Non-Tuberculous Mycobacterial Otomastoiditis with Intracranial Involvement
Images
Brought to you by Guerbet.
Case Summary
An 11-year-old presented with right ear pain, intermittent fevers, and new right-sided jaw pain with dizziness. Medical history included recurrent otitis media with otorrhea and bilateral ventilation tube placement. During the previous month, the patient reported a right ear infection that did not respond to medical management. On examination, the right external auditory canal demonstrated marked posterior wall swelling and edema, modest otorrhea, and tympanic membrane effusion. A contrast-enhanced CT scan of temporal bones was ordered.
Imaging Findings and Subsequent Course
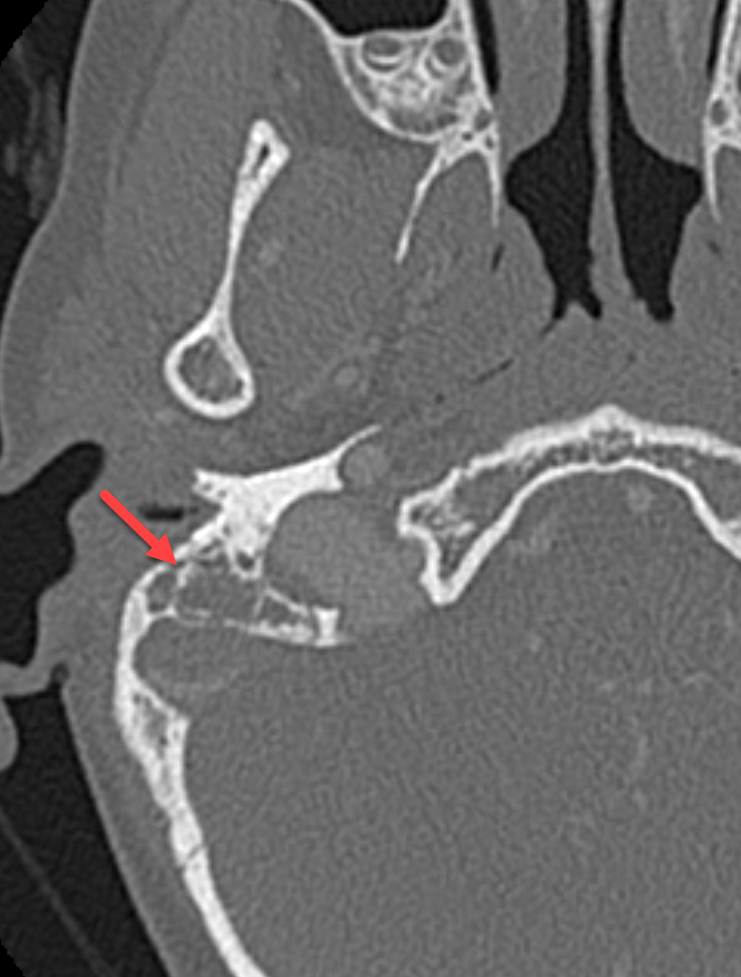
Contrast-enhanced CT demonstrated right-sided coalescent mastoiditis with occlusive thrombosis of the right sigmoid and transverse sinuses (Figure 1). Mastoidectomy, ventilation tube replacement, and three myringotomy incisions were performed. Subsequent anticoagulation and empiric oral antibiotics were started. Postoperatively, ventilation tube obstruction was observed and tube replacement in the clinical setting was unsuccessful, warranting revision surgery. Focal granulation tissue was appreciated intraoperatively along the posterior wall of the right ear canal and the posterior-superior aspect of the tympanic membrane. The canal was gently debrided and an occluded ventilation tube was identified and subsequently evacuated. After further debulking, three small myringotomies were created. Following this procedure, the patient reported right-sided otalgia, ear secretions, and nausea.
