Choledochocele
Images
Case Summary
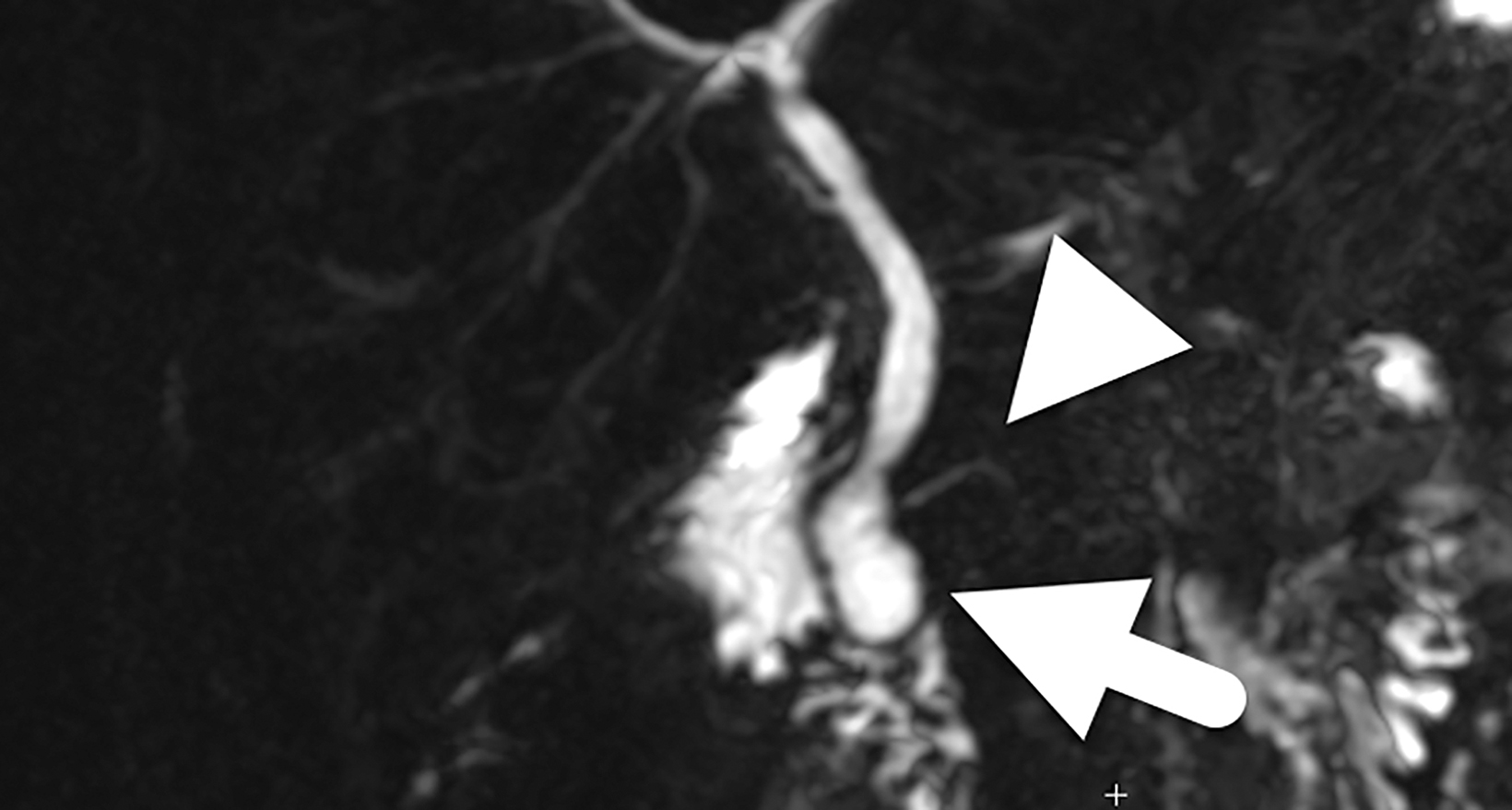
Abdominal imaging was performed to evaluate for distant disease in a child with scalp melanoma. A choledochocele was found incidentally during imaging evaluation. At that time, the child had no abdominal pain, jaundice, or hematemesis and did not exhibit signs of hepatic injury or cholestasis.
The patient was lost to follow-up for four years and presented again for re-evaluation. Magnetic resonance imaging (MRI) showed enlargement of the choledochocele. They again had no symptoms of abdominal pain, difficulty eating, or jaundice, but the child’s mother noted occasional, bright, green-colored stools. Liver function tests demonstrated mild elevation of AST and ALT but no cholestasis. The patient and her family elected for management of the choledochocele with an endoscopic retrograde cholangiopancreatography (ERCP) with endoscopic biliary sphincterotomy and biopsy of the cyst lining.
