FDA Clears Reality-Based Visualization and Navigation Platform
Images
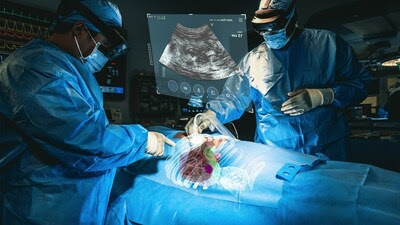
MediView XR, Inc. announced US FDA 510(k) clearance for its XR90 augmented reality-based surgical visualization and navigation platform. XR90 is intended to be used adjunctively for minimally invasive ultrasound and CT guided needle-based procedures for soft tissue and bone. "This is not only the first 510(k) clearance for MediView, but it is the first 510(k) clearance for an augmented reality device utilizing live imaging combined with 3D XR visualization for pre- and intra-operative indications for use, which sets the stage for further advancements in augmented reality in the healthcare space." said Adam Cargill, Director of Quality, Regulatory and Clinical Affairs at MediView.
MediView is utilizing augmented reality to address the long-standing limitations of current medical imaging technologies. Flat panel monitors limit practitioners to 2D imaging and require them to look away from the patient's procedural site, disrupting hand-eye-coordination, resulting in reduced clinician confidence and potentially impacting outcomes. Through Microsoft's HoloLens 2 AR headset, clinicians are able to visualize the patient's ultrasound, as well as displays of other procedural information to facilitate their workflow. XR90 overcomes the limitations of two-dimensional imaging by providing physicians with 3D "X-Ray vision" during procedures –the ability to visualize a patient's comprehensive internal anatomy in 3D underneath their skin, including bone, tissue, organs and vasculature. The device projects 3D virtual models of the patient's own anatomy based on their CT imaging and combines that CT with live ultrasound to perform minimally invasive procedures, such as biopsies and tumor ablations (the use of heat or cold to kill cancerous tumors). XR90's augmented reality capabilities include a Holographic Light Ray that tracks and displays the path of the physician's instrument, CT-based 3D holographic anatomy display, and live ultrasound that is projected and displayed anatomically into the patient as the clinician scans, similar to a flashlight beam. The system is designed to provide visual information and reference to clinicians for analysis of procedural options during pre-operative planning, to help facilitate workflow, and to provide enhanced ergonomics to the user for heads-up, intra-operative display of medical images during ultrasound- guided needle procedures. XR90 is indicated to be used adjunctively to standard of care imaging.
"MediView is excited to receive 510(k) clearance for our XR90 platform and appreciate the team's efforts and FDA's partnership in achieving this milestone. We are in a new era of visualization, collaboration, and data insights in healthcare that can create patient, clinician and financial benefits," said Mina Fahim, President and CEO of MediView. "XR90 expands the MediView portfolio of solutions available to practitioners as they look to simplify, democratize, and inform care delivery with the ultimate goal of improving and expanding access to the best care. Thank you to our employees, investors, and partners for making this possible. We are just at the beginning of a journey seeing the clinical adoption of game-changing data and digital healthcare solutions."
XR90 is a diverse imaging tool providing a new way to view patient anatomy and visualize instrumentation pathway while maintaining natural alignment of senses. Clinicians at remote locations can also collaborate real-time with shared visualization, communication, and the ability to provide guidance during procedures for collaborative patient care. These features can provide increased support for understaffed facilities, rural or underserved populations and a distinct ability to limit caregiver and patient exposure to COVID-19 while maintaining care levels.