Imaging Acute Face and Neck Infections
Images
This article is accredited for one SA-CME credit. Visit appliedradiology.org/SAM2 for full SA-CME information.
Avariety of acute infectious disorders of the face and neck, resulting from such common sources as pharyngitis, dental infection, and penetrating trauma, may present to the emergency department. Clinical localization of such infections, especially those involving the deep neck, is limited. In one series of patients with deep neck involvement, clinical examination localized the space involved in only 42.9% of cases.1 Accurate characterization of infections can determine the most appropriate therapy; ie, aspiration/drainage versus antibiotic therapy.
Nevertheless, despite widespread access to antibiotics, face and neck infections still present significant morbidity and mortality. Potentially life-threatening complications, including airway obstruction, mediastinitis, and septic emboli, are reported in 10-20% of deep neck infections.2,3 Emergency radiologists play a critical role in diagnosing, localizing, characterizing, and identifying the complications resulting from acute face and neck infections.
Dental Abscess
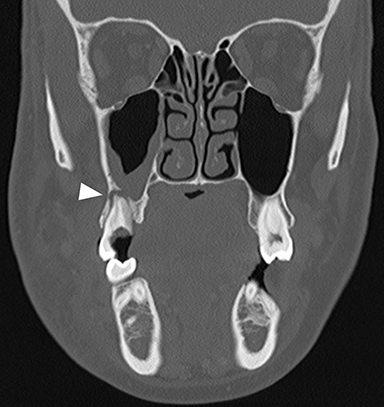
Dental caries, broken teeth, or periodontal disease can predispose teeth and adjacent soft tissues to bacterial infections. Dental abscesses, focal collections of pus associated with bacterial infection, occur most commonly adjacent to the tooth root tip, and are termed periapical abscesses. Infection can also arise from the tissues along the margins of the tooth roots, and is known as a periodontal abscess. Such abscesses can result in focal cortical destruction of the alveolar ridge and extend into the adjacent subperiosteal and extraosseous spaces, resulting in facial cellulitis.
On computed tomography (CT), osseous lucency around the tooth root apex suggests a periapical abscess (Figure 1A).4 Rim-enhancing fluid collections and associated inflammatory fat stranding in the adjacent buccal space are compatible with extension of infection (Figure 1B). An osseous breach of the alveolar ridge between the periapical abscess and adjacent subperiosteal or extraosseous abscess is frequently present.
Masticator Space Abscess
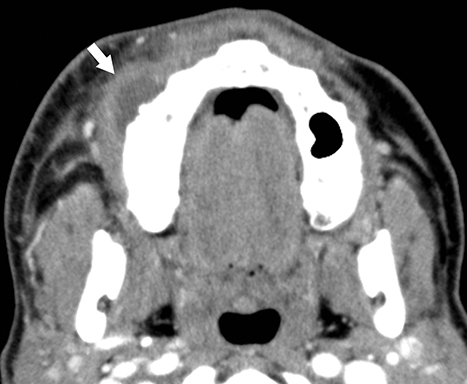
The masticator space is contained by the superficial layer of the deep cervical fascia, which encompasses the muscles of mastication and a portion of the posterior mandible. Dental infections of the posterior mandibular molars may breach into the masticator space, resulting in abscess formation that may clinically present as trismus.
On CT, a rim-enhancing fluid collection along the posterior margin of the mandible close to the muscles of mastication is compatible with a masticator space abscess (Figure 2A). Imaging should cover the superior extent of the temporalis muscles, as the masticator space continues superiorly above the zygomatic arch to the parietotemporal scalp (Figure 2B).5 The ipsilateral mandibular teeth should be assessed for underlying dental abscess. Additional potential infectious sources include septic arthritis of the temporomandibular joint and coalescent mastoiditis.
