CT imaging of endoscopy-confirmed gastric pathology
Images












Prior presentation of this article abstract: Rakita D, McElligott SE, Primakov D, Sideridis K, Davidoff S, Friedman B. Gastric Pathology Found during Routine Abdominopelvic CT Imaging with Confirmatory Endoscopic Correlation. Radiological Society of North America (RSNA) Annual Meeting 2006. Educational Exhibit.
Patients experiencing upper gastrointestinal discomfort are routinely referred for abdomino-pelvic computed tomography (CT) imaging.Although dedicated gastric CT imaging utilizing gastric distention is very effective,1 routine examinations usually are not tailored for evaluation of the stomach. Nevertheless, this modality can diagnose a large spectrum of gastric pathology, including inflammatory, neoplastic, and structural abnormalities. Most patients with gastric pathology are then referred for upper endoscopy, which is used for additional characterization, histologic confirmation, and sometimes therapy.
Imaging
At our institution, routine abdomino-pelvic CT scanning in the portal venous phase is performed following administration of positive oral and intravenous contrast. Most patients do not receive effervescent granules for stomach distention. Scanning takes place on a 16- or64-channel helical scanner at 5-mm collimation. Coronal reformatted images are generated routinely.
Gastric neoplasms
Gastric adenocarcinoma
The prevalence of gastric cancer peaks between 50 and 70 years of age. Risk factors include familial adenomatous polyposis, chronic atrophic gastritis, pernicious anemia, history of partial gastrectomy (after 15-20 years), and Ménétrier disease.2 Most gastric cancers are adenocarcinomas of mucous cell origin. Signet-ring cell carcinomas account for up to 15% of all gastric cancers and typically cause scirrhous infiltration of the gastric wall (linitis plastica).1
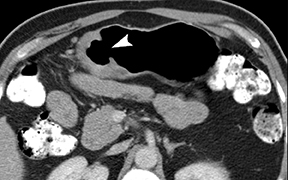
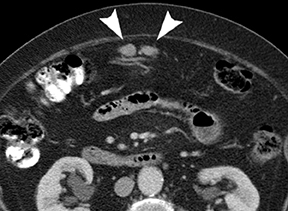
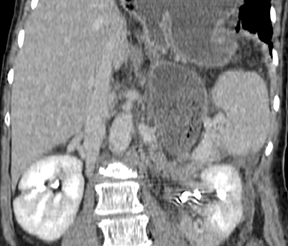
The CT appearance of gastric adenocarcinoma depends on the stage of the lesion. Early cancers appear as focal enhancing mucosal thickening or as polypoid lesions (Figure 1). Advanced cancers demonstrate various degrees of gastric wall thickening and ulceration as well as extension into perigastric fat and adjacent organs (Figure 2).1
Gastric lymphoma
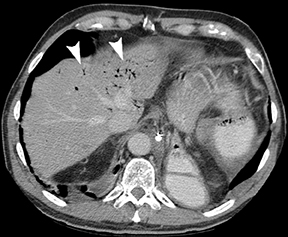
