Benign breast lesions that mimic cancer: Determining radiologic-pathologic concordance
Images
Mammography is the gold standard for early detection of breast cancer with a sensitivity of 60-90% and an overall specificity of approximately 93%, 1 with the average recall rate from screening being 9.8%. Of those recalled, approximately 12% of women necessitate biopsy and more than 60% of biopsies are benign yielding an average 4.8% positive predictive value (PPV).2,3,4 Ultrasound, though an important supplement to mammography and now used to screen women with dense breast tissue,5 has a relatively high false positive rate.6 Magnetic resonance imaging (MRI) is recommended in addition to mammography for women who are at increased lifetime risk of breast cancer of greater than 20-25%. Annual screening with MRI and mammography beginning at age 30 for high-risk women is felt to be effective. 7
Although breast cancer is relatively common and remains the second leading cause of death in women, the majority of findings discovered on imaging which undergo percutaneous biopsy are benign. Furthermore, when there is radiology-pathology discordance following image-guided biopsy, surgical excision is subsequently performed. The additional imaging work-up and in some cases, biopsy or even surgery for these benign lesions is associated with substantial patient anxiety, lost time from work, and added expense to the healthcare system.
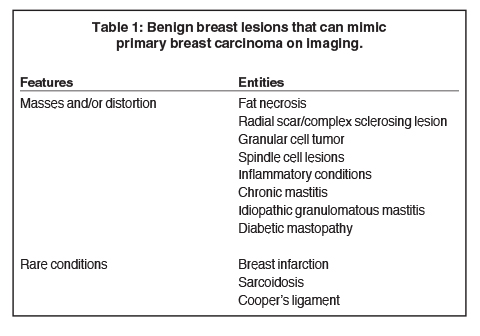
Using case examples, this article will discuss common and uncommon benign conditions that present as masses or architectural distortion on mammography, ultrasound and/or MRI and that can exhibit imaging features that mimic breast cancer (Table 1). As a result, radiologists will become more familiar with benign breast lesions that mimic breast cancer and gain a better understanding concerning their management.
Mammographic features predictive of malignancy include masses with spiculated margins (PPV 81%) and irregular shape (PPV 73%), while masses with round or oval shape, circumscribed margins, and low or fat-containing density are likely to be benign (negative predictive value [NPV] 95%).8,9,10 Sonographic features predictive of malignancy include masses with spiculated margins (PPV 86%), irregular shape (PPV 62%) and non-parallel orientation (PPV 69%), whereas masses with a thin echogenic capsule (NPV 95%), circumscribed margin (NPV 90%), and parallel orientation (NPV 78%) are predictive of benignity.11,12,13 On MRI, a mass with spiculated margins (PPV 88%), rim enhancement (PPV 79%), or washout (Type-III) kinetics (PPV 87%) suggests malignancy, while a lobulated mass with non-enhancing internal septations (NPV 95%) and persistent (Type-I) kinetics (NPV 94%) suggests benignity.14,15
Fat necrosis
Fat necrosis is a nonsuppurative inflammatory process secondary to accidental or surgical trauma (eg, blunt trauma, biopsy, lumpectomy, reduction, augmentation, reconstruction, radiation therapy) and is characterized histologically by foamy histiocytes, lipid-laden macrophages, inflammatory cells with peripheral fibrosis and necrosis. It may present as a painless palpable mass or may be discovered incidentally on imaging.
