Imaging infection and inflammation of the terminal ileocecal region: Emphasis on typhoid
Images
There is a broad spectrum of pathologies that can involve the ileocecal region of the bowel, caused by numerous pathogens and inflammatory conditions. Most likely these conditions will first be recognized by multidetector computed tomography (MDCT) supported by sonography, contrast enterography and magnetic resonance imaging (MRI). Both imaging and clinical features can overlap among these entities, requiring specific clinical details and a thorough knowledge of common and atypical imaging appearances to avoid delaying appropriate treatment. This article will review several of these conditions with a focus on the clinical and imaging presentations of typhoid. The importance of this condition relates to its impact as a worldwide health problem and the increasing potential to be encountered in the U.S. with the steady growth in international visitors to and from endemic areas
Infection presentation
Salmonella
Typhoid, a global health problem, can have a devastating impact on resource-poor countries. It is caused by Salmonella enteritidis serovar typhi (S. typhi)1,2 or by related but less virulent Salmonella paratyphi A. In Asia, the mean incidence of enteric fever is estimated to be 900 per 100,000 people per annum.3 This is a curable disease, but it has a mortality of 1-4%.4,5 Complications can occur in up to one-third of pediatric patients.5,6 The most lethal complications are gastrointestinal bleeding and ileal perforation.1 The clinical diagnosis can