Ludwig Angina
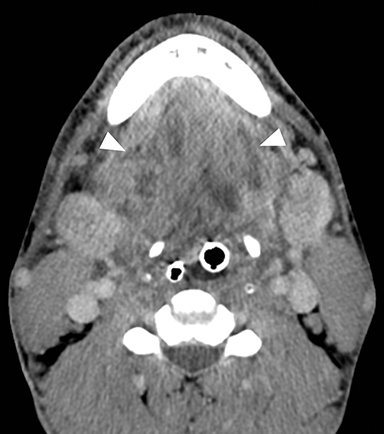
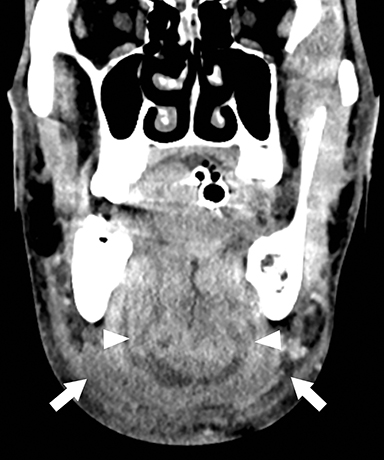
Ludwig angina is a potentially life- threatening bacterial cellulitis of the sublingual and submandibular spaces that can result in acute airway compromise. This infection is most commonly the result of extension of infection from an odontogenic source.6 On CT, Ludwig angina demonstrates inflammatory stranding and swelling within the bilateral sublingual spaces in the floor of mouth (Figure 3A).7 Additional inflammatory stranding in the fat of the submandibular spaces inferior and lateral to the mylohyoid muscles is typically present (Figure 3B). Rim-enhancing fluid collections compatible with abscesses occasionally may be seen. Presence of a periapical lucency with potential lingual cortical breakthrough involving a second or third mandibular molar suggests the inciting dental abscess.
Odontogenic Sinusitis
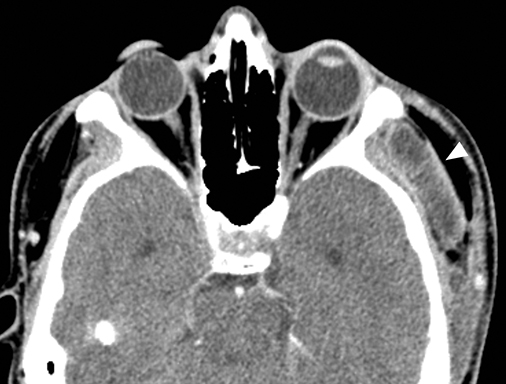
Dental abscesses adjacent to the roots of the maxillary premolars and molars may result in inflammatory mucosal thickening or acute bacterial sinusitis of the adjacent maxillary sinuses, termed odontogenic sinusitis. This often-overlooked etiology of paranasal sinus disease results in up to 40% of unilateral maxillary sinusitis.8 Although isolated maxillary sinusitis is the most common form, inflammatory mucosal thickening can extend through the maxillary infundibulum to the middle meatus, resulting in an ostiomeatal unit pattern of obstructive paranasal sinus disease with ipsilateral anterior ethmoid and frontal sinusitis. Treatment for odontogenic sinusitis centers around clearing the dental abscess with antibiotics and root canal or dental extraction.
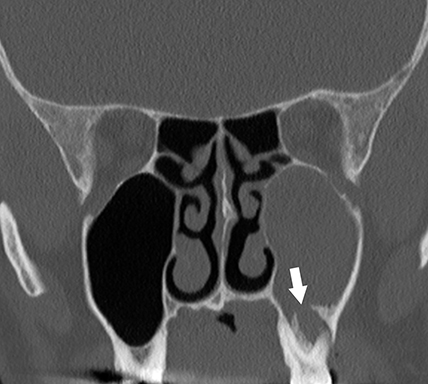
On CT, odontogenic sinusitis typically demonstrates unilateral maxillary sinus inflammatory mucosal thickening and sinus fluid with concomitant adjacent oroantral fistula, periodontal disease with projecting tooth root, or periapical abscess (Figure 4).9 Osseous dehiscence between the periapical lucency and maxillary sinus may be present.
Tonsillitis
Tonsillitis is a type of pharyngitis resulting from inflammation of the palatine tonsils, typically from a rapidly progressive infection. Most cases are the result of viral infection, most commonly due to adenovirus, rhinovirus, influenza, coronavirus, or respiratory syncytial virus.10 Epstein-Barr virus can also cause tonsillitis as part of infectious mononucleosis. Approximately 20% of tonsillitis results from bacterial infection, typically following a preceding viral infection. The most common bacterial etiology is Group A Streptococcus, known as strep throat. Haemophilus influenzae, Neisseria gonorrhoeae, and Corynebacterium diphtheriae are rare bacterial causes of tonsillitis. Treatment for tonsillitis is typically supportive with anti-inflammatories/analgesics for viral etiologies, with the addition of antibiotics for confirmed bacterial causes.
