Study Indicates Oral MR Imaging Agent Well Tolerated with Food
 The last patient visit has been completed in the study to evaluate the effect of food intake on the absorption of Orviglance (formerly called Mangoral). This Food Effect Study is part of the ongoing pivotal clinical program for Orviglance and will be included in the marketing authorization package to the health authorities, including US Food and Drug Administration (FDA) and European Medicines Agency (EMA).
The last patient visit has been completed in the study to evaluate the effect of food intake on the absorption of Orviglance (formerly called Mangoral). This Food Effect Study is part of the ongoing pivotal clinical program for Orviglance and will be included in the marketing authorization package to the health authorities, including US Food and Drug Administration (FDA) and European Medicines Agency (EMA).
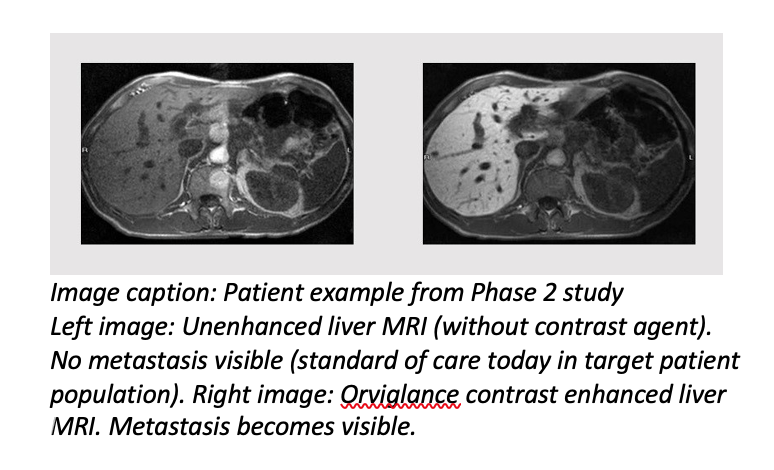
Orviglance (manganese chloride tetrahydrate) is Ascelia Pharma’s oral investigational MR imaging agent developed to improve the detection and visualization of focal liver lesions, including liver metastases and primary tumors, in patients with reduced kidney function. These patients are at risk of serious side effects from the currently available class of gadolinium-based contrast agents. Orviglance, which has been granted an Orphan Drug Designation by the FDA, is currently in Phase 3 development, including the global multi-center SPARKLE study.
The Food Effect Study is designed to assess whether the current requirement to fast before patients are given Orviglance is strictly necessary. A potential removal or adjustment of the fasting requirement will simplify and provide convenience for patients using Orviglance. More specifically, the objective of the Food Effect study is to evaluate the effect of food intake on pharmacokinetics, pharmacodynamics and safety of Orviglance. In this crossover study, Orviglance was administered in 24 healthy volunteers in fasting condition versus two conditions with food intake (snack or full meal).
Preliminary data indicate that Orviglance has been well tolerated in the study. Final results of the Food Effect Study are expected within four months.
“We are pleased to have completed the patient enrollment amid the Covid-19 pandemic. A potential removal of the current fasting requirement could further improve the convenience and ease the administration of Orviglance in clinical practice,” said Carl Bjartmar, Chief Medical Officer of Ascelia Pharma.