be delayed because the presentation may be atypical and cultures require at least 18-36 hours.4 The causative agent is a gram-negative facultative intracellular aerobe. Infection is acquired from ingestion of bacteria in contaminated food and water. Once a person is infected, organisms spread through a fecal-oral route, the bacterium proliferating in the small intestine and through the lymphatics of the intestine disseminates to various organs. Salmonella infections are most commonly self-limiting with symptoms treated empirically with antibiotics. Radiological investigations are limited to ultrasound and only rarely do complications occur which are likely to be investigated with cross-sectional imaging.
Salmonella: Organ involvement
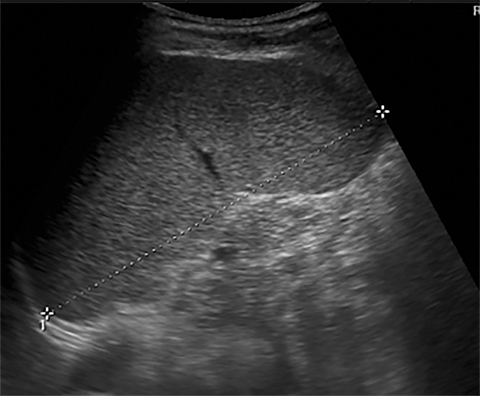
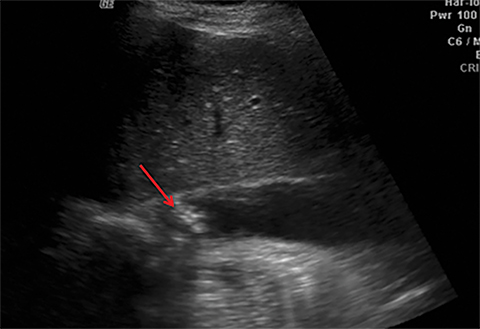
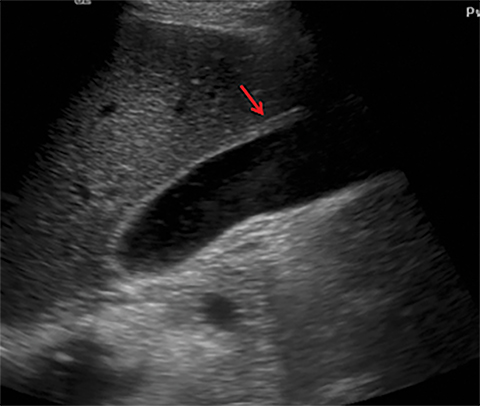
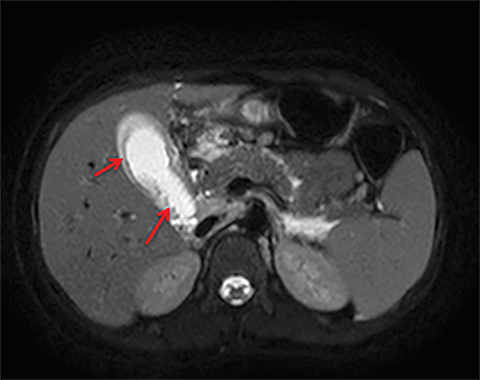
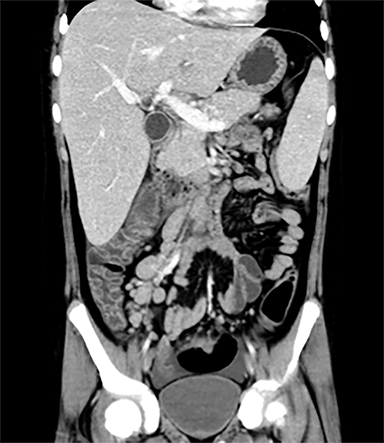
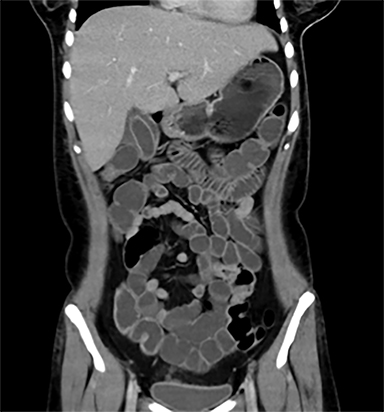
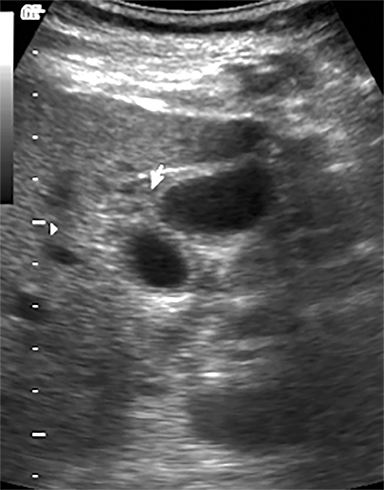
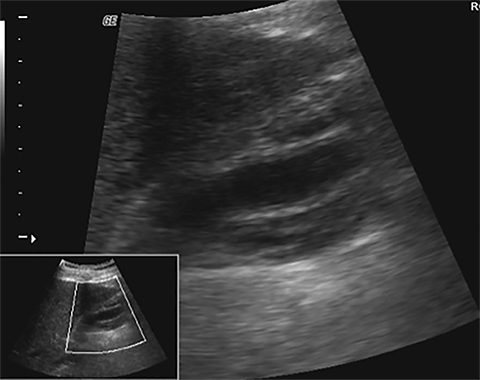
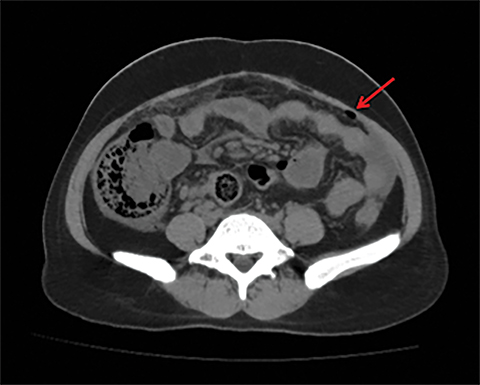
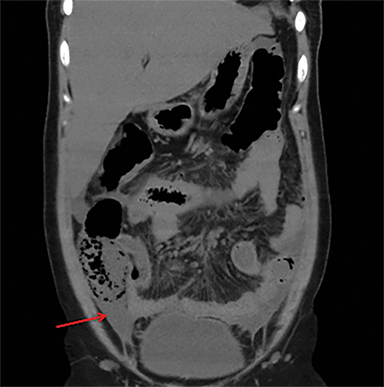
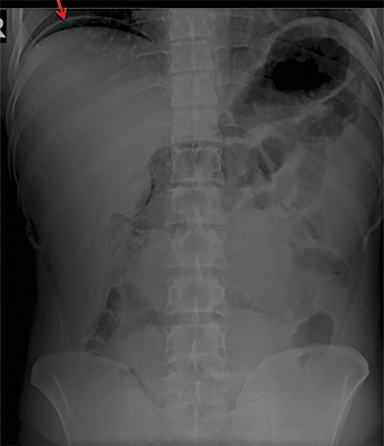
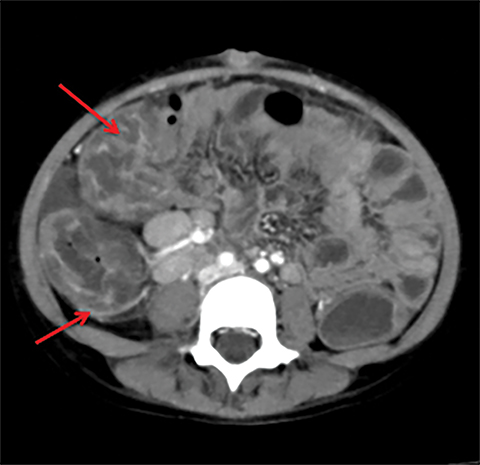
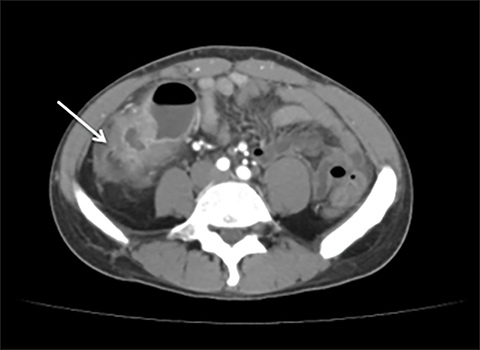
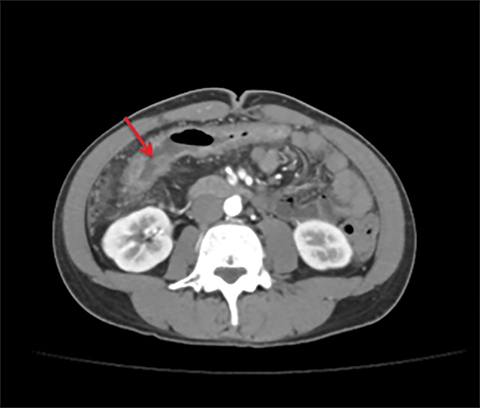
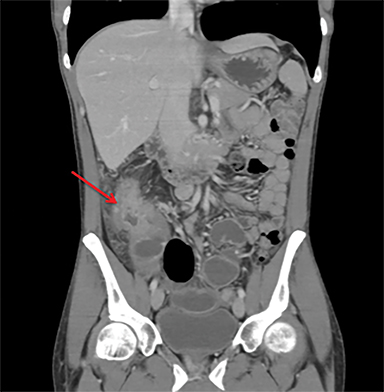
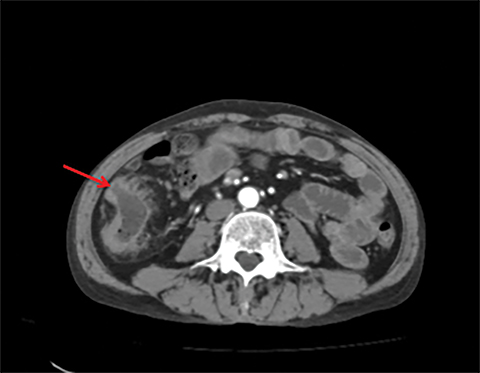
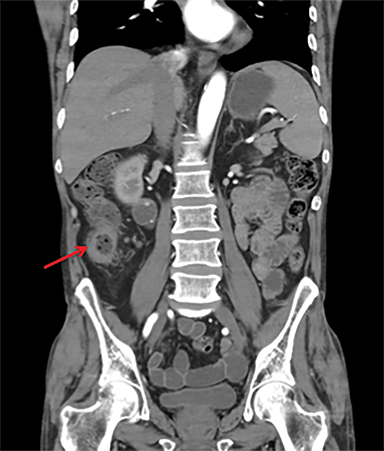