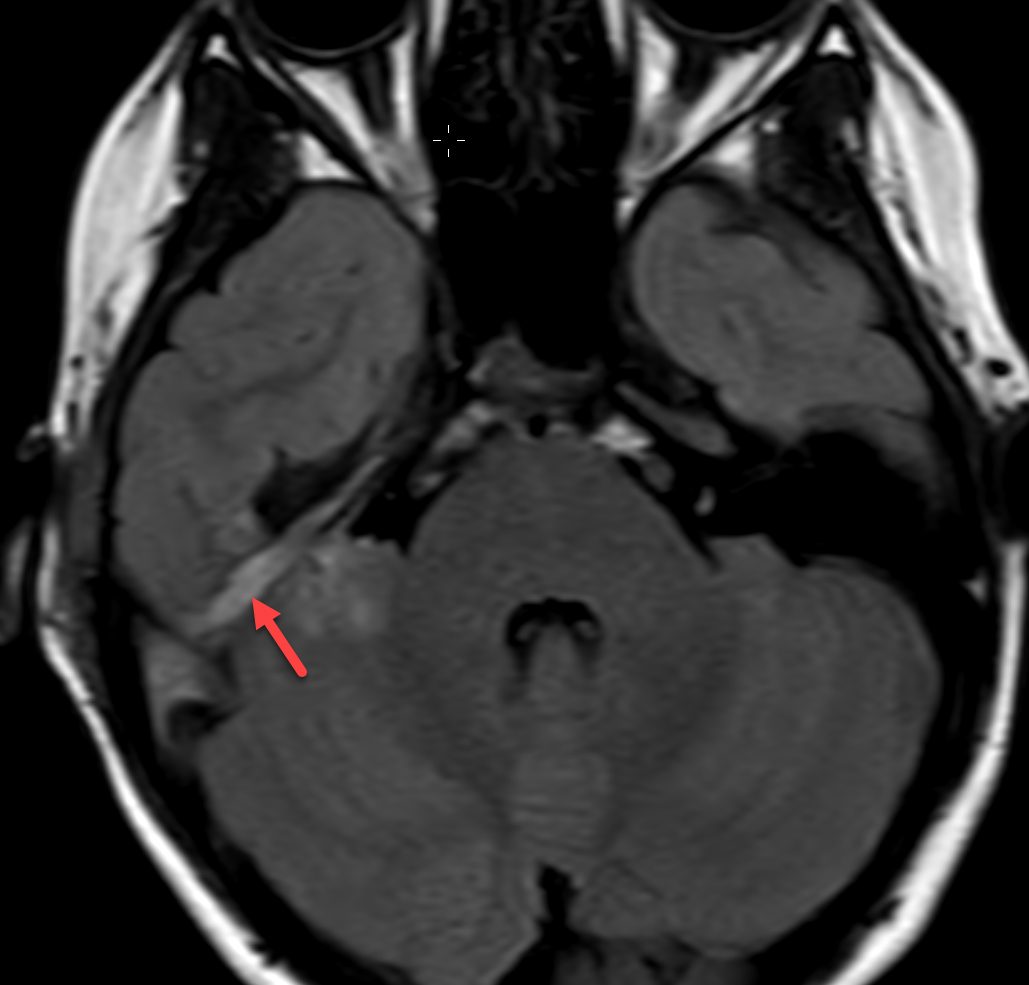
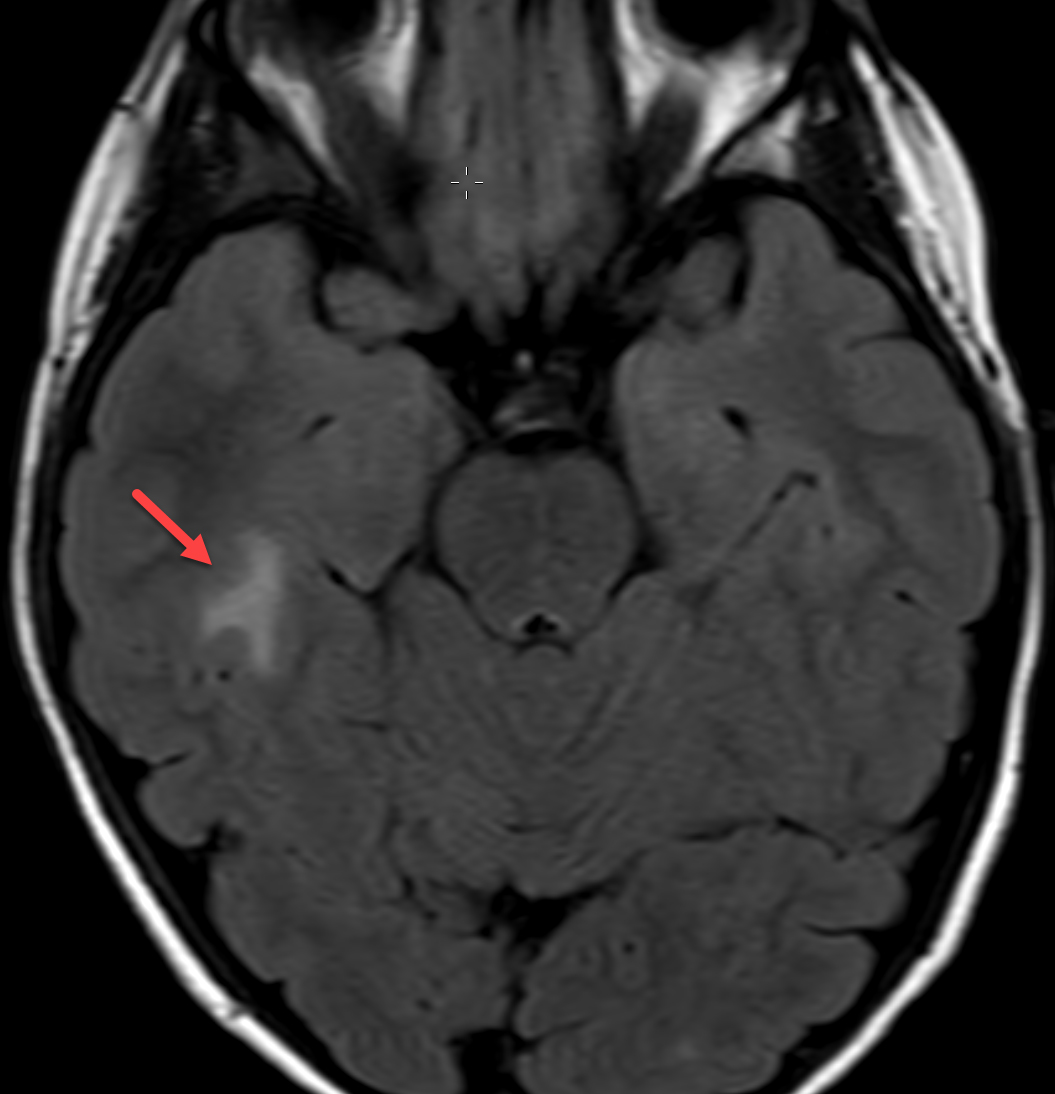
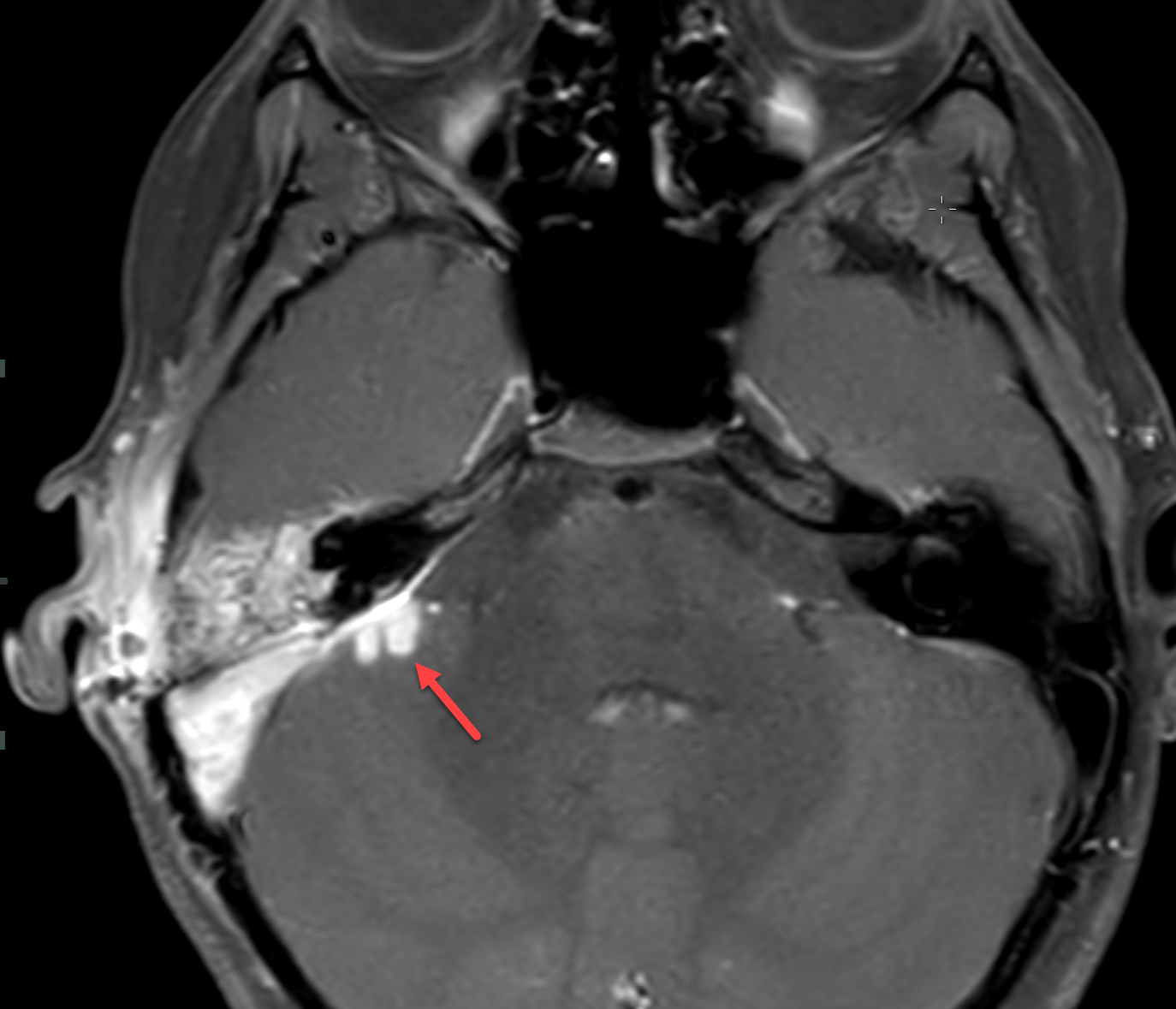
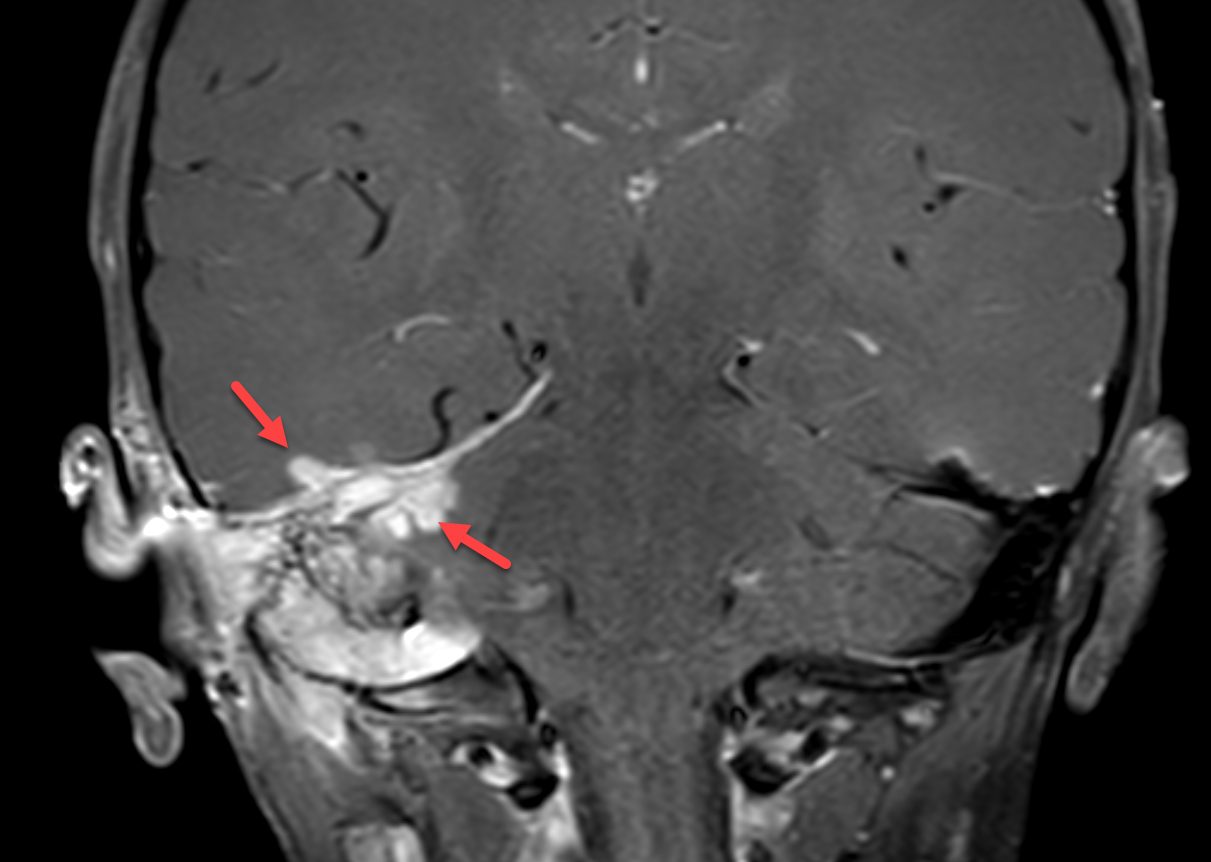
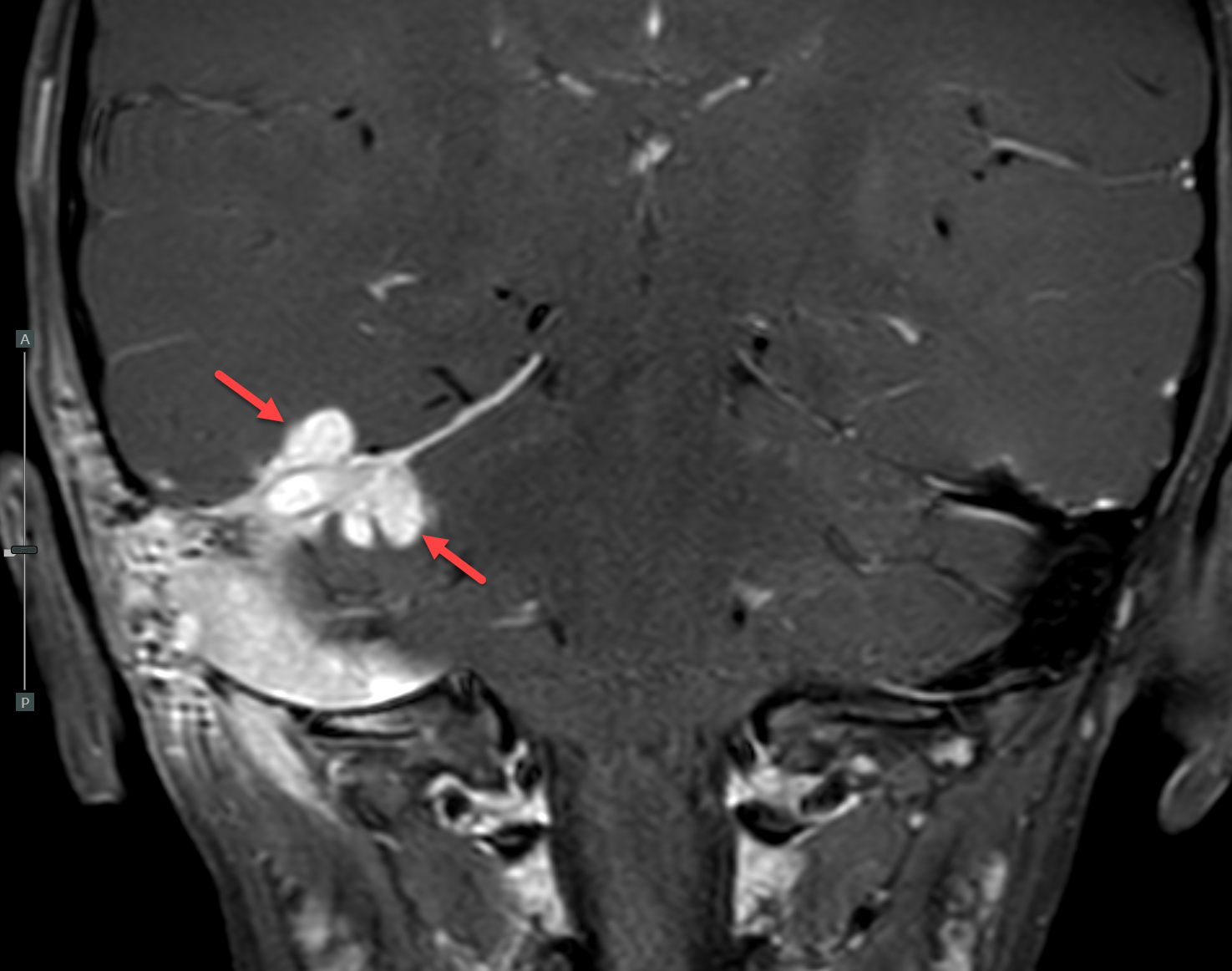
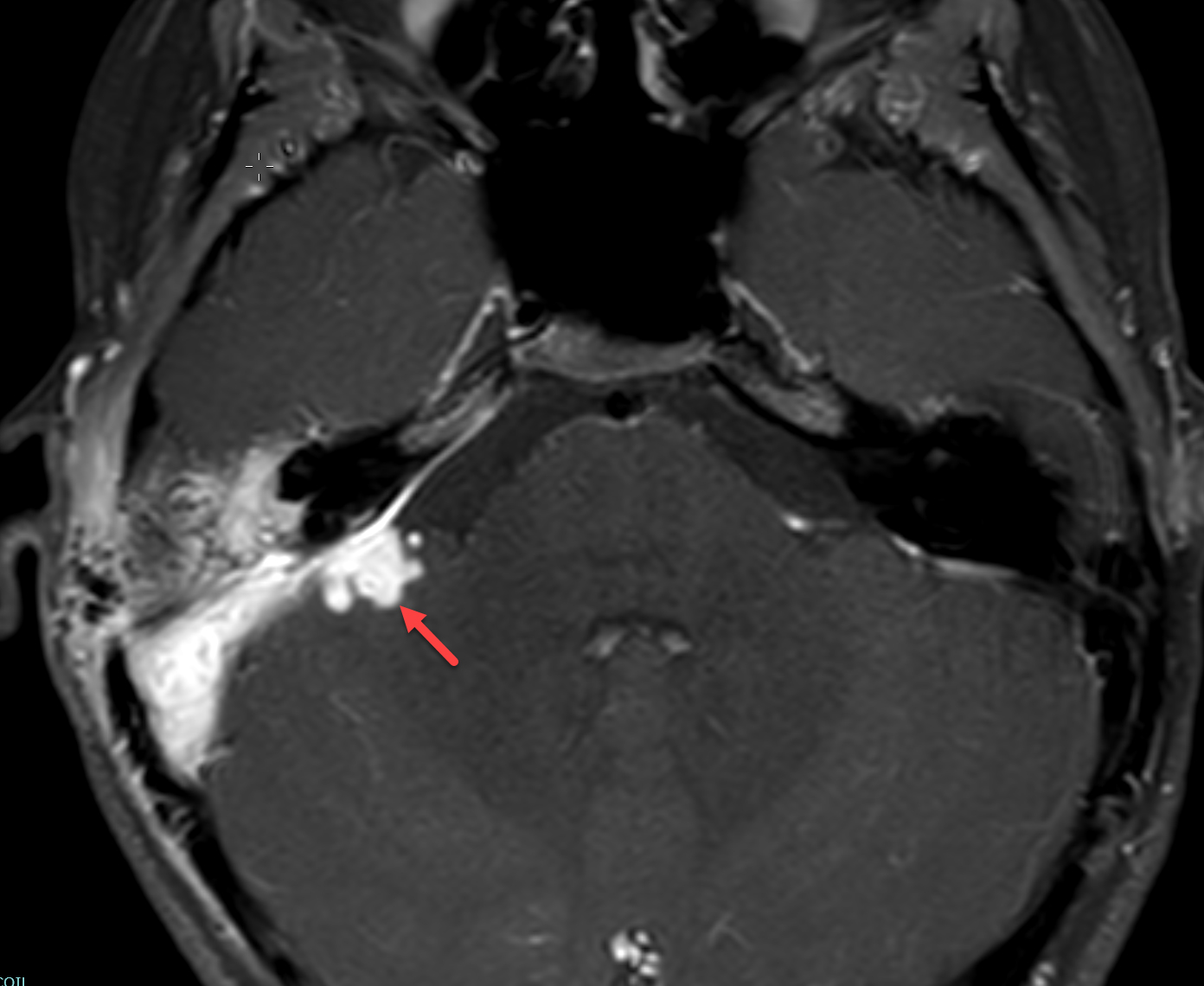