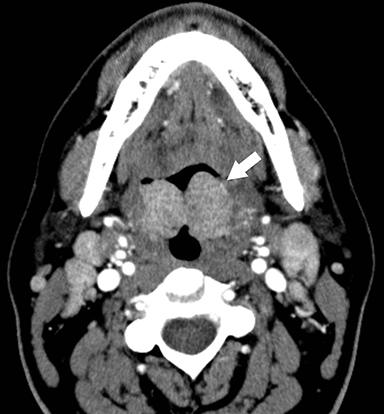
On contrast-enhanced CT, tonsillitis is evidenced by enlarged palatine tonsils, which can touch in the midline of the oropharynx, termed “kissing tonsils” (Figure 5).11 The tonsils also typically demonstrate a striated, “tiger stripe” appearance in which the enhancing mucosal lining along the crypts is contrasted by low-density edema within the submucosa of the glands.
Peritonsillar Abscess
The most common ENT emergency is peritonsillar abscess, a complication of untreated or incompletely treated bacterial tonsillitis. Peritonsillar abscess occurs as infection spreads deep between the tonsillar capsule and superior constrictor muscle, forming a collection of pus.12 Since peritonsillar abscesses necessitate more aggressive management, such as aspiration of pus and adding steroids to antibiotics, imaging characterization directly affects patient management.
On CT, peritonsillar abscesses will appear as rim-enhancing fluid collections along the deep margins of the inflamed palatine tonsil (Figure 6).13 These collections can result in localized mass effect with narrowing of the oropharyngeal airway. These abscesses also typically result in inflammatory stranding in the adjacent neck soft tissues, usually in the fat of the parapharyngeal and retropharyngeal spaces.
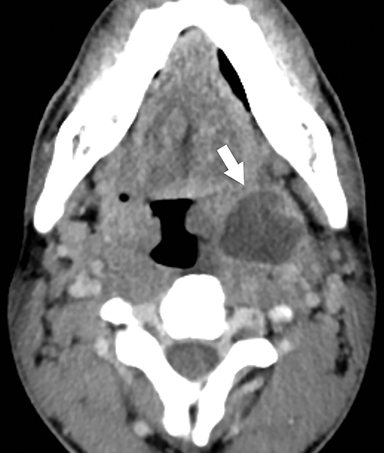
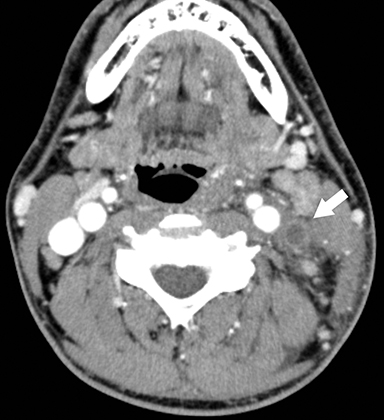
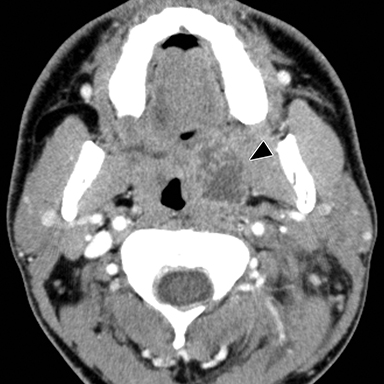
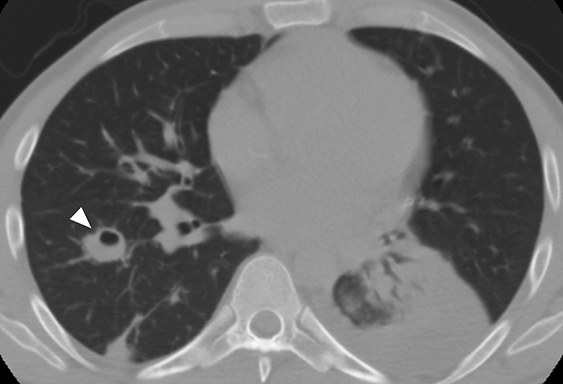
Lemierre Syndrome
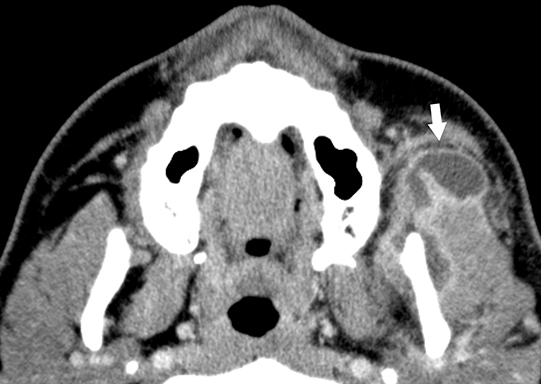
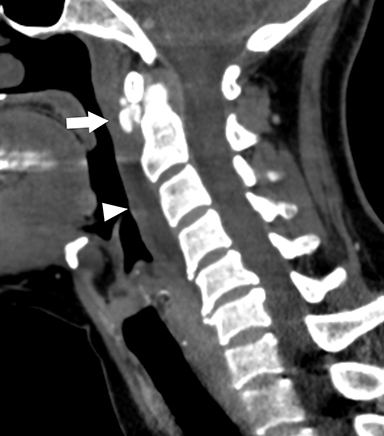