Imaging Findings
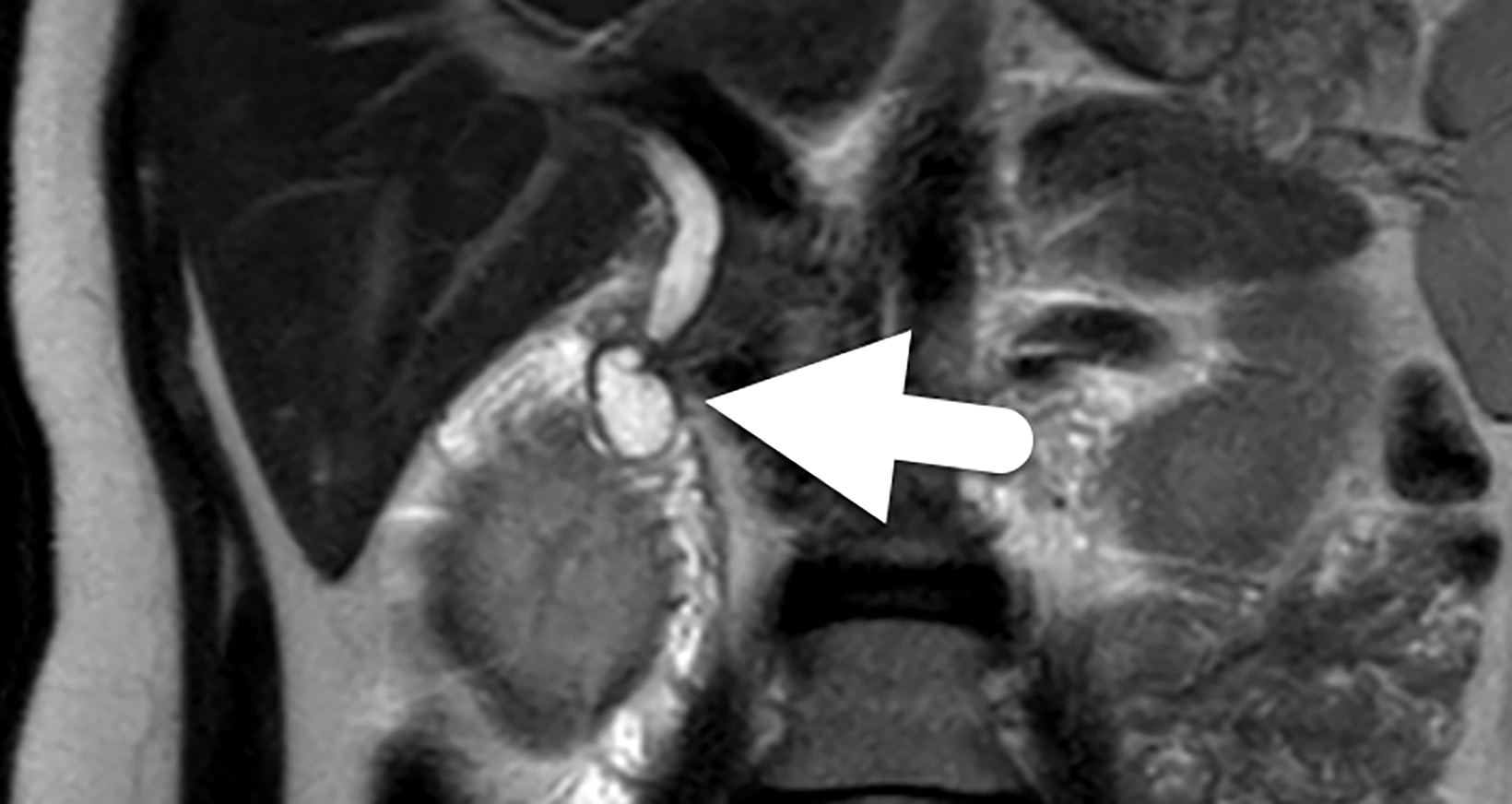
The initial MRI (Figure 1) identified a mildly enlarged common bile duct (CBD) (5 mm diameter) with mild central intrahepatic biliary ductal dilatation. The dilated CBD inserted into a bilobed cystic structure at the level of the pancreatic head. One lobe of this structure appeared partially intraluminal, extending into the second portion of the duodenum and was diagnosed as a choledochocele. The hepatobiliary phase images showed the cystic structure filling with contrast.