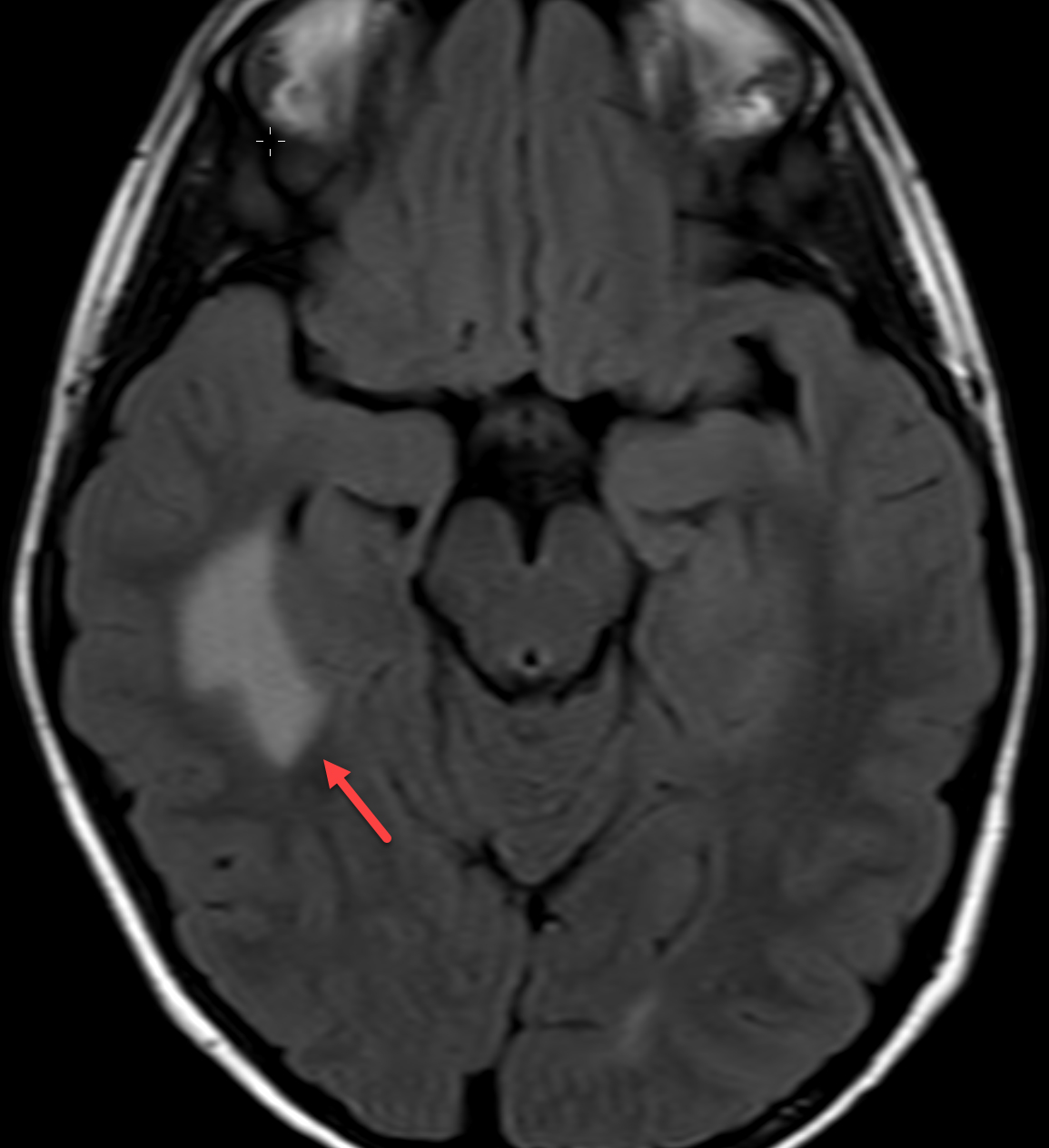
Magnetic resonance imaging (MRI) with and without contrast demonstrated contrast enhancement extending above the tegmen tympani with linear enhancement along the right tentorium. Also demonstrated were several foci of T2 hyperintensity and nodular enhancement on both sides of the right tentorium extending to the inferior aspect of the temporal lobe and the superior aspect of the right cerebellum (Figure 2). This was concerning for an infectious versus an inflammatory process disseminating through the tegmen tympani and petrous bone with involvement of the right tento rial dural lining. Vasogenic edema was also noted in the temporal lobe and cerebellum with redemonstration of sigmoid sinus thrombosis. The patient again underwent extensive debridement of the mastoid region, and right ear tissue cultures were collected. Gram stains revealed no organism growth and minimal leukocytes.