Primary gastric lymphoma is confined to the stomach and regional lymph nodes. These are predominantly non-Hodgkin’s lymphomas of B-cell origin and are more frequently found in the antrum.1,3 Secondary gastric involvement by advanced diffuse lymphoma is also common. It is found in 10% of patients with non-Hodgkin’s lymphomas at diagnosis, and in up to 60% of those with advanced non-Hodgkin’s lymphomas.1,3 The imaging appearances of gastric lymphoma includes focal or diffuse wall thickening, occasionally ulcerated polypoid lesions, and the submucosal nodular form (Figure 3).1,3
Gastric GIST
Gastro-intestinal stromal tumors (GIST) represent a unique group of mesenchymal neoplasms that are distinct from true smooth muscle and neural tumors. GIST is the most common mesenchymal neoplasm of the gastrointestinal tract and is most frequently found in the stomach, representing 2% of all gastric tumors.4 The defining feature of these tumors is the expression of c-KIT (CD117), a tyrosine kinase growth factor receptor. The immunoreactivity for c-KIT distinguishes GISTs from true leiomyomas, leiomyosarcomas, schwannomas, and neurofibromas. Additionally, it is important in targeted treatment of GIST by Gleevec.4
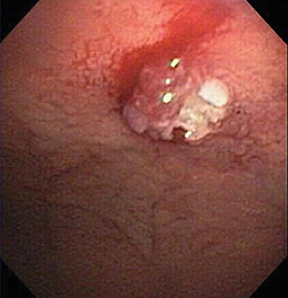
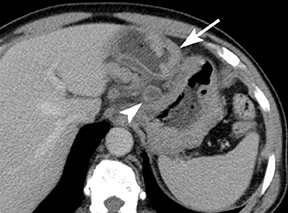
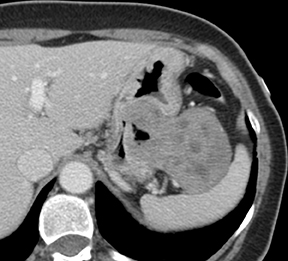
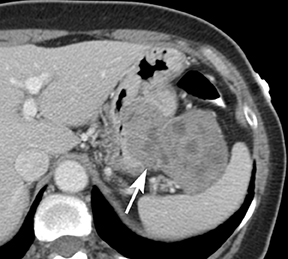
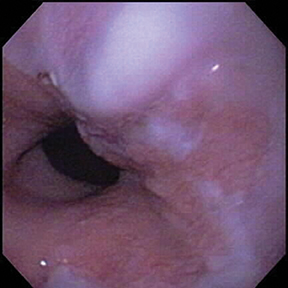
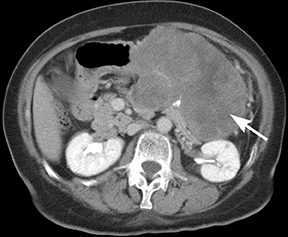
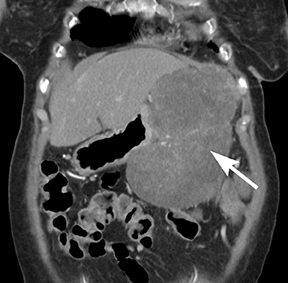
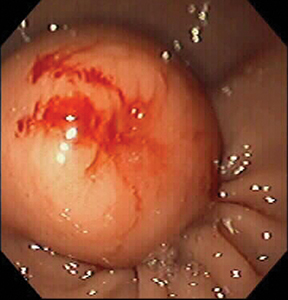
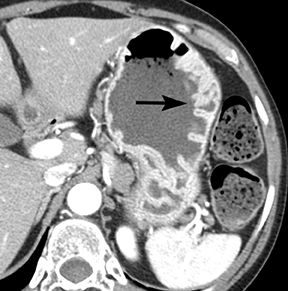
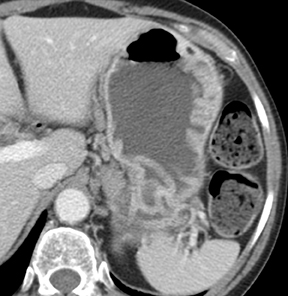
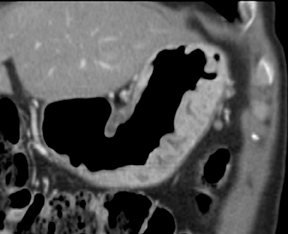
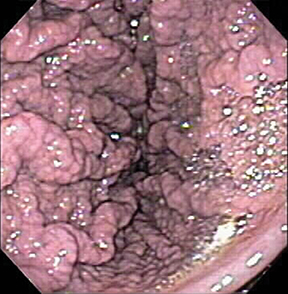
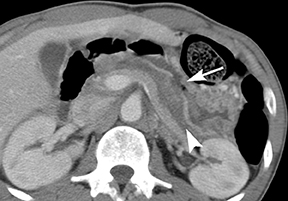
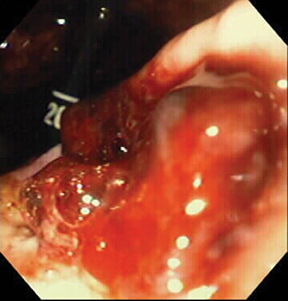
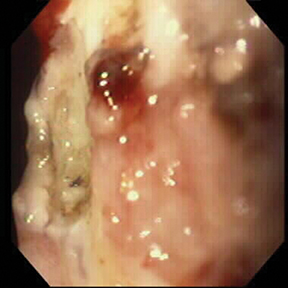
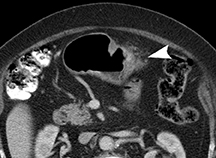
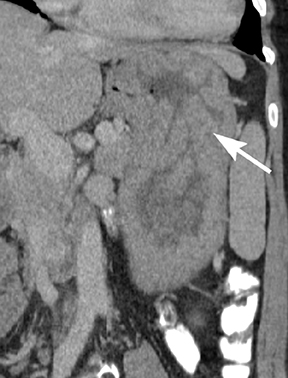
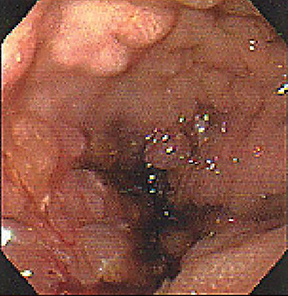
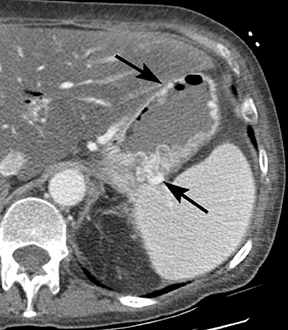
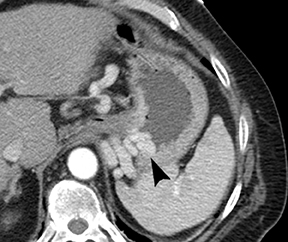
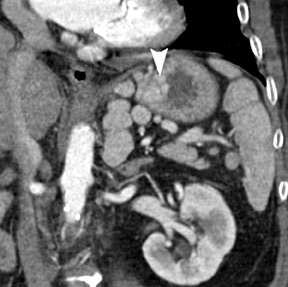
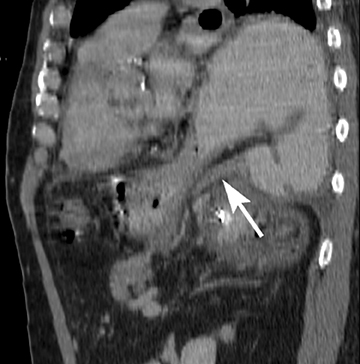
Of GIST tumors, 10% to 30% are malignant. The risk of malignancy is increased in tumors that are located outside the stomach, are greater than 5 cm in diameter, and demonstrate extension into adjacent organs. A characteristic CT imaging feature of gastric GISTis intraluminal and extraluminal ex-tension (Figures 4, 5).4 Endoscopy in GIST patients with a small intraluminal component can be under-whelming (Figure 5).