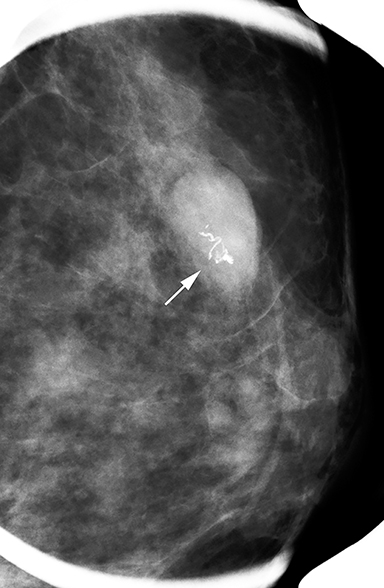
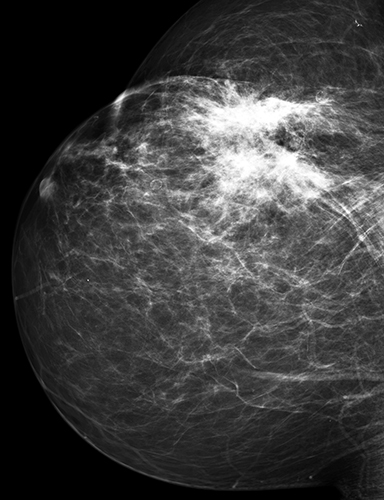
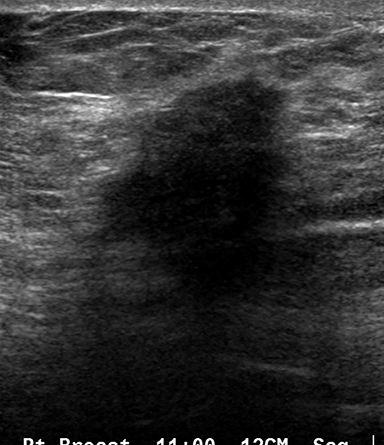
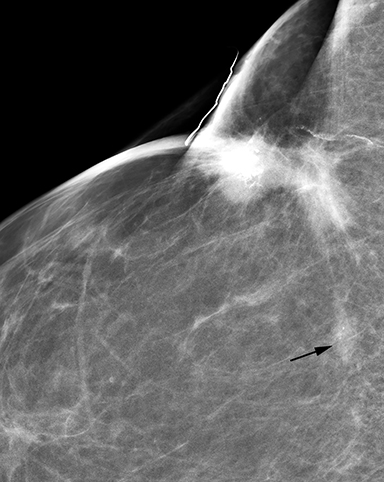
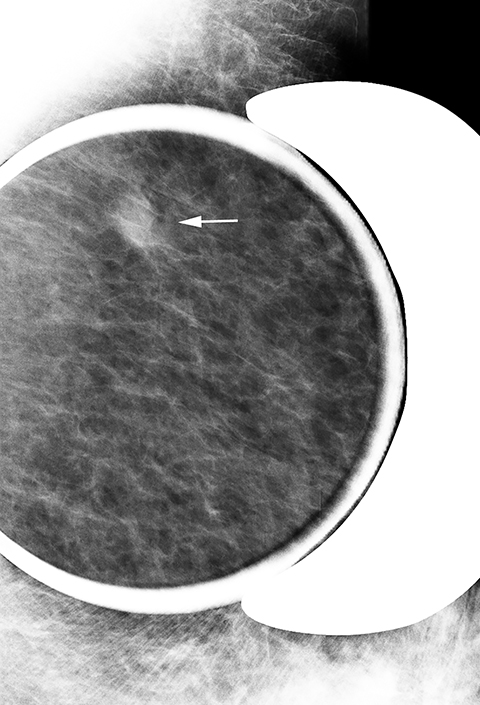
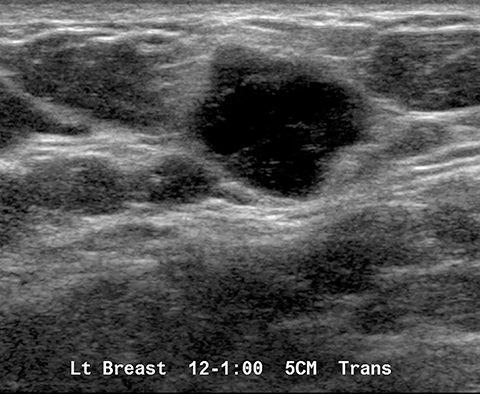
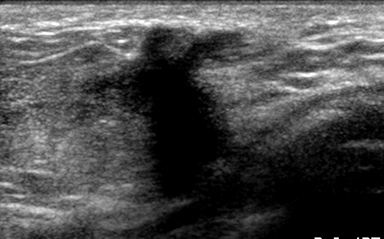
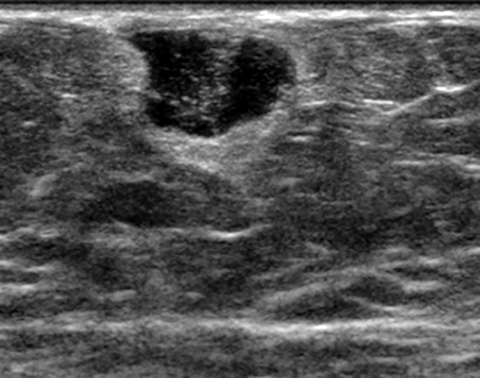
Fat necrosis has variable imaging appearances. The mammographic appearance of fat necrosis ranges from a lucent mass, ill-defined asymmetry, focal mass with microlobulated, ill-defined or spiculated margins, to grouped microcalcifications (Figure 1A,B, D). 16,17 The amount of fibrosis in the lesion determines its mammographic appearance. Sonographically, fat necrosis can present as a cyst, complex cystic or a solid mass with well circumscribed, ill-defined or spiculated margins and architectural distortion of the surrounding tissues (Figure 1c, e).18 On MRI, fat necrosis is characterized by fatty signal intensity mass, often containing a fat-fluid level with variable enhancement following contrast. Other MRI appearances include a solid irregular mass with variable signal intensity on T1- and T2-weighted images, and intense enhancement with Type-II or Type-III kinetics.19 Correlation with clinical history often aids in diagnosis; however, biopsy may be necessary to confirm the diagnosis.
