of this technique. Measurements differ between MRI scanners and calibration with normal individuals must precede use on patients.
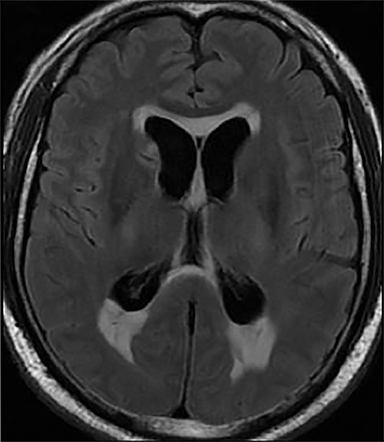
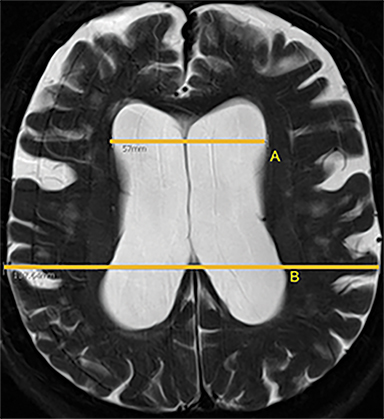
Morphologic imaging features are most commonly used in daily practice to support clinical suspicion of NPH. The Evans index (EI) is the oldest and most frequently used measure of ventricular enlargement. This is calculated as he ratio of the maximum diameter of the frontal horns to the maximum diameter of the skull on axial CT or MR images.40 An EI of 0.3 or greater suggests NPH. Specificity is low, as EI is also increased in the setting of parenchymal atrophy due to other causes.
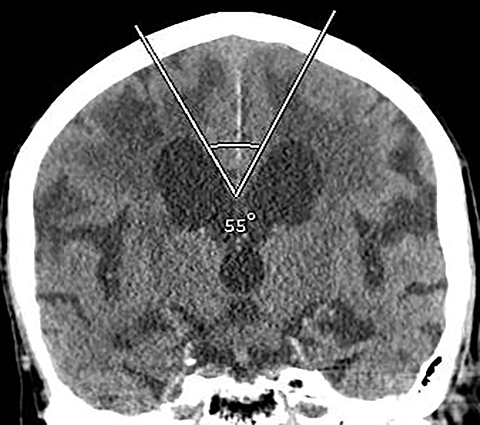
The callosal angle (CA), the angle between the frontal horns measured in the coronal plane at the level of the posterior commissure, is decreased in NPH. A CA less than 90 degrees may differentiate NPH from AD.41 Virhammar et al found that shunt response correlated with a preoperative CA significantly smaller than that of non-responders: 59 degrees versus 68 degrees. CA of less than 63 degrees had the best prognostic accuracy.42
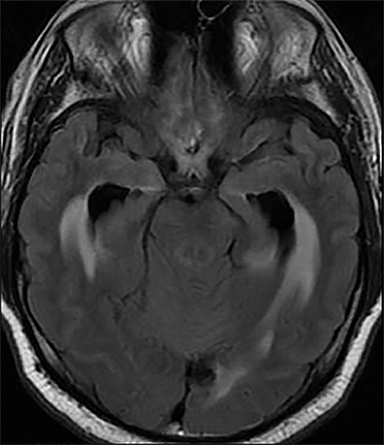
Disproportionally enlarged subarachnoid space hydrocephalus (DESH) describes narrowed frontoparietal sulci that abut the superior sagittal sinus and widening and upward displacement of the Sylvian fissures. MR imaging and DESH were utilized in the Study of Idiopathic Normal Pressure Hydrocephalus on Neurological Improvement (SINPHONI) trial, with DESH showing a high positive predictive value in identifying shunt responsive patients (Figure 10).43,44
Volumetric segmentation is commonly used in the evaluation of global and regional structural changes in AD. Miskin et al tested the utility of volumetric assessment versus CA and EI in discriminating between individuals with NPH, AD, and normal age matched controls.45 A combination of CA and EI demonstrated 89.6 - 93.4% accuracy for differentiating patients with NPH from those without it. In a three-way analysis discriminating between NPH, AD and healthy controls, volumetric assessment was superior to combined CA and EI with overall classification accuracy of 88.7% versus 71.7%. Currently, morphologic imaging with simple cutoff criteria serves as a useful screening tool for suspected NPH. As automated volumetric methods become more widely available, these metrics may provide greater diagnostic utility.
Conclusion
Despite over a century of study, the pathophysiology of hydrocephalus remains incompletely understood. Radiologists play a vital role in diagnosis, classification, and treatment planning. Identification of noninvasive imaging biomarkers for shunt-responsive NPH is an active area of radiology research that has produced novel imaging techniques and insights into CSF flow dynamics. Further study is necessary to expand treatment options and optimize patient outcomes.