Gastric lipomas
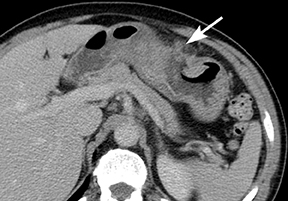
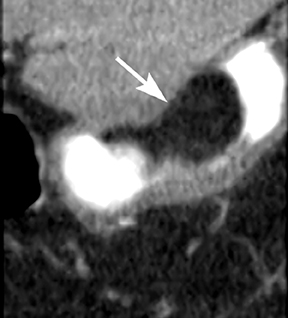
Gastric lipomas are typically found in patients in their fifth or sixth decade of life and 90% are submucosal in location. Hemorrhage, abdominal pain, obstruction, and dyspepsia represent the most common symptoms associated with gastric lipomas, although most by far are asymptomatic and discovered incidentally.5 Lipomas closest to the pylorus can cause obstructive symptoms, frequently by obstructing the pylorus or prolapsing into the duodenum.5 Their CT appearance is that of a submucosal homogeneous mass of fatty attenuation (Figure 6).5
Inflammatory conditions
Gastritis
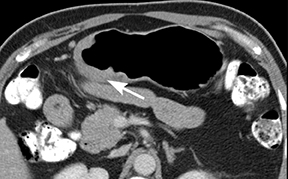
Gastritis is a term covering a broad spectrum of entities that induce inflammatory changes in the gastric mucosa. The common mechanism of injury is an imbalance between the aggressive and the defensive factors that maintain the integrity of the gastric mucosa. Acute gastritis can be broken down into 2 categories: erosive (caused by NSAIDs, alcohol, radiation, ischemia, stress) and nonerosive (generally caused by Helicobacter pylori). H. pylori infection is also the most common cause of chronic, nonerosive gastritis. Colonization of the mucosa by the bacterium leads to chronic inflammation and loss of gastric glands responsible for the production of acid, leading to an atrophic gastritis.6
The most common CT imaging findings of acute gastritis is nonspecific gastric wall thickening. Vivid enhancement of the gastric rugae is commonly noted (Figure 7). Shallow ulcerations and linear erosions of erosive gastritis as well as mucosal nodularity have been on barium radiography and are difficult to visualize on CT.6
Gastric ulcers
Peptic ulcers are mucosal breaks of 3 mm or larger. They are common, occurring in about 10% of adults in western countries. Gastric ulcers account for about 1/3 of peptic ulcers, and duodenal ulcers account for the rest. H. pylori infection and nonsteroidal anti-inflammatory drugs (NSAIDs) are the 2 main factors in the pathogenesis of peptic ulcers. Other factors include steroids, aspirin, smoking, alcohol or coffee consumption, stress, delayed gastric emptying, and duodenogastric bile reflux.7