Radial scar/complex sclerosing lesion
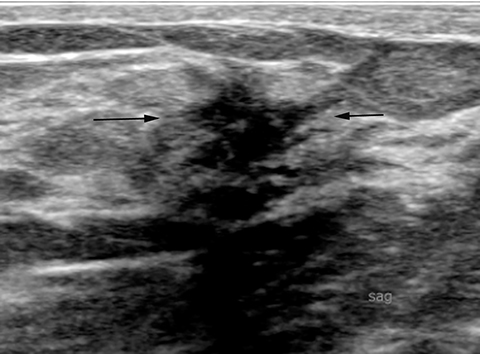
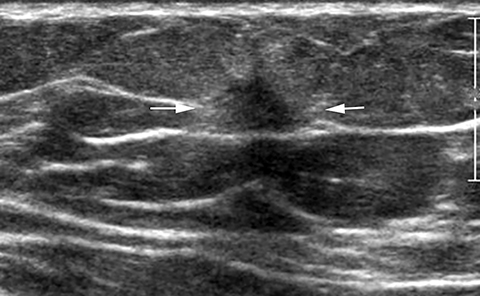
Radial scar (RS) is a pseudo-infiltrative lesion characterized by a fibroelastotic core with entrapped ducts and surrounding radiating ducts and lobules demonstrating a range of epithelial hyperplasia.20 The term “radial scar” is used for lesions < 1cm and the term “complex sclerosing” lesion is used for lesions > 1 cm in size. The epithelial component can display a variety of atypia and may represent a nidus for development of ductal carcinoma in situ. RS are commonly seen incidentally in pathology specimens obtained for other reasons, but can also be seen as non-palpable lesions detected on screening mammography.
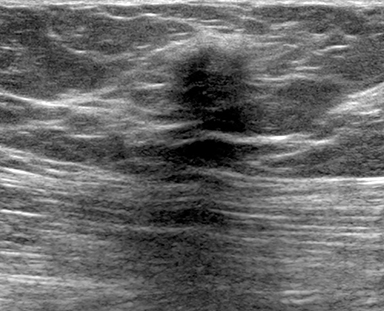
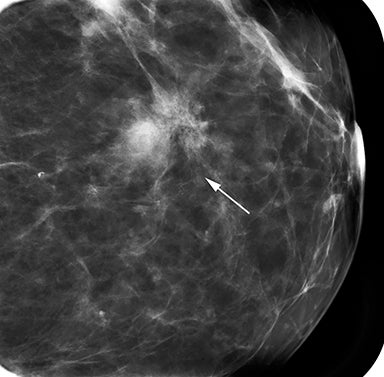
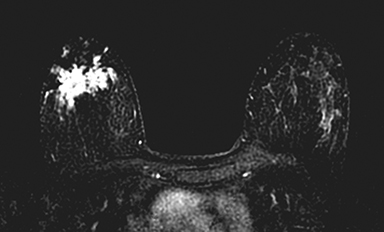
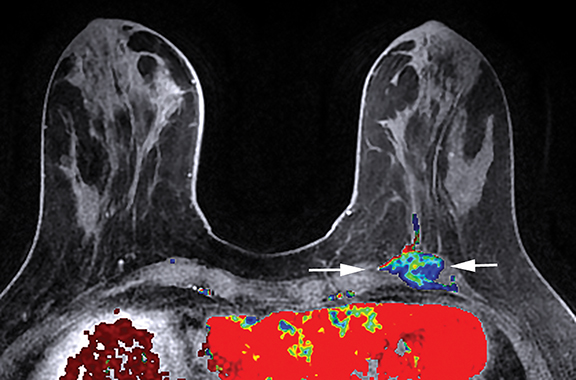
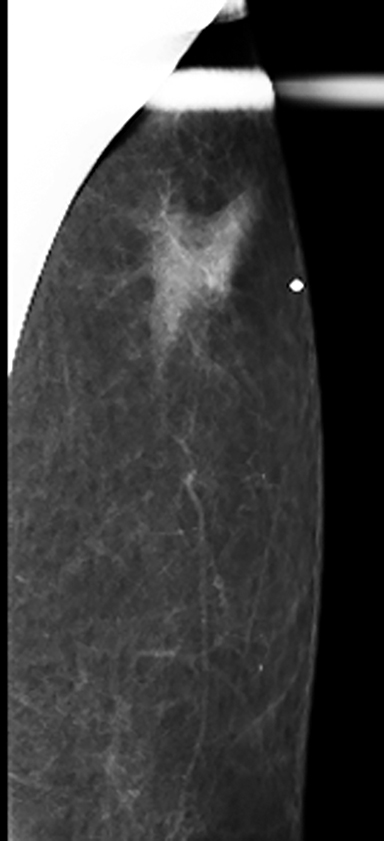
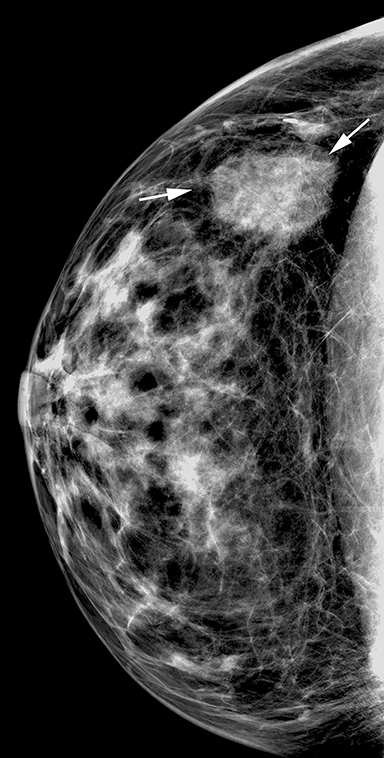
On mammography RS are commonly seen as an area of focal architectural distortion, and are better seen in one projection, without any discernable central mass or overlying skin retraction.21 The lesion has a “black star” appearance with long thin spicules radiating from a central radiolucent area (Figure 2A). Ultrasound may not always detect RS, but can show a poorly defined hypoechoic area, or an irregular hypoechoic mass with ill-defined, spiculated margins and varying degrees of posterior shadowing.22 There are no specific sonographic features that distinguish RS from breast cancer. MRI typically demonstrates a focal low signal area of architectural distortion on T1- and T2-weighted images with enhancement ranging from none/minimal to intense enhancement with, at times Type-III kinetics (Figure 2b). 23 Accumulating evidence indicates an association with atypia and/or malignancy and suggests that it is an independent risk factor for development of carcinoma in either breast. Therefore, excision is recommended following a diagnosis of radial scar on core needle biopsy.20,21
