Plasmablastic Lymphoma
Images
Case Summary
An adult presented with left shoulder pain and difficulty raising their arm after a fall three days previously. The medical history was notable for type II diabetes mellitus, hypertension, and hyperlipidemia. A physical examination showed weakness in abduction and external rotation of the left shoulder, with no evidence of neurovascular deficit. Complete blood count and chemistry panel were unremarkable. Laboratory tests were positive for Epstein-Barr virus (EBV) and negative for human immunodeficiency virus (HIV).
Imaging Findings
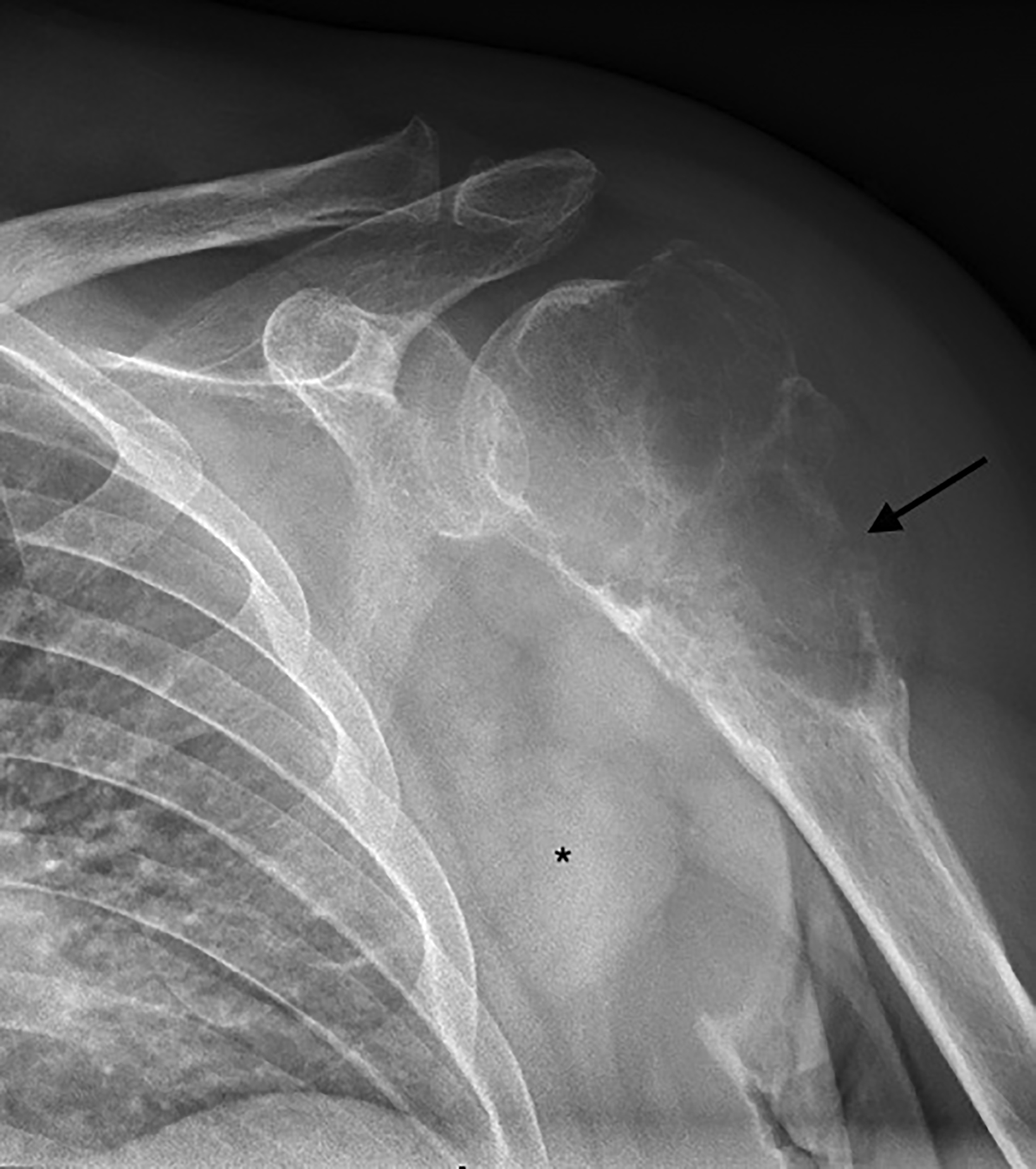
Radiography of the left shoulder revealed a lytic, expansile, eccentrically located lesion with a narrow zone of transition within the humoral head and extending to the subchondral bone without sclerotic margins (Figure 1). There was loss of cortex on the lateral aspect of the lesion, concerning for pathologic fracture. Based on the clinical history and appearance of the lesion, it was first thought to be a giant cell tumor (GCT).
