Genomics With Tau PET Provides a Personalized Approach for Subtyping Alzheimer Disease
Images
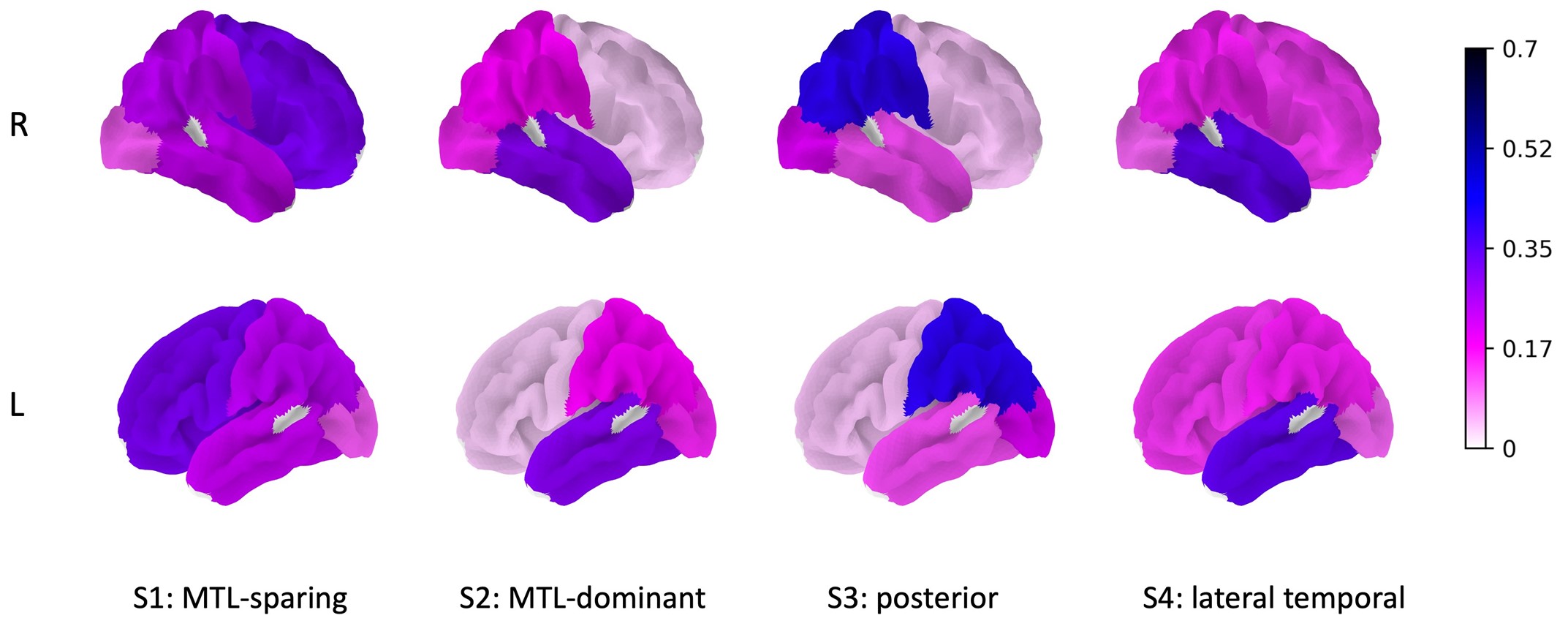
Research presented at the 2023 Society of Nuclear Medicine and Molecular Imaging Annual Meeting suggests that a new computational technique that combines genomic and tau PET imaging data promises a more personalized approach for subtyping Alzheimer disease. Based on a novel clustering framework using sparse canonical correlation analysis (SCCA), the integrated approach was successful in identifying four subtypes of Alzheimer’s disease and the top genes associated with each.
Alzheimer disease is a genetically complex disorder. Several subtypes of the disease have been identified based on PET imaging of amyloid plaques and tau neurofibrillary tangles, the pathological hallmarks of the disease. In addition, recent genome-wide association studies have identified many genes that play a role in the development of Alzheimer disease.
“By identifying different subtypes of Alzheimer disease using both imaging and genomic information, researchers could gain potential new insights into the underlying biology of the disease and its progression,” stated Joyita Dutta, PhD, associate professor in the Department of Biomedical Engineering at the University of Massachusetts in Amherst, Massachusetts. “Understanding the specific genetic associations for each subtype could also lead to the development of personalized treatment approaches in the future.”
Imaging and genomics data from the Alzheimer Disease Neuroimaging Initiative (ADNI) were utilized in the investigation. All ADNI participants who had undergone18F-flortaucipir PET and Illumina SNP genotyping were included (541 participants in total; 334 cognitively normal and 207 cognitively impaired). Tau PET standardized uptake value ratios from 10 broad regions were computed from tau PET imaging, and 145 genome variations associated with Alzheimer disease were identified and extracted from the SNP genotyping. The SCCA-clustering framework was then jointly applied to the tau PET and genomics datasets.
Four subtypes of Alzheimer disease were identified in the analysis: medial temporal lobe (MTL)-dominant, posterior, MTL-sparing, and lateral-temporal. In addition to the APOE gene, top genes that were associated with each subtype were also identified.
“Genomics- and imaging-guided individualized subtyping is vital for Alzheimer disease because different subtypes may also have distinct rates and profiles of cognitive decline, potentially affecting clinical trial outcomes and treatment response,” said Dutta. “By combining molecular imaging information with genomics, we have created a diagnostic technique that could be truly personalized for each patient. This has potential for broad diagnostic utility across many disease types, not only Alzheimer disease.”