Granular cell tumor
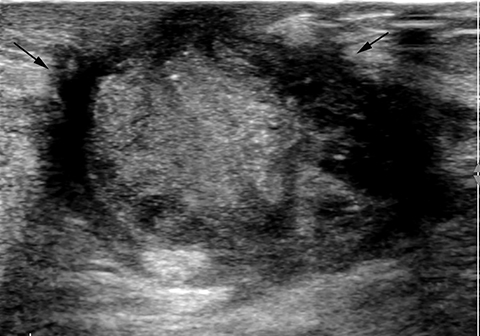
Granular cell tumor of the breast is a rare tumor that arises from Schwann cells. It may occur anywhere in the body with approximately 5-8% of cases seen in the breast. 24 Patients present with a single, unilateral, hard, painless mass mimicking cancer and may have skin retraction and nipple inversion.25 Mammographic features range from a round, circumscribed mass to an indistinct or spiculated mass with or without calcifications (Figure 3a).26 On ultrasound, these present as a hypoechoic poorly defined solid mass with marked posterior shadowing, though it may also present as a well-circumscribed solid mass with variable echogenicity (Figure 3b). A peripheral echogenic halo has also been described.26 MRI appearances are non-specific and include a focal mass with variable high signal on T2-weighted sequence and homogenous or rim enhancement following contrast administration.27 Treatment ranges from imaging follow-up to surgical wide excision.26,27 Local recurrence can occur.
Spindle cell lesions
Spindle cell lesions comprise a wide spectrum of proliferative diseases, some of which are benign. Many spindle cell lesions result from overgrowth of fibroblasts and myofibroblasts, which are a normal part of the lobular mammary stroma. 28,29 Benign proliferations of fibro- and myofibroblasts have a range of clinical characteristics and include pseudoangiomatous stromal hyperplasia (PASH), myofibroblastoma, and fibromatosis. It is difficult to discern between the various types of spindle cell lesions both on imaging and on histopathology from core biopsy. Obtaining sufficient tissue samples during biopsy is critical in making an accurate diagnosis on needle biopsy and obviating the need for subsequent surgical excision.