Terminal ileal and proximal colonic thickening is typical of typhoid and should be considered in appropriate clinical settings. Uncommon abdominal manifestations of typhoid include cystitis, pyelonephritis and renal abscess.7 Khan et al have reported a rare case of typhoid rhabdomyolysis leading to acute renal failure.8 Neha et al have reported epididymoorchitis caused by salmonella.9 Pleural effusion pleurisy, broncho and lobar pneumonia have also been described7.Cardiac manifestations include endo, myo and pericarditis,but these are quite uncommon.10 Salmonella is one of the most common causes of infectious aneurysm.10 Central nervous system involvement can cause meningitis and encephalitis resulting in hydrocephalus and cerebral abscess.7 Salmonella osteomyelitis is well known to occur given preexisting sickle cell disease. Other complications include septic arthritis, periostitis and abscess formation. Common sites of involvement include the gastrointestinal tract, hepatobiliary system, spleen, peritoneum and mesentery. Mesenteric and peritoneal involvement in salmonella is seen as ascites, localized or generalized, with mesenteric thickening and lymphadenopathy. Nonspecific organomegaly is the most common sign of liver and spleen involvement (Figure 1),1 rarely presenting as an organ abscess.7 Splenic rupture secondary to trivial trauma of an enlarged spleen is a rare complication. Acute acalculus cholecystitis is a known manifestation in salmonella infection seen as gallbladder wall thickening more than 3mm, gross distention more than 40mmin length] with pericholecystic fluid seen on both ultrasound and CT (Figure 2 – 4). 11 Gallbladder perforation and cholangitis are rare.