References
- Rekate HL. A consensus on the classification of hydrocephalus: its utility in the assessment of abnormalities of cerebrospinal fluid dynamics. Childs Nerv Syst. 2011;27(10):1535-1541.
- Raybaud C. MR assessment of pediatric hydrocephalus: a road map. Childs Nerv Syst. 2016; 32(1):19-41.
- Bradley WG. CSF flow in the brain in the context of normal pressure hydrocephalus. AJNR Am J Neuroradiol. 2015;36(5):831-838.
- Bateman GA, Smith RL, Siddique SH. Idiopathic hydrocephalus in children and idiopathic intracranial hypertension in adults: two manifestations of the same pathophysiological process? J Neurosurg. 2007;107(6 Suppl):439-444.
- Wagshul ME, Eide PK, Madsen JR. The pulsating brain: A review of experimental and clinical studies of intracranial pulsatility. Fluids Barriers CNS. 2011;8(1):5.
- Kahle KT, Kulkarni AV, Limbrick DD, et al. Hydrocephalus in children. Lancet. 2016;387 (10020):788-799.
- Desai B, Hsu Y, Schneller B, et al. Hydrocephalus: the role of cerebral aquaporin-4 channels and computational modeling considerations of cerebrospinal fluid. Neurosurg Focus. 2016;41(3):E8.
- Iliff JJ, Wang M, Liao Y, et al. A paravascular pathway facilitates CSF flow through the brain parenchyma and the clearance of interstitial solutes, including amyloid ß. Sci Transl Med. 2012; 4(147):147ra111.
- Dandy WE, Blackfan KD. Internal hydrocephalus: an experimental, clinical and pathological study. Am J Dis Child. 1914;8(6):406-482.
- Kulkarni AV, Drake JM, Kestle JR, et al. Endoscopic third ventriculostomy vs cerebrospinal fluid shunt in the treatment of hydrocephalus in children: a propensity score-adjusted analysis. Neurosurgery. 2010;67(3):588-593.
- Vinchon M, Baroncini M, Delestret I. Adult outcome of pediatric hydrocephalus. Childs Nerv Syst. 2012;28(6):847-854.
- Rekate HL, Blitz AM. Hydrocephalus in children. Handb Clin Neurol. 2016;136:1261-1273.
- Yamada S, Miyazaki M, Kanazawa H, et al. Visualization of cerebrospinal fluid movement with spin labeling at MR imaging: preliminary results in normal and pathophysiologic conditions. Radiology. 2008;249(2):644-652.
- Dinçer A, Kohan S, Ozek MM. Is all “communicating” hydrocephalus really communicating? Prospective study on the value of 3D-constructive interference in steady state sequence at 3T. AJNR Am J Neuroradiol. 2009;30(10):1898-1906.
- Chen Q, Feng Z, Tan Q, et al. Post-hemorrhagic hydrocephalus: Recent advances and new therapeutic insights. J Neurol Sci. 2017;375:220-230.
- Yu H, Zhan R, Wen L, et al. The relationship between risk factors and prognostic factors in patients with shunt-dependent hydrocephalus after aneurysmal subarachnoid hemorrhage. J Craniofac Surg. 2014;25(3):902-906.
- Germanwala AV, Huang J, Tamargo RJ. Hydrocephalus after aneurysmal subarachnoid hemorrhage. Neurosurg Clin N Am. 2010;21(2):263-270.
- Morgan T, Awad I, Keyl P, et al. Preliminary report of the clot lysis evaluating accelerated resolution of intraventricular hemorrhage (CLEAR-IVH) clinical trial. Acta Neurochir Suppl. 2008;105:217-220.
- Naff N, Williams MA, Keyl PM, et al. Low-dose recombinant tissue-type plasminogen activator enhances clot resolution in brain hemorrhage: the intraventricular hemorrhage thrombolysis trial. Stroke. 2011;42(11):3009-3016
- Ziai WC, Tuhrim S, Lane K, et al. A multicenter, randomized, double-blinded, placebo-controlled phase III study of Clot Lysis Evaluation of Accelerated Resolution of intraventricular hemorrhage (CLEAR III). Int J Stroke. 2014;9(4):536-542.
- Bodilsen J, Schønheyder HC, Nielsen H. Hydrocephalus is a rare outcome in community-acquired bacterial meningitis in adults: a retrospective analysis. BMC Infect Dis. 2013;13:321.
- Kasanmoentalib ES, Brouwer MC, Van der ende A, et al. Hydrocephalus in adults with community-acquired bacterial meningitis. Neurology. 2010;75(10):918-923.