Fibromatosis
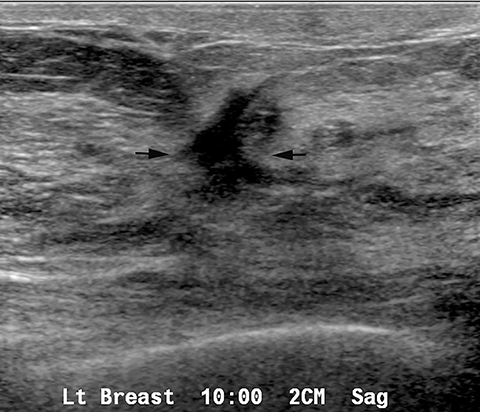
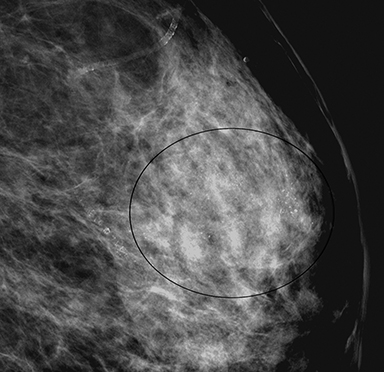
Mammary fibromatosis, also known as desmoid tumor, is a rare, benign tumor of fibroblasts and myofibroblasts.30,31 Clinically, it presents as a firm, painless, mobile mass, although patients can present with focal pain if the tumor involves the chest wall. Additional physical findings may include skin retraction, dimpling, or nipple retraction, arousing suspicion for breast carcinoma.30,31 On mammography, fibromatosis typically appears as a spiculated mass whereas on ultrasound it typically presents as a hypoechoic mass with spiculated, irregular or microlobulated margins30 (Figure 4a). On MRI it usually appears as a mass with low to medium signal intensity on T1-weighted images and variable signal intensity of T2-weighted mages, with benign or Type I kinetics.32 Chest wall involvement is best evaluated by MRI (Figure 4b). Wide surgical excision is preferred, as these tumors tend to be locally aggressive.31
Myofibroblastoma
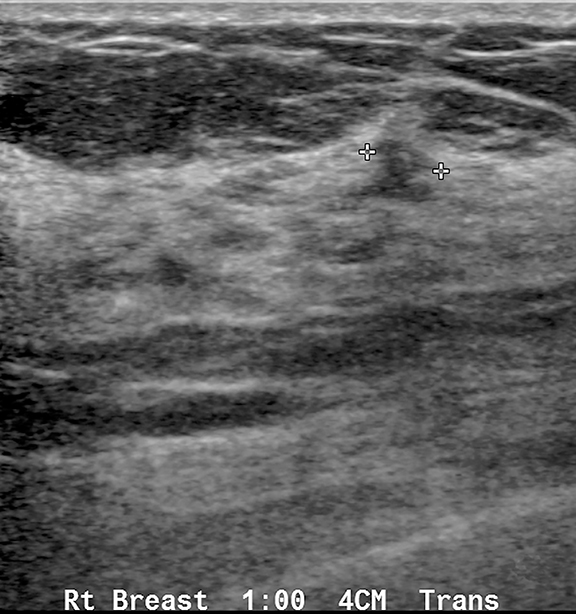
Myofibroblastoma is a benign tumor of myofibroblasts and is more common in men than women.33 Clinically, it presents as a single, slow-growing, painless and mobile mass. On mammography, it appears as a well-defined, round or ovoid dense mass ranging from 1 to 4 cm in diameter (Figure 4c).34 On ultrasound, it typically presents as a solid, circumscribed, homogeneously hypoechoic mass; however, it may also present as an ill-defined mass with posterior shadowing (Figure 4d).35 Most clinicians recommend surgical excision of these lesions as well.
Pseudoangiomatous stromal hyperplasia (PASH)
PASH, a type of spindle cell lesion, is a benign growth of stromal myofibroblasts most commonly found in premenopausal women.36 Patients may present with a painless palpable lump or PASH may be discovered incidentally on imaging. On mammography PASH typically presents as a non-calcified oval mass or as a developing focal asymmetry (Figure 4e).37 On ultrasound it most commonly presents as a well-circumscribed oval hypoechoic mass.36,37 Histologically PASH is characterized by slit-like spaces in the stroma lined by spindle-shaped myofibroblasts. These slit-like spaces are easily mistaken for vascular spaces (hence the name pseudoangiomatous), and may be misinterpreted as angiosarcoma.37 The pathogenesis is poorly understood, however ovarian hormones may play a role. PASH can enlarge, but is benign and does not need to be excised, although some patients opt for surgical removal.