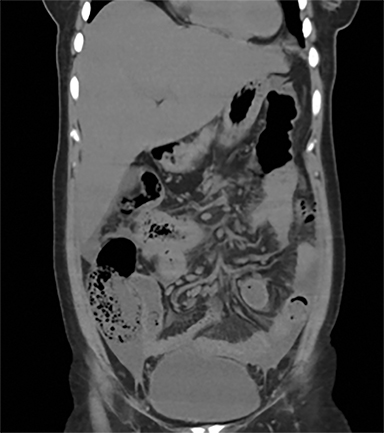
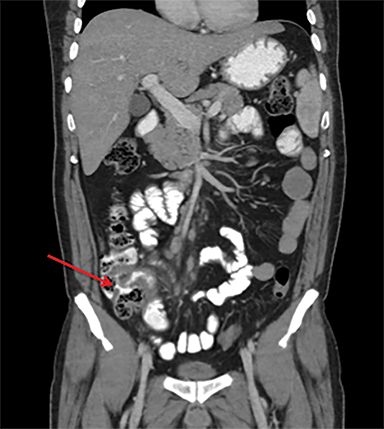
Intestinal complications of salmonella include GI bleed and perforation of the terminal ileum. The latter occurs due to necrosis of the Peyer’s patches in the terminal ileum, resulting in ulceration, bleeding and, in extreme cases, full thickness perforation (Figure 5).11 Causes of perforation include inadequate antimicrobial treatment prior to admission, short duration of symptoms and leucopenia.1 Active bleeding is seen as contrast extravasation on CT angiography.
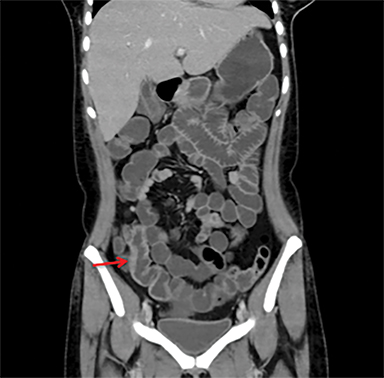
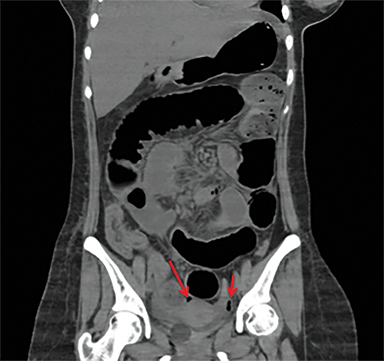
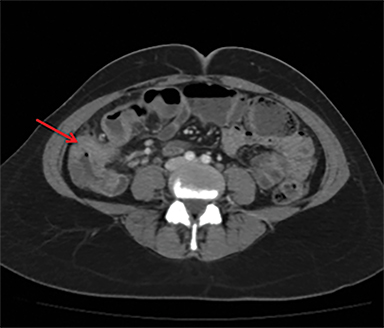
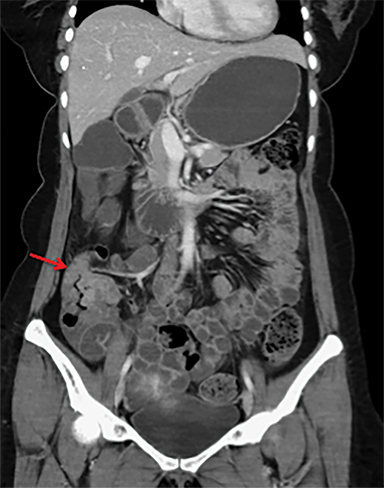
Overcoming gastric acidity and competing with the normal intestinal flora, Salmonella bacterium elaborates several toxins that affect intestinal cells. At CT, involved bowel typically shows wall thickening, which usually demonstrates homogeneous contrast enhancement. Low attenuation intramural edema may be detected. Ascites or inflammation of the pericolic fat can also be present. Multiple air-fluid levels can be present in the colon due to increased fluid and liquid feces.4, 12 Enlarged mesenteric lymph nodes are seen adjacent to the involved segment .4, 7 Thickening of the terminal ileum and proximal colon is typical (Figure 6).7 Salmonella enteritis may simulate other causes of intestinal wall thickening such as tuberculosis, Crohn’s, neutropenic typhilitis, ischemic bowel, other infectious enteritis and malignancies of the ileocecal region.