The differential diagnosis included aneurysmal bone cyst, which was considered less likely given the patient’s age and subchondral extension of the lesion. The concern for a soft-tissue component or other aggressive process led to further imaging and biopsy.
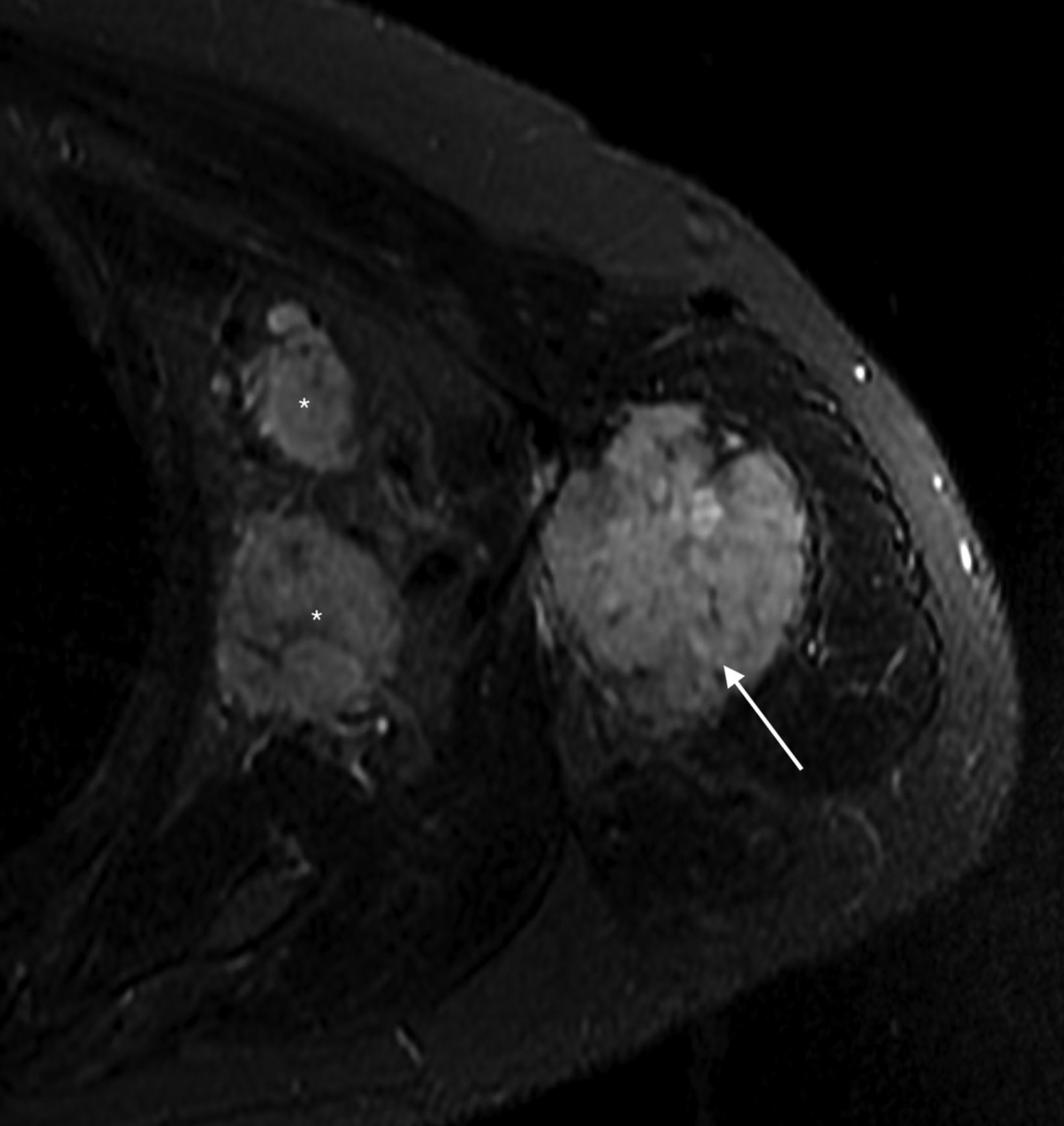
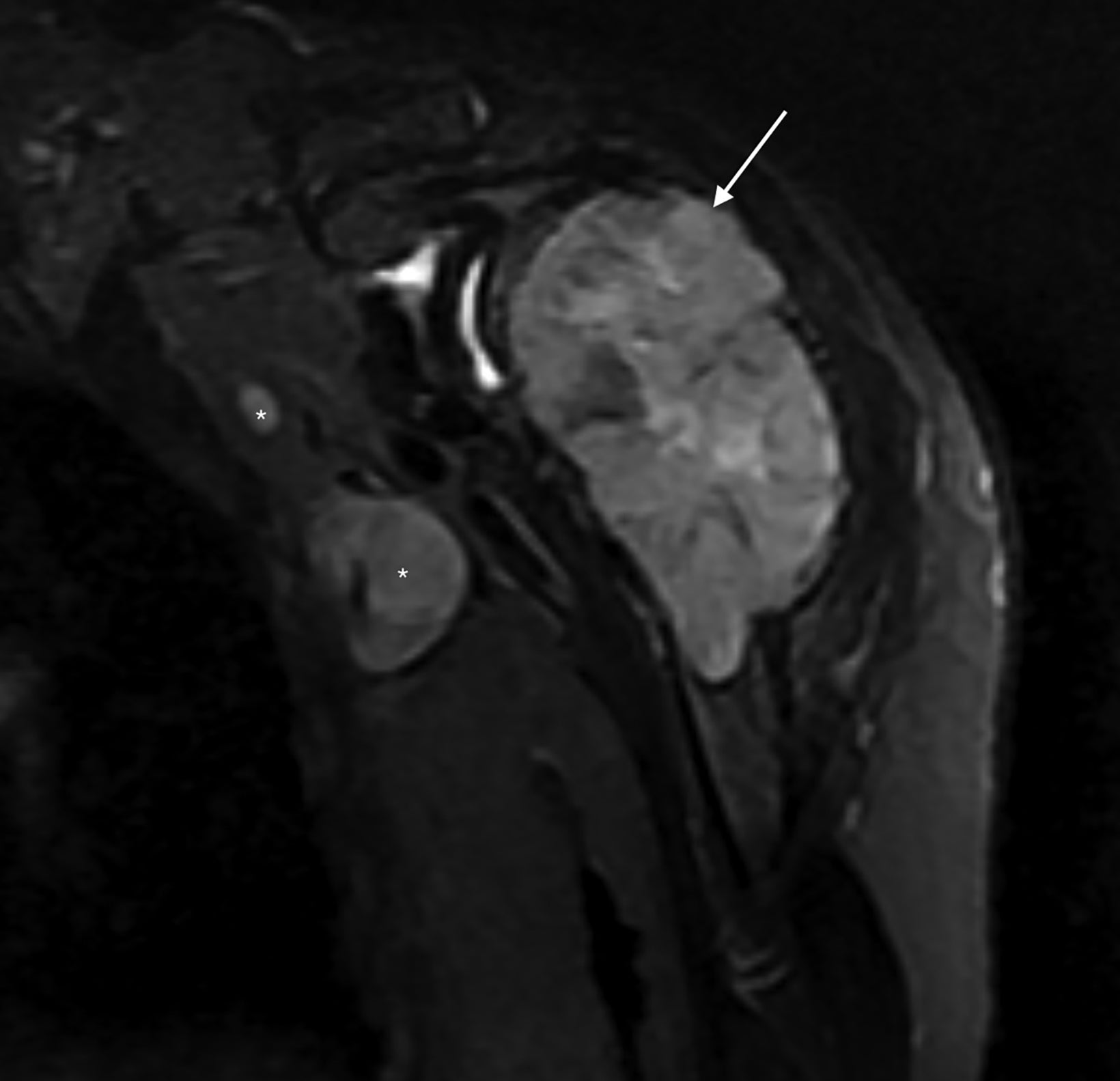
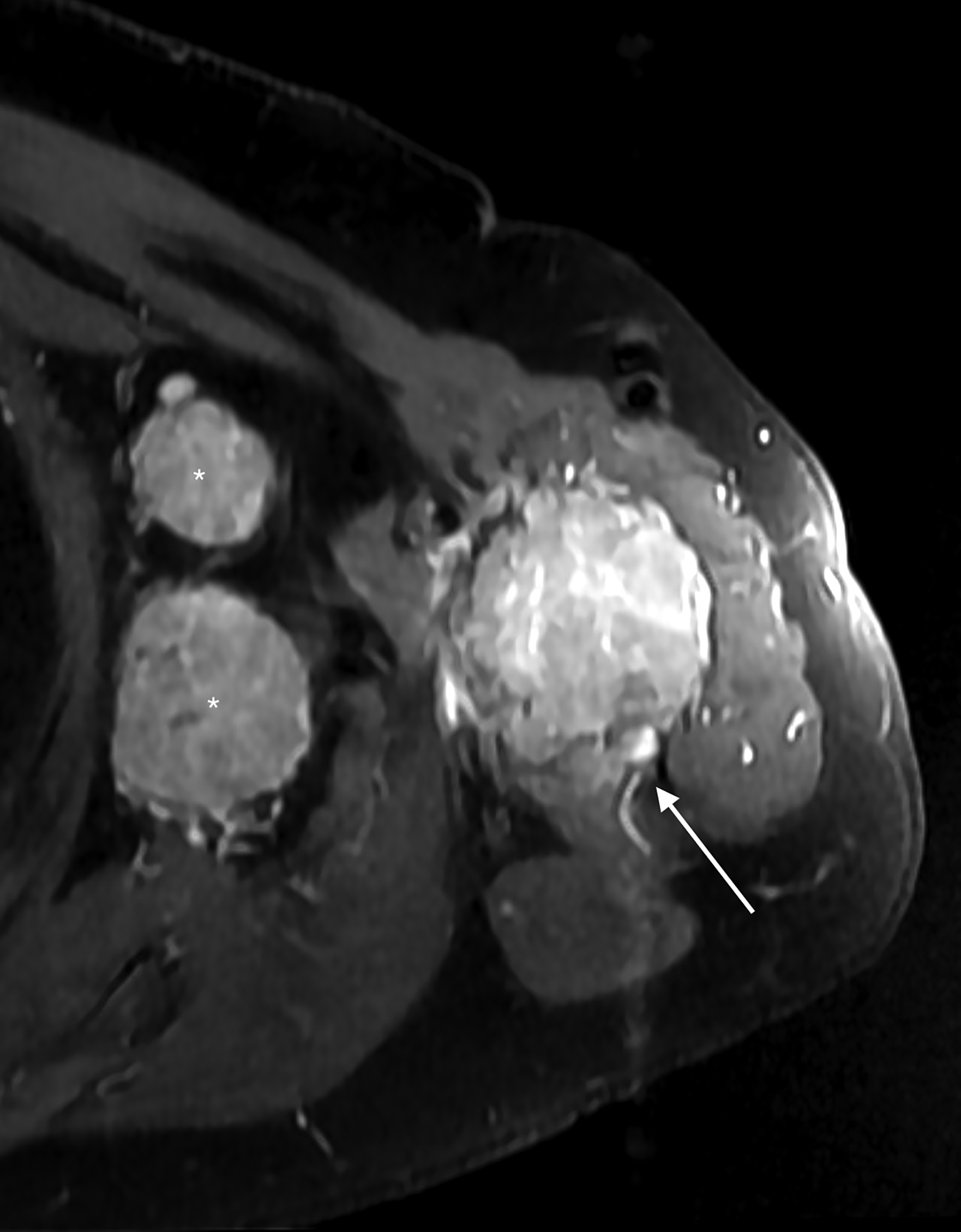
Subsequent pre- and post-Gadolinium contrast magnetic resonance imaging showed the lesion to be 5.6 cm × 5.5 cm × 9.8 cm with low signal on T1 sequences, heterogeneous
hyperintensity with areas of low signal intensity on fluid sensitive sequences, including short tau inversion recovery (STIR), and mild postcontrast enhancement (Figure 2).