Inflammatory conditions
Mastitis
Mastitis is a focal or diffuse breast infection, seen both in puerperal and non-puerperal states. Patients with acute mastitis typically present with tenderness, redness, swelling and warmth of the breast. Diagnosis is often made on clinical symptoms and response to antibiotics. Chronic mastitis, seen commonly in non-puerperal states, presents clinically with asymmetric breast thickening, breast lump, nipple discharge, and/or axillary lymphadenopathy without any constitutional symptoms.
Mammography may be difficult to perform due to tenderness and breast swelling and can be negative or show focal/diffuse skin thickening or an ill-defined focal asymmetry. 38 Sonography demonstrates skin thickening, increased echogenicity of fat, loss of tissue planes, and/or dilated ducts (over 3mm) or lymphatics.38 A focal heterogeneous hypoechoic mass with thick irregular walls and increased peripheral vascularity, representing an abscess can also frequently be seen (Figure 5a). Aspiration may be nondiagnostic as the abscess contents are thick and contain granulation tissue and cultures are frequently negative for microorganisms. Treatment is percutaneous drainage and antibiotics, although surgical intervention is sometimes needed. MRI can show focal or diffuse skin thickening, thick walled focal mass with variable enhancement and ductal enhancement and lymphadenopathy.39 Imaging findings are non-specific and a core biopsy should be performed to exclude inflammatory carcinoma if there is no clinical improvement on antibiotics.
Granulomatous mastitis
Granulomatous mastitis is a rare condition commonly found in young women and is associated with pregnancy, breast-feeding and oral contraception. 40 Clinically, it often mimics inflammatory cancer and presents as poorly defined areas of thickening and axillary lymphadenopathy. On mammography, it typically presents as an ill-defined focal asymmetry, although it may also present as multiple, small ill-defined masses.40 41 On ultrasound, it typically presents as an ill-defined hypoechoic mass with increased vascularity in close proximity to ducts (Figure 5b). 40 41 MRI typically shows a low signal mass with ill-defined margins, mimicking malignancy.41 Diagnosis is made on core biopsy with histology revealing non-caseating, non-infectious granulomas within the breast lobules. Many patients respond to treatment with corticosteroids and are monitored by close clinical and imaging follow up, though some eventually require surgical wide excision.42 Recurrence is seen in up to 25% of patients.
Diabetic mastopathy
Diabetic mastopathy (DM) is a rare and benign fibroinflammatory condition of the breast. DM is typically found in premenopausal women suffering from chronic type-I diabetes mellitus, but can also be found in non-diabetic patients with other autoimmune conditions.43 DM is characterized clinically and radiologically by dense, fibrous breast tissue that is difficult to distinguish from breast cancer. Clinically, it presents as unilateral or bilateral, single or multiple hard, palpable, irregular, painless masses.43 On mammography it frequently presents as focal asymmetry or mass.44 On ultrasound it typically shows substantial posterior acoustic shadowing, with irregular and diffuse shadowing zones (Figure 5C).44,45 The pathogenesis is poorly understood, however current theories suggest that glycosylation due to hyperglycemia may induce extracellular matrix expansion and an autoimmune response, which leads to further matrix proliferation.43
Other benign conditions
Other benign entities that mimic breast cancer include Cooper’s ligament, sarcoidosis and breast infarct. Cooper’s ligaments can demonstrate shadowing on ultrasound (Figure 6A), but imaging in the orthogonal plane will demonstrate no persistent abnormality, thus confirming a normal anatomic structure.
Sarcoidosis is a benign granulomatous systemic condition of unknown etiology that can secondarily affect the breast in <1% of cases46 in patients with active systemic disease. Most commonly, it presents as bilateral axillary and/or intramammary lymphadenopathy, best appreciated as dense enlarged nodes on mammography or as abnormally enlarged nodes with thickened cortices on ultrasound. In rare cases, sarcoidosis can present as an irregular or spiculated breast mass or as a developing asymmetry on mammography, or as an irregular hypoechoic mass on ultrasound (Figure 6B). On MRI, a suspiciously enhancing irregular mass with progressive to washout kinetics has been reported.47 In almost all cases, biopsy to exclude malignancy is indicated.
Breast infarct is a rare condition that typically presents as a painful palpable lump but also rarely may present as grouped calcifications on mammography (Figure 6C). 48 It is associated with pregnancy and lactation and can be seen in patients taking systemic anticoagulation such as warfarin. Biopsy often is performed to exclude malignancy.
Conclusion
Although breast cancer has characteristic imaging features, it is often difficult to diagnose cancer based on imaging alone and a biopsy is necessary for accurate diagnosis. The high incidence of benign breast conditions that mimic malignancy indicates that careful correlation between the radiologic and pathologic findings is needed. Familiarity with benign breast lesions increases radiologists’ confidence after a core needle biopsy, allays patient fears, and prevents unnecessary surgical excision. Finally, the combination of multimodality imaging and assessment of the patient’s risk factors and clinical presentation will ensure optimal patient care.