Tuberculosis
Tuberculosis most commonly involves the ileocecal portion of the gastrointestinal tract. Characteristic CT features include asymmetric thickening of the ileocecal valve and medial wall of the cecum (Figure 7), engulfing the terminal ileum and massive lymph nodes with central low-attenuation areas. Thickening of the valve lips or wide gaping of the valve with narrowing of the terminal ileum (the Fleischner sign) has been described as a characteristic of tuberculosis. Fistulas and sinus tracts are rare. Massive adenopathy is more suggestive of tuberculosis13. Associated findings such as peritoneal involvement, a history of tuberculosis, a positive tuberculin skin test and characteristic chest radiographic findings provide additional diagnostic clues. Other causes of infectious terminal ileitis include Yersinia and Campylobacter, but they generally involve the jejunum as well.12
Inflammation
Fat stranding that is disproportionately more severe than the degree of wall thickening is a finding that suggests inflammatory diseases such as diverticulitis, appendicitis, or epiploic appendagitis.
Crohn’s disease
Crohn’s colitis imaging features include abnormal wall thickening with skip areas, colonic stenosis and dilatation. Active disease may manifest as mucosal hyper enhancement or mural stratification (Figure 8) Associated findings include pericolonic fat stranding, fistulas, abscesses, fibro-fatty proliferation and mesenteric adenopathy.
Neutropenic colitis (Typhilitis)
Patients being treated for underlying leukemia or lymphoma may rarely present with neutropenic colitis (typhilitis) as an acute complication. CT findings include bowel wall thickening, intramural air (pneumatosis), and pericecal inflammatory fat stranding or fluid. The CT density of the bowel wall may be of low attenuation secondary to edema.12 The inflammatory changes may also encompass the ascending colon and the distal ileum and have also been found isolated to the small bowel.14, 15
Ischemic typhilitis
Ischemic typhilitis has similar CT findings at the ileocecal region; however the presence of pneumatosis favors an ischemic cause.12 The presence of thrombus within the mesenteric vessels is diagnostic; however , it need not be present in all cases, as small vessel vasculitis may just be seen as bowel wall thickening, as seen in in one of our cases (Figure 9), where intestinal perforation was the initial clinical presentation. The CT images of this patient (Figure 9) revealed distal small bowel thickening with ischemic changes and contained local perforation. Peri-operatively, a long ischemic segment was identified and resected, later typhoid induced mesenteric vasculitis was diagnosed.
Diverticulitis
Diverticulitis in this region can mimic typhoid typhilitis. The presence of focal thickening and stranding of mesenteric fat out of proportion to the bowel thickening and the identification of diverticula aids in the diagnosis.15
Most benign conditions, infective or inflammatory, including tuberculosis have a stratified contrast enhancement pattern in the bowel wall seen as “double halo” or “target” configuration, a key feature to differentiate them from malignant bowel wall thickening. However, this stratification pattern may not always be present in chronic inflammatory conditions, hence clinical history plays an important role in leading to the correct diagnosis.