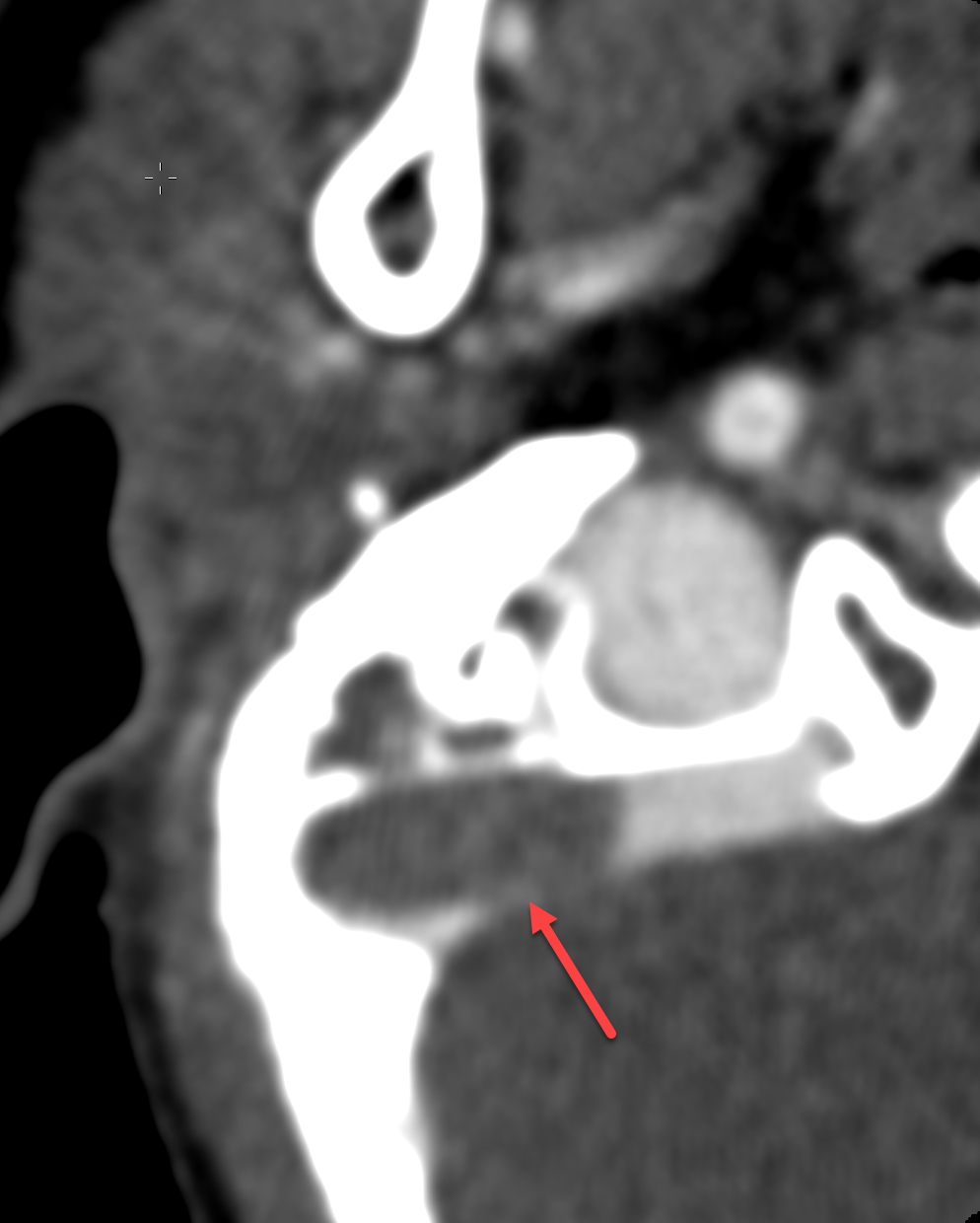
MRI was repeated six weeks later, demonstrating stable enhancement throughout the right mastoid and involving the right petrous apex, concerning for petrous apicitis and osteomyelitis. Also, increased vasogenic edema was appreciated in the adjacent right temporal lobe and cerebellum, while the right sigmoid sinus thrombus was stable (Figure 3). Meanwhile, a mycobacterial culture grew Mycobacterium abscessus. The patient was started on tigecycline, azithromycin, and amikacin, and was later discharged to home. At 3-month follow-up, clinical examination revealed significant symptomatic improvement, while a repeat MRI demonstrated resolution of the nodular areas of enhancement and the vasogenic edema.
Diagnosis
Non-tuberculous mycobacterial otomastoiditis with intracranial involvement
Discussion
Non-tuberculous mycobacterium (NTM) describes mycobacterial species not including M tuberculosis or M leprae. These organisms are ubiquitous in nature, with transmission usually taking place via contaminated soil and water but also via milk, food, and animals.1–4 Human-to-human transmission has not been described.1 Although these species are among the most common organisms identified from infections in HIV/AIDS patients, particularly those with CD4 count <50 cells/µL, NTM infections within the otomastoid region is rare, especially in immunocompetent patients due to low virulence.1,5–11 Lundman et al (2015) identified 89 such cases in the literature thus far.
As in our case, most NTM otomastoiditis cases are caused by M abscessus which, along with M fortuitum, represents the faster-growing NTM and typically infects male children younger than 6 years.1,4,9,10 Major risk factors across all age groups include recurrent otitis media and tympanic membrane perforation.1,4 Among children, specifically, insertion of ventilation tubes was identified most commonly 82% (54 out of 66), followed by tympanic membrane perforation, indicating transmission is likely via tympanic membrane defects.1,4 M avium complex, specifically, usually arises in younger patients (around 2 years old). It can also be acquired via consumption of contaminated soil as indicated by the relatively greater prevalence of cervical lymphadenopathy.1 Because these transmission methods bypass inhalation, patients are not necessarily more likely to be immunocompromised. In fact, most cases involve otherwise healthy children with no history of other disease.1,4,9
Once symptomatic, children almost always present with treatment-resistant ear canal secretions.1,4 Other common findings include polyp formation, granulation tissue in the middle ear, insidious mastoiditis, and facial nerve involvement.1,4,11 Imaging with CT, MRI, or both allows for evaluation of the cranial structures and the extent of spread.1,9,12 Typical findings include opacification of the middle ear cavity and mastoids with associated bone destruction and middle ear attenuation.1,4,11 About 81% (42 out of 52) of cases were limited to the temporal bone (6% limited to the middle ear alone), but as reported in the present case, NTM otomastoiditis can rarely extend intracranially to the temporal lobe, cerebellum, or meninges.1,4 NTM infections with central nervous system involvement can present vaguely with fever, headache, or vomiting, or more severely with hemiplegia, seizure, or altered mental status and yield heterogenous imaging findings ranging from no obvious inflammation to meningitis to abscess.1,4,6,11 In the present case, however, CT suggested NTM extension through the tegmen tympani, along the tentorium, involving the temporal lobe and cerebellum with venous sinus thrombosis.