References
- Carney PA, Miglioretti DL, Yankaskas BC, et al. Individual and combined effects of age, breast density, and hormone replacement therapy use on the accuracy of screening mam mography. Ann Intern Med. 2003;138:168–175.
- Yankaskas BC, Cleveland RJ, Schell MJ, Kozar R. Association of recall rates with sensitivity and positive predictive values of screening mammography. AJR Am J Roentgenol. 2001; 177:543-549.
- Rosenberg RD, Yankaskas BC, Abraham LA, et al. Performance benchmarks for screening mammography. Radiology. 2006; 241:55-66.
- Sickles EA, Miglioretti DL, Ballard-Barbash R, et al. Performance benchmarks for diagnostic mammography. Radiology. 2005;235:775-790.
- Hooley RJ, Greenberg KL, Stackhouse RM, et al. Screening US in patients with mammographically dense breasts: initial experience with Connecticut Public Act 09-41. Radiology. 2012;265:59-69.
- Corsetti V, Houssami N, Ferrari A, et al. Breast screening with ultrasound in women with mammography-negative dense breasts: evidence on incremental cancer detection and false positives, and associated cost. Eur J Cancer. 2008;44:539-544.
- Lowry KP, Lee JM, Kong CY, et al. Annual screening strategies in BRCA1 and BRCA2 gene mutation carriers: a comparative effectiveness analysis. Cancer. 2012;15;118:2021-2030.
- D’Orsi CJ, Sickles EA, Mendelson EB, Morris EA, et al. ACR BI-RADS ® Atlas, Breast Imaging Reporting and Data System. Reston, VA, American College of Radiology; 2013.
- Liberman L, Abramson AF, Squires FB, et al. The breast imaging reporting and data system: positive predictive value of mammographic features and final assessment categories. AJR Am J Roentgenol. 1998;171:35-40.
- D’Orsi CJ, Kopans DB. Mammographic feature analysis. Semin Roentgenol. 1993;28:204–230.
- Lazarus E, Mainiero MB, Schepps B, et al. BI-RADS lexicon for US and mammography: interobserver variability and positive predictive value. Radiology. 2006; 239:385-391.
- Hong AS, Rosen EL, Soo MS, Baker JA. BI-RADS for sonography: positive and negative predictive values of sonographic features. AJR Am J Roentgenol. 2005;184:1260–1265.
- Stavros T, Thickman D, Rapp CL, et al. Solid breast nodules: use of sonography to distinguish between benign and malignant lesions. Radiology. 1995;196:123–134.
- Ikeda DM, Hylton NM, Kinkel K, et al. Development, standardization, and testing of a lexicon for reporting contrast- enhanced breast magnetic resonance imaging studies. J Magn Reson Imaging. 2001;13:889-895.
- Liberman L, Morris EA, Lee, MJ, et al. Breast lesions detected on MR imaging: Features and positive predictive value. AJR Am J Roentgenol. 2002;179:171-178.
- Hogge JP, Robinson RE, Magnant CM, et al. The mammographic spectrum of fat necrosis of the breast. RadioGraphics. 1995;15:1347-1356.
- Taboada JL, Stephens TW, Krishnamurthy S, et al. The many faces of fat necrosis in the breast. AJR Am J Roentgenol. 2009;192:815–825.
- Soo MS, Kornguth PJ, Hertzberg BS. Fat necrosis in the breast: sonographic features. Radiology. 1998;206:261–269.
- Daly CP, Jaeger B, Sill DS. Variable appearances of fat necrosis on breast MRI. AJR Am J Roentgenol. 2008;191:1374-1380.
- Kennedy M, Masterson AV, Kerin M, Flanagan F. Pathology and clinical relevance of radial scars: a review. J clin pathol. 2003;56:721–724.
- Shaheen R, Schimmelpenninck CA, Stoddart L, et al. Spectrum of diseases presenting as architectural distortion on mammography: multimodality radiologic imaging with pathologic correlation. Semin ultrasound CT MR. 2011;3:351–362.
- Cohen MA, Sferlazza SJ. Role of sonography in evaluation of radial scars of the breast. AJR Am J Roentgenol. 2000;174:1075-1078.
- Pediconi F, Occhiato R, Venditti F, et al. Radial scars of the breast: contrast enhanced magnetic resonance mammography appearance. Breast J. 2005;11:23-28.
- Adeniran A, Al-Ahmadie H, Mahoney MC, Robinson-Smith TM. Granular cell tumor of the breast: a series of 17 cases and review of the literature. Breast J. 2004;10:528–531.