Malignancy
Malignant causes of ileocecal junction thickening like adenocarcinoma, can usually be obviously differentiated from inflammation or infection as they are seen as irregular short, asymmetric wall thickening (Figure 10), with abrupt margins and/or annular stricture. Fat stranding is usually minimal unless there is perforation. Lymphoma at the ileocecal region is seen as pliable and long symmetric thickening, polypoidal lesion, or exocentric mass with no or minimal fat stranding. A stratified enhancement pattern in a thickened segment of bowel wall is used to exclude malignant conditions and is, therefore, a very important criterion to use to establish a correct diagnosis. In general, benign conditions result in bowel wall thickening of less than 2 cm, whereas wall thickening greater than 3 cm usually indicates a neoplastic condition. With exceptions (mainly lymphoma), long segments of involvement indicate a benign condition.15
Conclusion
Typhoid remains an important public health problem in the world, especially in developing countries, which have episodic typhoid epidemics. Given the ever-increasing travel of persons to and from endemic regions, typhoid should be considered in the diagnosis of acute ileocecal pathology in the U.S. and other developed nations where the disease is not commonly encountered. Imaging findings in salmonella infection can be nonspecific and thus mimic other diseases. Differentiation of this easily treatable entity from other various mimics is essential. Imaging plays a pivotal role in some cases where the clinical presentation is atypical and can determine the necessity of further studies and prompt initiation of treatment. Potentially serious complications might involve multiple organs and may present in unusual fashion. In general, greater awareness of and familiarity with this disease and its complications among radiologists will allow for earlier diagnosis and will, with supportive care and adequate antimicrobial therapy, reduce morbidity and mortality.
References
- Hosoglu S, Aldemir M, Akalin S, et al. Risk factors for enteric perforation in patients with typhoid fever. Am J Epidemiol. 2004;160(1):46-50.
- Cotran, Kumar, Collins. Robbins Pathologic Basis of Disease, 6th ed. Philadelphia: Saunders; 1999:835.
- Ivanoff, B. Typhoid fever: global situation and WHO recommendations. In: Proceedings of the 2nd Asia Pacific Symposium on Typhoid Fever and Other Salmonellosis. Bangkok: Infectious Disease Association of Thailand 1994:39.
- Puylaert J B, Sjofn Kristjansdottir S, Golterman K L, et al. Typhoid fever: diagnosis by using ultrasonography. AJR Am J Roentgenol. 1989; 153(4):745-746.
- Surgical complications of typhoid fever. Ameh E, Abantanga F. In Ameh E, Bickler S, Lahooo K, eta al: Paediatric Surgery: A Comprehensive Text For Africa. Chapter 17. Available at: http://www.global-help.org/publications/books/help_pedsurgeryafricavolume01.pdf:103-110.
- Malik AS. Complications of bacteriologically confirmed typhoid fever in children. J Trop Pediatr. 2002; 48(2):102-108.
- Hennedige T, Bindl D S, Bhasin A, et al. Spectrum of imaging findings in Salmonella infections. AJR Am J Roentgenol. 2012; 198(6):W534-539.
- Khan FY, Al- Ani A, Ali HA. Typhoid rhabdomyolysis with acute renal failure and acute pancreatitis: a case report and review of the literature. Int J Infect Dis. 2009; 13(5); e282-285.
- Bansal N, Kaistha N, Chander J. Epididymo-orchitis: An unusual manifestation of salmonellosis. J Microbiol Immunol Infect. 2012;45(4): 318-320.
- Hibbert B , Costiniuk C, Hibbert R, et al. Cardiovascular complications of Salmonella enteritidis infection. Can J Cardiol.Oct 2010;26(8): e323–e325.
- O’Connor O J, Maher M M. Imaging of cholecystitis. AJR Am J Roentgenol. 2011;198(4):367-374.
- Horton KM, Corl F M, Fishman E K, MD CT evaluation of the colon: inflammatory disease. RadioGraphics. 2000; 20(2):399–418.
- Engin G, Acunas B, Acunas G, et al. Imaging of extrapulmonary tuberculosis. RadioGraphics. 2000; 20(2):471–488.
- Silva A C, Beaty S D, Hara A K, et al. Spectrum of normal and abnormal CT appearances of the ileocecal valve and cecum with endoscopic and surgical correlation. RadioGraphics. 2007; 27(4):1039 –1054.
- Hoeffel C, Crema M D, Belkacem A, et al. Multi-detector row CT: spectrum of diseases involving the ileocecal area. RadioGraphics. 2006; 26(5):1373–1390.
Citation
A R, S N, A P, MallarajapatnaG, M H, SG M. Imaging infection and inflammation of the terminal ileocecal region: Emphasis on typhoid. Appl Radiol. 2016;(8):22-27.
August 1, 2016