Infectious thrombophlebitis of the internal jugular vein (IJV), also known as Lemierre syndrome, is a rare and potentially life-threatening complication of peritonsillar abscess.14 This infection most commonly occurs following the spread of the anaerobic bacteria Fusobacterium necrophorum, a constituent of the normal oral flora, to the IJV. Infectious thrombi can break off from the IJV and travel to the lungs, resulting in pulmonary septic emboli. Less commonly, septic microemboli can bypass the pulmonary capillaries and cause infection or infarctions elsewhere in the body, typically septic arthritis of the large joints. Intravenous antibiotics targeting anaerobic organisms and peritonsillar abscess drainage (if present) is first-line treatment for Lemierre syndrome. Ligation of the IJV occasionally may be necessary.
On CT, IJV thrombophlebitis shows non-opacification of the vein lumen with a thickened, enhancing wall and adjacent inflammatory stranding (Figure 7A).15 There is typically an ipsilateral peritonsillar abscess (Figure 7B). Pulmonary septic emboli in the partially visualized portions of the lung apices are also sometimes detected on CT of the neck. Pulmonary septic emboli appear as nodular opacities with ill-defined margins, typically within internal cavitation (Figure 7C).
Retropharyngeal Suppurative Lymphadenitis
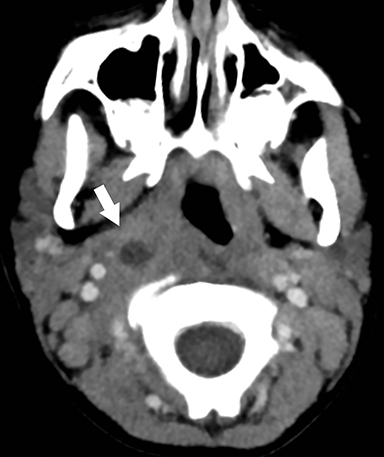
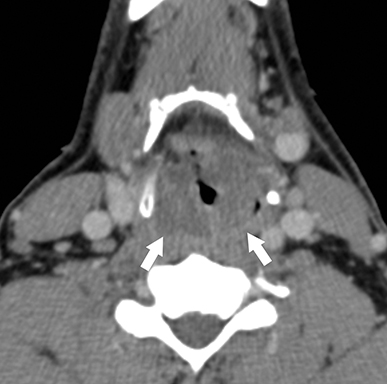
Retropharyngeal suppurative lymphadenitis denotes lateral retropharyngeal lymph node inflammation resulting from a bacterial infection that has progressed to internal liquefaction. This condition is most commonly encountered in young children (2-6 years of age) exhibiting fever, neck pain, dysphagia, and/or torticollis. The most commonly involved bacteria are Streptococcus sp. and Staphylococcus aureus.16 Intravenous antibiotics are the first-line treatment for this infection, with aspiration or surgical drainage reserved for refractory cases or large fluid collections.