- Lack EE, Worsham GF, Callihan MD, et al. Granular cell tumor: a clinicopathologic study of 110 patients. J Surg Oncol. 1980;13:301–316.
- Yang WT, Edeiken-Monroe B, Sneige N, Fornage BD. Sonographic and mammographic appearances of granular cell tumors of the breast with pathological correlation. J Clin Ultrasound. 2006;34:153–160.
- Scaranelo AM, Bukhanov K, Crystal P, et al. Granular cell tumour of the breast: MRI findings and review of the literature. Br J Radiol. 2007;80:970–974.
- Brogi E. Benign and malignant spindle cell lesions of the breast. Semin Diagn Pathol. 2004;21: 57–64.
- Schnitt SJ. Spindle cell lesions of the breast. Surg Path Clin. 2009;2:375-390.
- Erguvan-Dogan B, Dempsey PJ, Ayyar G, Gilcrease MZ. Primary desmoid tumor (extraabdominal fibromatosis) of the breast. AJR Am J Roentgenol. 2005;185:488–489.
- Schwarz GS, Drotman M, Rosenblatt R, Milner L, et al. Fibromatosis of the breast: case report and current concepts in the management of an uncommon lesion. Breast J. 2006;12:66–71.
- Nakazono T, Satoh T, Hamamoto T, Kudo S. Dynamic MRI of fibromatosis of the breast. AJR Am J Roentgenol. 2003;181:1718–1719.
- Mele M, Jensen V, Worenecki A, Lelkaitis G. Myofibroblastoma of the breast: a case report and literature review. Int J Surg Case Rep. 20112:93-6.
- Dockery WD, Singh HR, Wilentz RE. Myofibroblastoma of the male breast: imaging appearance and ultrasound-guided core biopsy diagnosis. Breast J. 2001 May-Jun;7:192-4.
- Yoo EY, Shin JH, Ko EY, et al. Myofibroblastoma of the female breast mammographic, sonographic, and magnetic resonance imaging findings. J Ultrasound Med. 2010;29:1833-1836.
- Celliers L, Wong D, Bourke A. Pseudoangiomatous stromal hyperplasia: a study of the mammographic and sonographic features. Clin Radiol. 2010;65:145–149.
- Hargaden G. Analysis of the mammographic and sonographic features of pseudoangiomatous stromal hyperplasia. AJR Am J Roentgenol. 2008;191:3593-63.
- Ferron S, Asad-Syed M, Boisserie-Lacroix M, et al. Imaging benign inflammatory syndromes. Diagn Interv Imaging. 2012;93:85–94.
- Rieber A, Tomczak RJ, Mergo PJ, et al. MRI of the breast in differential diagnosis of mastitis versus inflammatory carcinoma and follow up. J Comput Assist Tomogr. 1997;21:128-132.
- Hovanessian Larsen LJ, Peyvandi B, Klipfel N, et al. Granulomatous lobular mastitis: imaging, diagnosis, and treatment. AJR Am J Roentgenol. 2009;193:574–581.
- Dursun M, Yilmaz S, Yahyayev A, et al. Multimodality imaging features of idiopathic granulomatous mastitis: outcome of 12 years of experience. Radiol Med. 2012 Jun;117:529-38. doi: 10.1007/s11547-011-0733-2. Epub 2011 Oct 21.
- Taghizadeh R, Shelley OP, Chew BK, Weiler-Mithoff EM. Idiopathic granulomatous mastitis: surgery, treatment, and reconstruction. Breast J. 2007;13:509–513.
- Chan CL, Ho RS, Shek TW, Kwong A. Diabetic mastopathy. Breast J. 2013;19:533–538.
- Neetu G, Pathmanathan R, Weng NK. Diabetic mastopathy: A Case report and literature review. Case Rep Oncol. 2010;3:245–251.
- Francisco C, Júlio C, Fontes AL, et al. Diabetic mastopathy: A case report. Clin Imaging. 2012;36:829–32.
- Isley LM, Cluver AR, Leddy RJ, Baker MK. Primary sarcoid of the breast with incidental malignancy. J Clin Imaging Sci. 2012;2:46.
- Kenzel PP, Hadijuana J, Hosten N, et al. Sarcoidosis of the breast: Mammographic, ultrasound, and MR findings. J Comput Assist Tomogr. 1997;21:439–441.
- Lucey J J. Spontaneous infarction of the breast. J Clin Pathol. 1975; 28: 937–943.
Citation
Mario J, Venkataraman ;S, Dialani ;V, Slanetz ;APJ. Benign breast lesions that mimic cancer: Determining radiologic-pathologic concordance. Appl Radiol. 2015;(9):28-32.
September 2, 2015