On a routine CT, gastric ulcers are usually not visualized unless they are penetrating or perforated. The ulcers can appear as mucosal defects and luminal outpouchings, with varying degrees of surrounding wall inflammation. Differentiating between benign and malignant ulcers on CT imaging is not always easy; however, benign ulcers more commonly appear as mucosal defects with smooth, flat borders (Figure8), and malignant ulcers as elevated ulcerated masses (Figure 9),1,7 although there is imaging overlap.
Hyperplastic gastric polyposis
Hyperplastic polyps are the most common benign epithelial tumor in the stomach, constituting as many as 80% to 90% of all gastric polyps. They are non-neoplastic proliferations of surface epithelium, presumably resulting from excessive regenerative hyperplasia in areas of chronic inflammation. Histologically, these lesions consist of hyperplastic foveolar cells that form elongated, cystically dilated glandular structures. Unlike adenomatous polyps, which consist of dysplastic cells that may degenerate to form invasive adenocarcinoma, hyperplastic polyps are composed of nondysplastic cells with virtually no malignant potential.8
Hyperplastic polyposis in the stomach is a frequent finding in familial polyposis and Gardner’s syndrome. Long term use of proton pump inhibitors has also been associated with development of hyperplastic and fundic gland type polyps.8 These polyps are asymptomatic and do not need any treatment.8 (Figure 10).
Gastric varices
Gastric varices, as well as esophageal varices, are typically induced by portal hypertension, which is commonly a consequence of cirrhosis.
Gastric varices can also be seen in cases of isolated splenic vein thrombosis. Gastric varices are classified according to their distribution and whether associated esophageal varices are present. Most gastric varices appear as a continuation of esophageal varices and extend 2 to 5 cm below the gastroesophageal junction, along the lesser curvature of the stomach.9
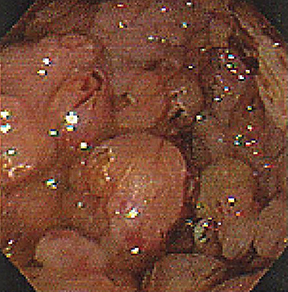
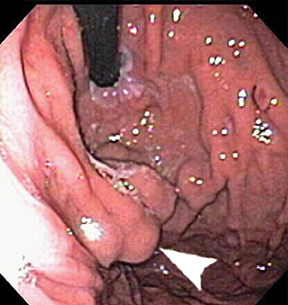
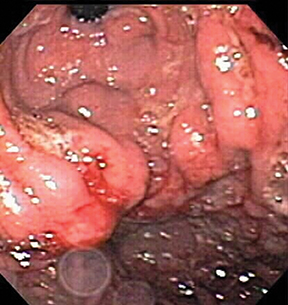
Submucosal, intramural, and perigastric serpentine vascular structures are well demonstrated on CT (Figure 11), best performed in the portal venous phase. Occasionally gastric varices appear as polypoid masses on endoscopy (Figure 12), leading to biopsy.