On CT, a rim-enhancing fluid collection confined to the lateral margin of the retropharyngeal space with adjacent inflammatory stranding is compatible with retropharyngeal suppurative lymphadenitis (Figure 8).17 There is typically associated retropharyngeal edema or effusion, but rim-enhancing fluid should not cross the midline or fill the retropharyngeal space, which would suggest development of a retropharyngeal abscess.
Retropharyngeal Abscess
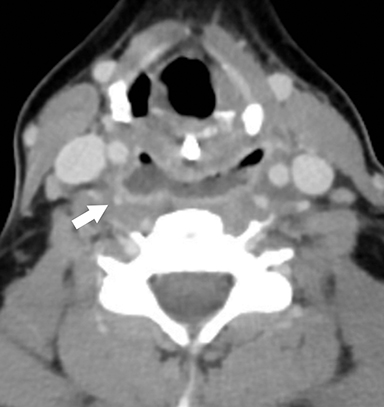
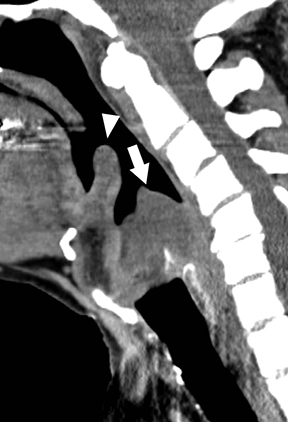
Infection from pus-forming bacteria in the retropharyngeal space results in retropharyngeal abscess. In adults, this condition most frequently is the result of a penetrating trauma with inoculation of normal oral flora into the retropharyngeal space. Retropharyngeal spread of infection from adjacent cervical discitis-osteomyelitis or ruptured retropharyngeal suppurative lymphadenitis in children are other potential etiologies. Retropharyngeal abscesses may spread to the danger space, a potential space situated between the true retropharyngeal space and prevertebral musculature. This permits free communication to the mediastinum, resulting in the dreaded complication of mediastinitis.18 Treatment with IV antibiotics and surgical drainage is standard of care.
On CT, retropharyngeal abscesses are rim-enhancing fluid collections that fill the retropharyngeal space (Figure 9A).19 Larger fluid collections typically demonstrate bowing of the walls of the retropharyngeal space. In cases of retropharyngeal abscess, it is essential to image the chest to search for inflammatory stranding or rim-enhancing fluid collections in the mediastinum, indicating the complication of mediastinitis (Figures 9B & C).
Calcific Tendinitis of the Longus Colli
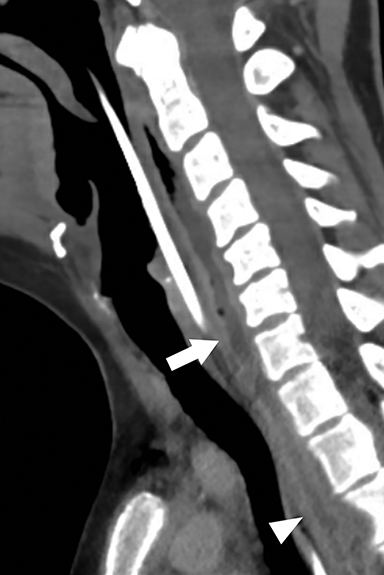
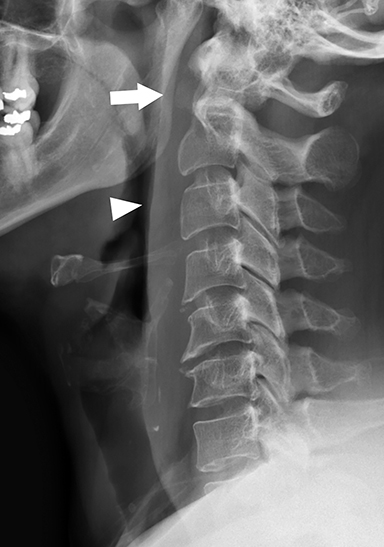
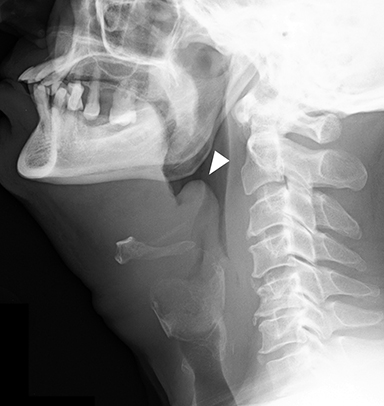
Calcific tendinitis is a self-limiting inflammatory response to calcium hydroxyapatite crystal deposition into the tendons of the longus colli muscles, typically at their superior insertions at the C1-C2 level. Although not an infection, this inflammation affects the prevertebral and retropharyngeal spaces, and can result in symptoms similar to retropharyngeal abscess, including neck pain, fever, dysphagia, odynophagia, and elevated serum inflammatory markers (eg, C-reactive protein, erythrocyte segmentation rate). Imaging is critical in differentiating this entity from retropharyngeal abscess, as calcific tendinitis of the longus colli is treated conservatively with nonsteroidal anti-inflammatory drugs.