There was cortical erosion with extension of the lesion into the surrounding soft tissues. Additionally, there were two pathologically enlarged axillary lymph nodes (2.4 and 3.3 cm in short axes). A review of the initial radiograph showed these lymph nodes to be present in retrospect (Figure 1).
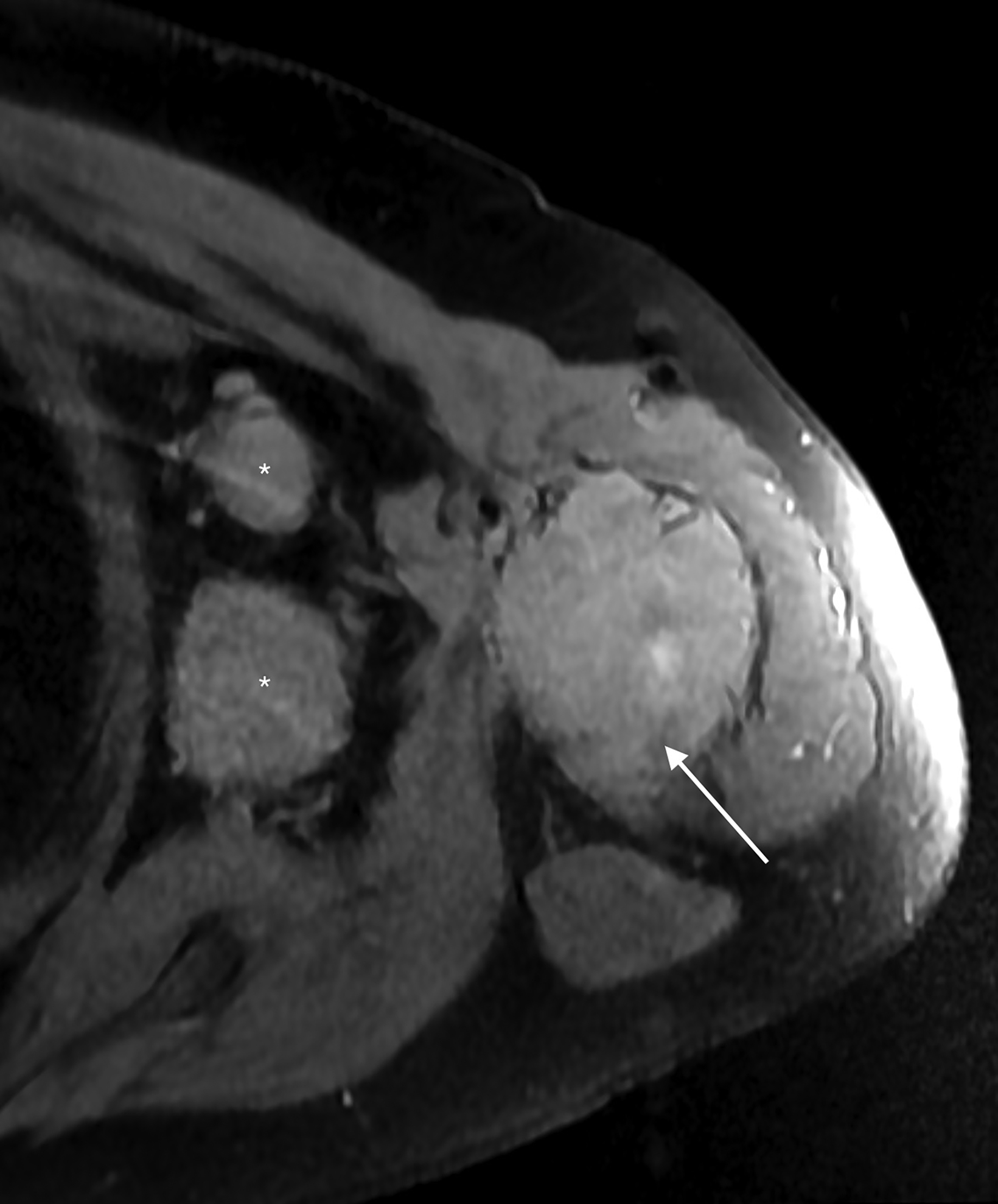
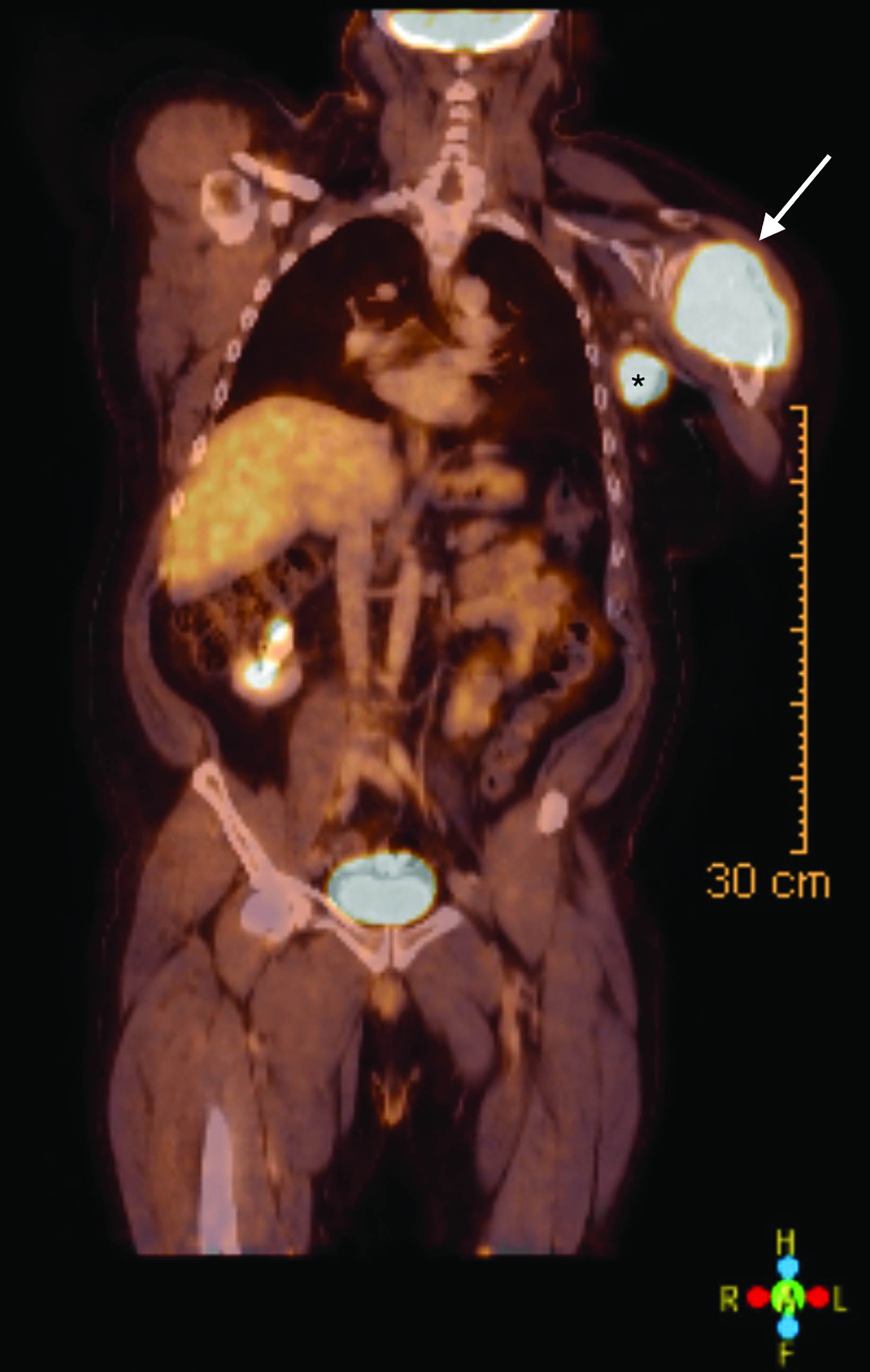
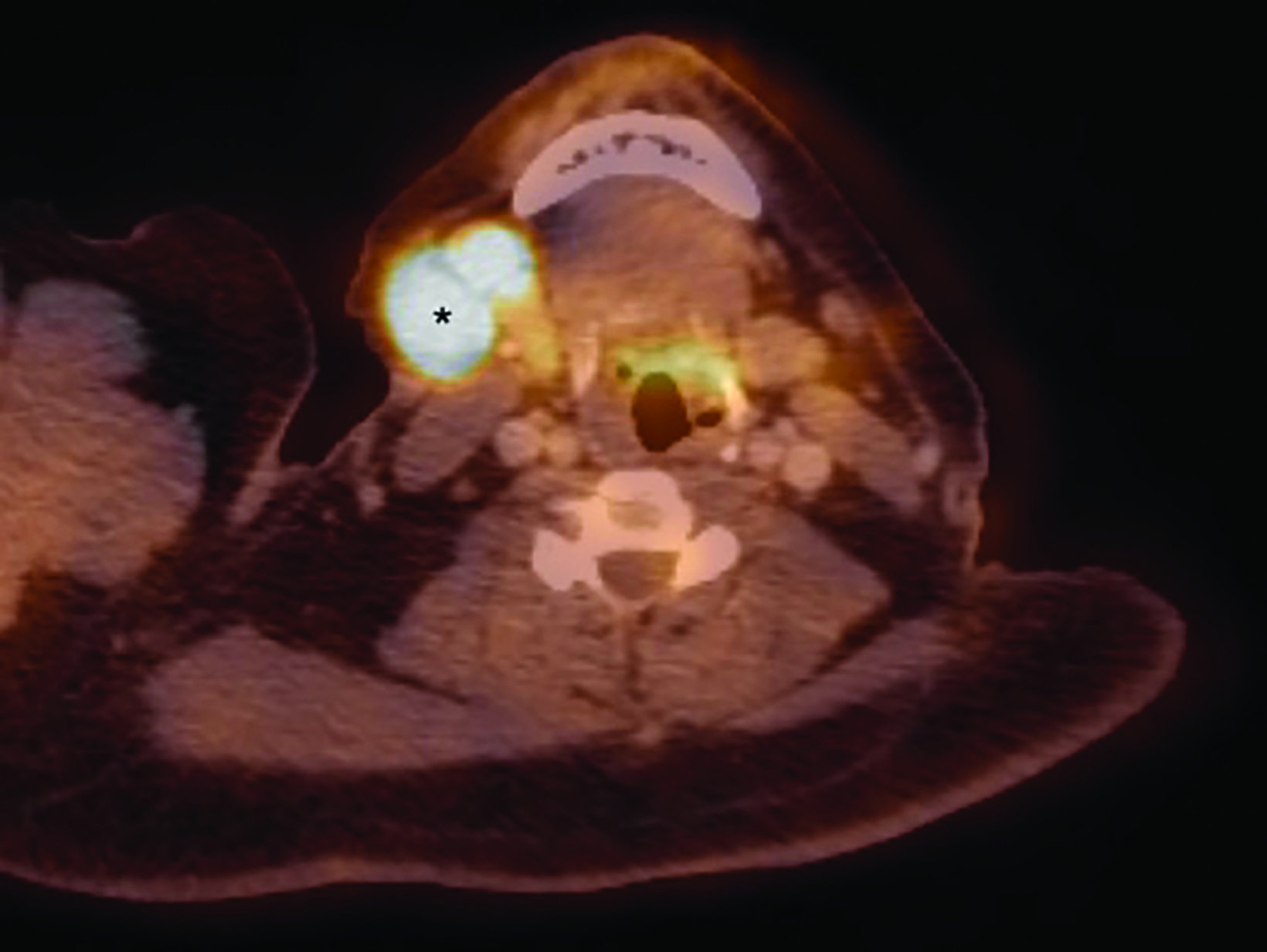
Staging (18)F-fluorodeoxyglucose (FDG) positron emission tomography/computed tomography (PET/CT) showed hypermetabolism of the humoral lesion with a maximum standardized uptake value (SUV) of 11.7 and of the axillary lymph nodes with maximum SUV of 11.6 (Figure 3). There were also hypermetabolic right cervical (maximum SUV 14.3), left subpectoral (maximum SUV 3.7), and right axillary lymph nodes (max SUV 4.4) concerning for additional sites of disease.