Structural abnormalities
Gastric volvulus
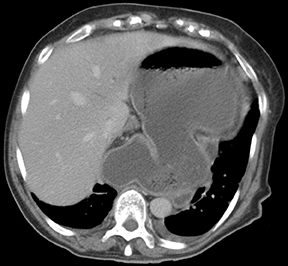
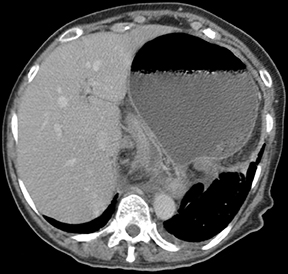
Gastric volvulus is defined as an abnormal rotation of the stomach of more than 180°, creating a closed loop obstruction that can result in incarceration and strangulation. There are 2 forms of gastric volvulus, organoaxial and mesentericoaxial.10 Organoaxial volvulus is the more common and is usually associated with diaphragmatic defects. The stomach rotates around the axis, which connects the gastroesophageal junction and the pylorus, with the greater curvature rotating from an inferior to a superior position. Strangulation and necrosis are common,reported in 5% to 28% of cases.10 (Figures 13 and 14).
The less common form is the mesentericoaxial volvulus. The antrum rotates anteriorly and superiorly, with the axis of rotation bisecting both the lesser and greater curvatures. Rotation is usually incomplete and occurs intermittently; vascular compromise is uncommon. This type occurs more commonly in young children, is associated with ligamentous laxity, and is not associated with diaphragmatic defects.10
Conclusion
Patients experiencing upper gastrointestinal discomfort are routinely referred for abdomino-pelvic CT imaging and endoscopy, which are complimentary in evaluation of gastric pathology. Although dedicated CT imaging of a distended stomach is very effective, routine abdomino-pelvic CT imaging can diagnose a wide range of gastric pathology, including neoplastic, inflammatory, and structural abnormalities. Correlative endoscopic images were provided for most cases.
REFERENCES
- Ba-Salamah A, Prokop M, Uffmann M, et al. Dedicated multidetector CT of the stomach: Spectrum of diseases. Radiographics. 2003;23:625-644.
- Oiso T. Incidence of stomach cancer and its relation to dietary habits and nutrition in Japan between 1900 and 1975. Cancer Res. 1975;35:3254-3258.
- Choi D, Lim HK, Lee SJ, et al. Gastric mucosa-associated lymphoid tissue lymphoma: Helical CT findings and pathologic correlation. AJR Am J Roentgenol. 2002;178:1117-1122.
- Levy AD, Remotti H, Thompson W, et al. From the archives of the AFIP: Gastrointestinal stromal tumors: Radiologic features with pathologic correlation. Radiographics. 2003;23:283-304.
- Thomson WM, Kende AI, Levy AD. Imaging characteristics of gastric lipomas in 16 adult and pediatric patients. AJR Am J Roentgenol. 2003;181:981-85.
- Gelfand DW, Ott DJ, Chen MY. Radiologic evaluation of gastritis and duodenitis. AJR Am J Roentgenol. 1999;173(2): 357-361.
- Levine MS. Peptic ulcers. In: Gore RM, Levine MS, eds. Textbook of Gastrointestinal Radiology. 2nd ed. Philadelphia: WB Saunders, 2000:514-545.
- Choudhry U, Boyce HW, Coppola D. Proton pump inhibitor-associated gastric polyps: A retrospective analysis of their frequency, and endoscopic, histologic, and ultrastructural characteristics. Am J Clin Pathol. 1998;110(5):615-621.
- Afessa B, Kubilis P. Upper gastrointestinal bleeding in patients with cirrhosis: Clinical course and mortality prediction. Am J Gastroenterol. 2000;95:484-489.
- Milne LW, Hunter JJ, Anshus JS, Rosen P. Gastric volvulus: Two cases and a review of the literature. J Emerg Med. 1994; 12:299-306.
Citation
CT imaging of endoscopy-confirmed gastric pathology. Appl Radiol.
November 4, 2013