- Wang KW, Chang WN, Chang HW, et al. Clinical relevance of hydrocephalus in bacterial meningitis in adults. Surg Neurol. 2005;64(1):61-65.
- Jung TY, Chung WK, Oh IJ. The prognostic significance of surgically treated hydrocephalus in leptomeningeal metastases. Clin Neurol Neurosurg. 2014;119:80-83.
- Volkov AA, Filis AK, Vrionis FD. Surgical treatment for leptomeningeal disease. Cancer Control. 2017;24(1):47-53.
- Chamberlain MC. Neoplastic meningitis. Oncologist. 2008;13(9):967-977.
- Rekate HL. The definition and classification of hydrocephalus: a personal recommendation to stimulate debate. Cerebrospinal Fluid Res. 2008;5:2.
- Owler BK, Jacobson EE, Johnston IH. Low pressure hydrocephalus: issues of diagnosis and treatment in five cases. Br J Neurosurg. 2001;15(4):353-359.
- Lesniak MS, Clatterbuck RE, Rigamonti D, et al. Low pressure hydrocephalus and ventriculomegaly: hysteresis, non-linear dynamics, and the benefits of CSF diversion. Br J Neurosurg. 2002;16(6):555-561.
- Hamilton MG, Price AV. Syndrome of inappropriately low-pressure acute hydrocephalus (SILPAH). Acta Neurochir Suppl. 2012;113:155-159.
- Adams Rd, Fisher Cm, Hakim S, et al. Symptomatic occult hydrocephalus with “normal” cerebrospinal-fluid pressure: A treatable syndrome. N Engl J Med. 1965;273:117-126.
- Miskin N, Patel H, Franceschi AM, et al. Diagnosis of normal-pressure hydrocephalus: Use of traditional measures in the era of volumetric MR imaging. Radiology. 2017;285(1):197-205.
- Borgesen SE, Gerris F. The predictive value of conductance to outflow of CSF in normal pressure hydrocephalus. Brain. 1982;105(pt. 1):65–86.
- Oi S, Di Rocco C. Proposal of “evolution theory in cerebrospinal fluid dynamics” and minor pathway hydrocephalus in developing immature brain. Childs Nerv Syst. 2006;22(7):662–669.
- Bradley WG. Magnetic resonance imaging of normal pressure hydrocephalus. Semin Ultrasound CT MR. 2016;37(2):120-128.
- Stephensen H, Tisell M, Wikkelso C. There is no transmantle pressure gradient in either communicating or non-communicating hydrocephalus. Neurosurgery. 2002;50(4):763–773.
- Owler BK, Pickard JD. Normal pressure hydrocephalus and cerebral blood flow: a review. Acta Neurol Scand. 2001;104(6):325-342.
- Bradley WG Jr, Scalzo D, Queralt J, et al. Normal-pressure hydrocephalus: evaluation with cerebrospinal fluid flow measurements at MR imaging. Radiology. 1996;198(2):523–529.
- Dixon GR, Friedman JA, Luetmer PH, et al. Use of cerebrospinal fluid flow rates measured by phase-contrast MR to predict outcome of ventriculoperitoneal shunting for idiopathic normal-pressure hydrocephalus. Mayo Clin Proc. 2002;77(6):509-514.
- Evans WA. An encephalographic ratio for estimating ventricular enlargement and cerebral atrophy. Arch NeurPsych. 1942;47:931–937.
- Ishii K, Kanda T, Harada A, et al. Clinical impact of the callosal angle in the diagnosis of idiopathic normal pressure hydrocephalus. Eur Radiol. 2008;18(11):2678-2683.
- Virhammar J, Laurell K, Cesarini KG, et al. The callosal angle measured on MRI as a predictor of outcome in idiopathic normal-pressure hydrocephalus. J Neurosurg. 2014;120(1):178-184.
- Hashimoto M, Ishikawa M, Mori E, Kuwana N. Diagnosis of idiopathic normal pressure hydrocephalus is supported by MRI-based scheme: a prospective cohort study. Cerebrospinal Fluid Res. 2010;7:18.
- Shinoda N, Hirai O, Hori S, et al. Utility of MRI-based disproportionately enlarged subarachnoid space hydrocephalus scoring for predicting prognosis after surgery for idiopathic normal pressure hydrocephalus: clinical research. J Neurosurg. 2017;3:1-7.
- Miskin N, Patel H, Franceschi AM, et al. Diagnosis of normal-pressure hydrocephalus: use of traditional measures in the era of volumetric MR imaging. Radiology. 2017;285(1):197-205.
Citation
WN G, LN T.Imaging of hydrocephalus. Appl Radiol. 2018; (5):5-13.
May 8, 2018