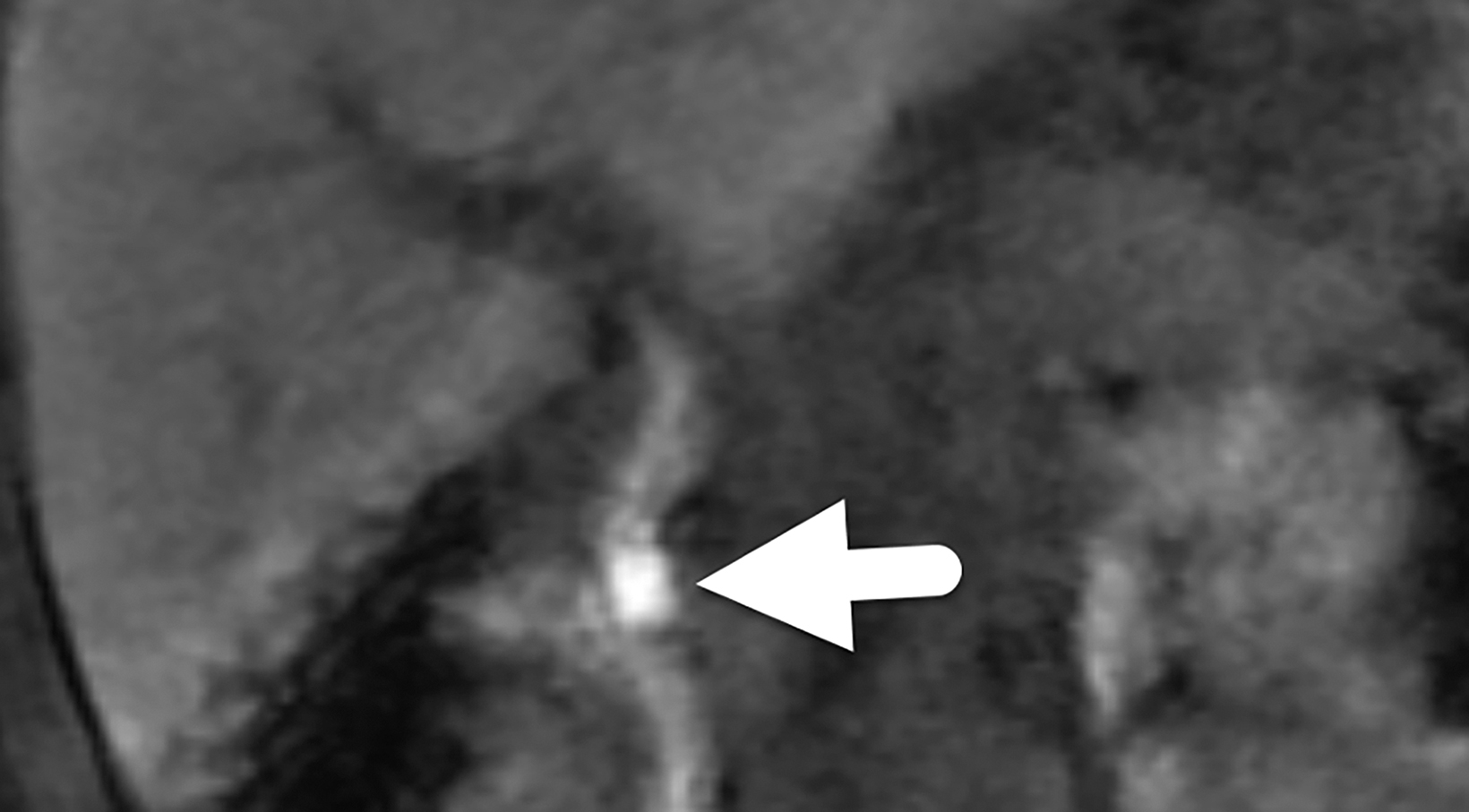
Follow-up magnetic resonance cholangiopancreatography (MRCP, Figure 2) four years later showed similar prominence of the central confluence of the hepatic ducts. The CBD remained dilated, measuring 7 mm in diameter and tapering to a point at the level of the ampulla. The choledochocele was again seen protruding into the duodenal lumen. The pancreatic duct was normal in caliber.