On lateral neck radiography, calcific tendinitis of the longus colli will demonstrate thickening of the prevertebral soft tissues and may identify amorphous calcifications along the inferior margin of the C1 anterior arch (Figure 10A).20 On CT, the calcifications are more easily identified at the tendinous insertion of the longus colli muscles. In addition, inflammatory stranding and/or effusion expands the retropharyngeal space (Figure 10B).21 In contrast to retropharyngeal abscess, the retropharyngeal effusion has little to no rim enhancement.
Epiglottitis/Supraglottitis
Epiglottitis (termed supraglottitis in adult patients) is an acute bacterial infection resulting in inflammation of the epiglottis and adjacent supraglottic larynx. In children, epiglottitis is a life-threatening emergency causing fever, respiratory distress, stridor, drooling, muffled voice, and tripod posturing,requiring emergent airway management. The incidence of childhood epiglottitis dropped following institution of routine vaccination against Haemophilus influenzae type B.22 With increasing incidence in adults, supraglottitis more commonly presents with dysphagia and odynophagia, but it may still progress to respiratory distress. Aside from airway protection, epiglottitis is treated with intravenous antibiotics and corticosteroids.
In children, diagnostic evaluation with upright frontal and lateral radiographs of the neck is standard of care, as supine positioning for radiographs or CT may precipitate respiratory compromise. On lateral radiographs, the epiglottis is thickened, giving the classic “thumb sign” of epiglottitis (Figure 11A).23 Other findings include thickening of the aryepiglottic folds, distention of the hypopharynx, and effacement of the valleculae, termed the “vallecula sign.” In adults, CT is typically preferred first-line imaging, as the symptoms of supraglottitis are similar to other etiologies of odynophagia, such as peritonsillar or retropharyngeal abscesses, and because CT is more sensitive and specific compared to conventional radiography.24 Imaging findings include thickening of the epiglottis, submucosal edema/inflammation within the supraglottic larynx, narrowing of the supraglottic airway, and potential submucosal rim-enhancing fluid collections (Figures 11B, 11C).25
Conclusion
Acute infections of the face and neck are frequent indications for imaging in the emergency department. Knowledge of the commonly encountered infection imaging patterns helps to identify the source and potential complications, and to permit proper clinical management.
References
- Wang B, Gao BL, Xu GP, et al. Images of deep neck space infection and the clinical significance. Acta Radiol. 2014;55(8):945-951.
- Bakir S, Tanriverdi MH, Gün R, et al. Deep neck space infections: a retrospective review of 173 cases. Am J Otolaryngol. 2012;33(1):56-63.
- Boscolo-Rizzo P, Stellin M, Muzzi E, et al. Deep neck infections: a study of 365 cases highlighting recommendations for management and treatment. Eur Arch Otorhinolaryngol. 2012;269(4):1241-1249.
- Chapman MN, Nadgir RN, Akman AS, et al. Periapical lucency around the tooth: radiologic evaluation and differential diagnosis. Radiographics. 2013;33(1):E15-32.