As there are no imaging findings specific to NTM infection, culture and acid-fast bacilli stain are the gold standards for diagnosis.1,2,6,8 However, imaging can assist in guiding biopsy or culture.6 Owing to the organisms’ slow growth in vitro, other diagnostic methods, like PCR, can also be considered, while some cases can be diagnosed after surgical tissue/specimen is obtained intraoperatively.1,8
NTM otomastoiditis is typically treated with antibiotics under recommendations of the American Thoracic Society and the Infectious Disease Society of America.8 Owing to general susceptibility variance, pathogen-specific treatment is lacking and is typically guided by culture susceptibility.1,4,8 More recently, some authors have also proposed boric acid.9,11 Surgery is indicated in cases of extensive disease and abscess formation, and may require revision resulting from wound rup ture or granulation tissue obstructing the ear canal or ventilation tubes, as with our patient.1 Among immunocompetent or otherwise healthy patients, including those with CNS involvement, no fatal cases have been described.1,4 In immunocompromised patients, CNS involvement is often fatal.1–3,5,6 Long-term sequelae, meanwhile, correlate with disease severity and most commonly manifest as hearing difficulty, especially after surgical intervention.1,4,9
Conclusion
Non-tuberculous mycobacterial otomastoiditis is an infrequent yet serious condition. As non-routine testing is required for diagnosis, its rarity and resulting low clinical suspicion can delay appropriate management. Therefore, suspicion for NTM otomastoiditis is recommended for patients presenting with treatment-resistant ear secretions and insidious mastoiditis. Further, the potential for intracranial spread underlines the utility of CT/MRI imaging. Concrete treatment recommendations following diagnosis and evaluation, however, are lacking, due to the condition’s scarcity in the literature. Hence, continued reporting is warranted to better characterize NTM mastoiditis.
References
- Lundman L, Edvardsson H, Ängeby K. Otomastoiditis caused by non-tuberculous mycobacteria: Report of 16 cases, 3 with infection intracranially. Journal of Laryngology and Otology. 2015;129(7):644-655. doi:10.1017/S0022215115001309
- Padmanaban V, Hussein R, Rizk E. Nontuberculous Mycobacterial Infection in Patients with Neurosurgical Hardware: Two Cases and A Review of the Literature. Cureus. Published online March 24, 2020. doi:10.7759/cureus.7398
- Cai R, Qi T, Lu H. Central nervous system infection with non-tuberculous mycobacteria: a report of that infection in two patients with AIDS. Drug discoveries & therapeutics. 2014;8(6):276-279. doi:10.5582/ddt.2014.01047
- Yeh CF, Tu TY, Wang MC, et al. Emergence of Refractory Otomastoiditis Due to Nontuberculous Mycobacteria: Institutional Experience and Review of the Literature. Clinical Infectious Diseases. 2015;62(6):739-745. doi:10.1093/cid/civ1033
- Karne SS, Sangle SA, Kiyawat DS, Dharmashale SN, Kadam DB, Bhardwaj RS. Mycobacterium avium-intracellulare brain abscess in HIV-positive patient. Annals of Indian Academy of Neurology. 2012;15(1):54-55. doi:10.4103/0972-2327.93282
- Kwon LM, Kim ES, Lee K, Lee Y, Song JH. Nontuberculous mycobacterial meningoencephalitis in a young healthy adult: A case report and literature review. Radiology of Infectious Diseases. 2018;5(2):85-90. doi:10.1016/j.jrid.2017.07.001
- Little AA, Gebarski SS, Blaivas M. Nontuberculous mycobacterial infection of a metastatic brain neoplasm in an immunocompromised patient. Archives of Neurology. 2006;63(5):763-765. doi:10.1001/archneur.63.5.763
- Griffith DE, Aksamit T, Brown-Elliott BA, et al. An official ATS/IDSA statement: Diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. American Journal of Respiratory and Critical Care Medicine. 2007;175(4):367-416. doi:10.1164/rccm.200604-571ST
- Sédillot-Daniel È, Voizard B, Vallières É, Woods O, Quintal MC. Chronic suppurative otomastoiditis due to nontuberculous mycobacteria: A case series. International Journal of Pediatric Otorhinolaryngology. 2020;138:110375-. doi:10.1016/j.ijporl.2020.110375
- Lefebvre MA, Quach C, Daniel SJ. Chronic suppurative otitis media due to nontuberculous mycobacteria: A case of successful treatment with topical boric acid. International Journal of Pediatric Otorhinolaryngology. 2015;79(7):1158-1160. doi:10.1016/j.ijporl.2015.04.043
- van Ingen J, Looijmans F, Mirck P, Dekhuijzen R, Boeree M, van Soolingen D. Otomastoiditis caused by mycobacterium abscessus, the Netherlands. Emerging Infectious Diseases. 2010;16(1):166-168. doi:10.3201/eid1601.090473
- Walliczek-Dworschak U, Diogo I, Strack L, et al. Indications of cone beam CT in head and neck imaging in children. Acta Otorhinolaryngologica Italica. 2017;37(4):270-275. doi:10.14639/0392-100X-1219