Diagnosis
Plasmablastic lymphoma
Discussion
Plasmablastic lymphoma (PBL) is a rare, highly aggressive, diffuse large B-cell lymphoma variant characterized by proliferation of CD20-neoplastic cells with immunophenotype resembling plasmablasts, a plasma cell precursor. Initially described as an oral cavity
lesion in HIV-positive patients,1 PBL is now known to occur in other anatomic locations, including the gastrointestinal tract, skin, and, less commonly, the bones,2-4 as well as in non-HIV-associated immunosuppressed and even immunocompetent patients.4
Extra-oral involvement appears to be more common in HIV-negative patients, while oral involvement remains the predominant manifestation in HIV-positive patients.2 Additionally, Epstein-Barr virus infection is present in a high percentage of cases.3
Osseous PBL has varied and nonspecific imaging findings, with case reports showing overlap with osteosarcoma and plasmacytoma, resulting in a significant risk of misdiagnosis.4,5 Differentiation occurs on the basis of histopathology and immunohistochemistry.4
Our case showed imaging findings mimicking those of giant cell tumor6 and, to a lesser extent, aneurysmal bone cyst,7 on initial plain films. Subsequent cross-sectional imaging findings of lymphadenopathy pointed toward a more malignant etiology, which was confirmed by tissue biopsy.
Owing to the rarity of this disease, there are currently no consensus treatment guidelines.5 A number of chemotherapy regimens have been tried, with mixed results.5 The median overall survival is 6-19 months.5 Given PBL’s poor prognosis, it is important to differentiate this disease from benign entities.
Conclusion
Plasmablastic lymphoma has imaging features overlapping with other benign and malignant entities, including giant cell tumors and aneurysmal bone cysts. In a patient with a presumed benign lytic lesion such as giant cell tumor, findings of regional lymphadenopathy should prompt suspicion for an alternative malignant etiology, including plasmablastic lymphoma.
References
- Delecluse HJ, Anagnostopoulos I, Dallenbach F, et al. Plasmablastic lymphomas of the oral cavity: a new entity associated with the human immunodeficiency virus infection. Blood. 1997;89(4):1413-20.
- Hansra D, Montague N, Stefanovic A, et al. Oral and extraoral plasmablastic lymphoma: similarities and differences in clinicopathologic characteristics. Am J Clin Pathol. 2010;134:710-719.
- Lopez A, Abrisqueta P. Plasmablastic lymphoma: current perspectives. Blood Lymphat Cancer. 2018;8:63-70.
- Sarker AK, Im HJ, Paeng JC, et al. Plasmablastic lymphoma exclusively involving bones mimicking osteosarcoma in an immunocompetent patient: A case report. Medicine (Baltimore). 2016;95(28):e4241.
- Diaz R, Amalaseelan J, Imlay-gillespie L. Plasmablastic lymphoma masquerading solitary plasmacytoma in an immunocompetent patient. BMJ Case Rep. 2018;2018:1-4.
- Chakarun CJ, Forrester DM, Gottsegen CJ, Patel DB, White EA, Matcuk GR. Giant cell tumor of bone: review, mimics, and new developments in treatment. Radiographics. 2013;33(1):197-211.
- Kransdorf MJ, Sweet DE. Aneurysmal bone cyst: concept, controversy, clinical presentation, and imaging. AJR Am J Roentgenol. 1995;164(3):573-80.
References
Citation
X B, R V,. Plasmablastic Lymphoma. Appl Radiol. 2022;(4):45-48.
July 13, 2022