- Schuknecht B, Stergiou G, Graetz K. Masticator space abscess derived from odontogenic infection: imaging manifestation and pathways of extension depicted by CT and MR in 30 patients. Eur Radiol. 2008;18(9):1972-1979.
- Botha A, Jacobs F, Postma C. Retrospective analysis of etiology and comorbid diseases associated with Ludwig’s angina. Ann Maxillofac Surg. 2015;5(2):168-173.
- Nguyen VD, Potter JL, Hersh-Schick MR. Ludwig angina: an uncommon and potentially lethal neck infection. AJNR Am J Neuroradiol. 1992;13(1):215-219.
- Vestin Fredriksson M, Öhman A, Flygare L, et al. When maxillary sinusitis does not heal: Findings on CBCT scans of the sinuses with a particular focus on the occurrence of odontogenic causes of maxillary sinusitis. Laryngoscope Investig Otolaryngol. 2017;2(6):442-446.
- Bomeli SR, Branstetter BF 4th, Ferguson BJ. Frequency of a dental source for acute maxillary sinusitis. Laryngoscope. 2009;119(3):580-584.
- Huovinen P, Lahtonen R, Ziegler T, et al. Pharyngitis in adults: the presence and coexistence of viruses and bacterial organisms. Ann Intern Med. 1989;110(8):612-616.
- Capps EF, Kinsella JJ, Gupta M, et al. Emergency imaging assessment of acute, nontraumatic conditions of the head and neck. Radiographics. 2010;30(5):1335-1352.
- Schraff S, McGinn JD, Derkay CS. Peritonsillar abscess in children: a 10-year review of diagnosis and management. Int J Pediatr Otorhinolaryngol. 2001;57(3):213-218.
- Maroldi R, Farina D, Ravanelli M, et al. Emergency imaging assessment of deep neck space infections. Semin Ultrasound CT MR. 2012;33(5):432-442.
- Lemierre A. On certain septicæmias due to anaerobic organisms. Lancet. 1936; 227:701–703.
- Weeks DF, Katz DS, Saxon P, et al. Lemierre syndrome: report of five new cases and literature review. Emerg Radiol. 2010;17(4):323-328.
- Hoffmann C, Pierrot S, Contencin P, et al. Retropharyngeal infections in children. Treatment strategies and outcomes. Int J Pediatr Otorhinolaryngol. 2011;75(9):1099-1103.
- Shefelbine SE, Mancuso AA, Gajewski BJ, et al. Pediatric retropharyngeal lymphadenitis: differentiation from retrophyarngeal abscess and treatment implications. Otolaryngol Head Neck Surg. 2007;136(2):182-188.
- Debnam JM, Guha-Thakurta N. Retropharyngeal and prevertebral spaces: anatomic imaging and diagnosis. Otolaryngol Clin North Am. 2012;45(6):1293-1310.
- Hoang JK, Branstetter BF 4th, Eastwood JD, et al. Multiplanar CT and MRI of collections in the retropharyngeal space: is it an abscess? AJR Am J Roentgenol. 2011;196(4):W426-432.
- Chung T, Rebello R, Gooden EA. Retropharyngeal calcific tendinitis: case report and review of literature. Emerg Radiol. 2005;11(6):375-380.
- Eastwood JD, Hudgins PA, Malone D. Retropharyngeal effusion in acute calcific prevertebral tendinitis: diagnosis with CT and MR imaging. AJNR Am J Neuroradiol. 1998;19(9):1789-1792.
- Mayo-Smith MF, Spinale JW, Donskey CJ, et al. Acute epiglottitis. An 18-year experience in Rhode Island. Chest. 1995;108(6):1640-1647.
- Grover C. Images in clinical medicine. “Thumb sign” of epiglottitis. N Engl J Med. 2011;365(5):447.
- Tan TH, Hsu CC, Liao TT, et al. Computed tomography scan as a diagnostic tool for supraglottitis in adults. Am J Emerg Med. 2014;32(1):1279-1280.
- Smith MM, Mukherji SK, Thompson JE, et al. CT in adult supraglottitis. AJNR Am J Neuroradiol. 1996;17(7):1355-1358.
Citation
Winegar, EA N, WS K. Imaging Acute Face and Neck Infections. Appl Radiol. 2020;(3):28-33.
May 4, 2020