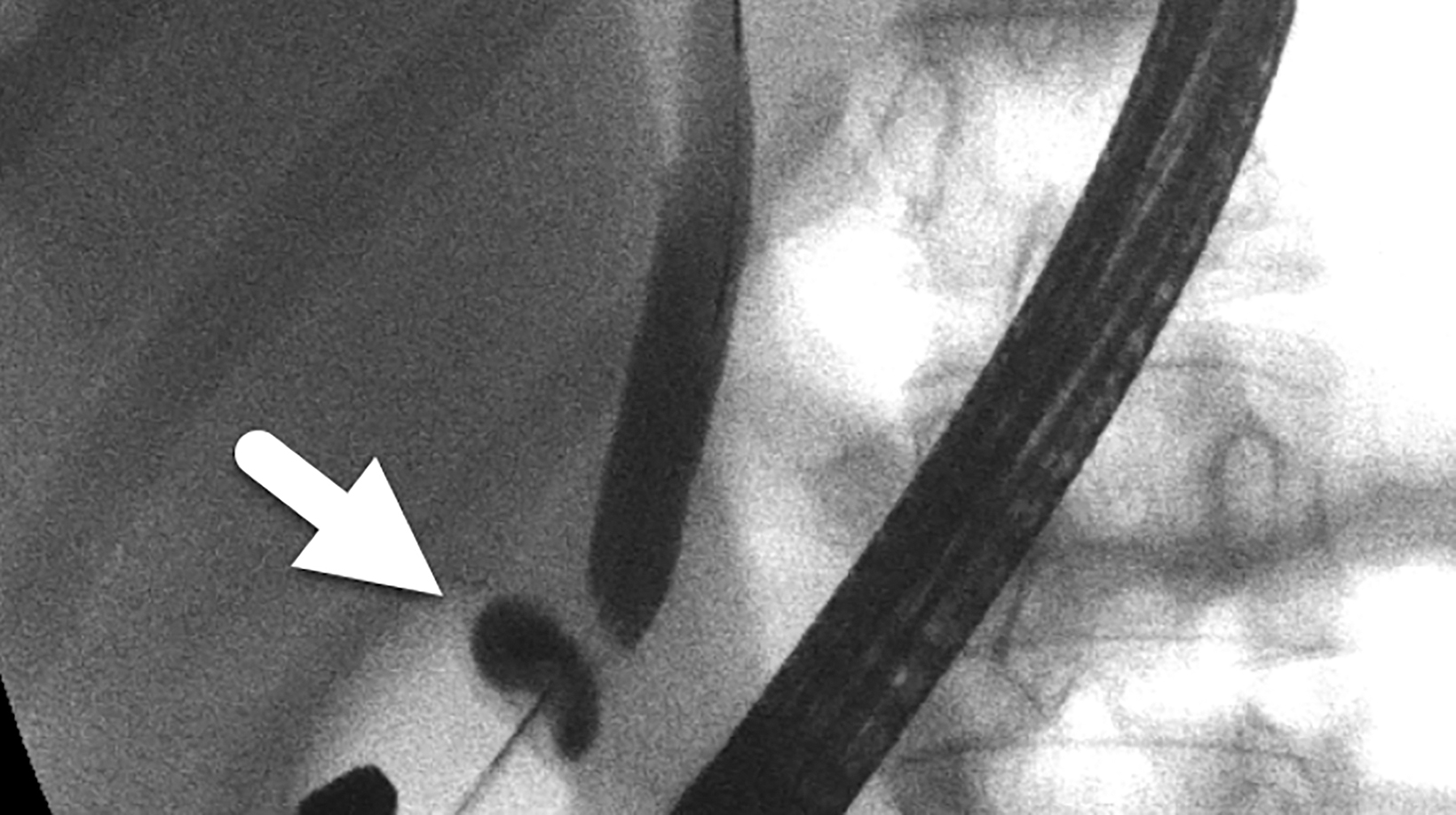
An ERCP confirmed the diagnosis of a Type III choledochal cyst — a choledochocele (Figure 3). A cholangiogram showed a dilated main bile duct with mildly dilated intrahepatic ducts. A needle knife cystostomy followed by sphincterotome extension of the cystostomy and biliary sphincterotomy was performed.
Diagnosis
Choledochocele (Type III choledochal cyst).
The differential diagnosis includes duodenal duplication cyst, other types of choledochal cysts, pancreatic pseudocyst, impacted gallstone, and biliary stricture.
Discussion
Choledochal cysts are dilations in the biliary tree. The Todani classification system defines six types of choledochal cysts, depending on the location of dilation. Type III choledochal cysts are limited to the intraduodenal portion of the CBD. These cysts are recognized as congenital anomalies and are more commonly known as choledochoceles, a term first applied in 1940, owing to their similarity to ureteroceles.2 There are two subtypes of choledochoceles, A and B.2,3 Type A choledochoceles are characterized by both the pancreatic duct and CBD opening into the cystic space, which then communicates with the duodenum via a separate orifice.3,6 Type B choledochoceles occur when a diverticulum arises from the intra-ampullary pancreaticobiliary duct or from the intraduodenal CBD. The bile and pancreatic secretions then drain into the duodenum via the ampulla of Vater.6,8 In both subtypes, the dilation can be visualized during endoscopic evaluation as an intraduodenal bulge at the level of the major duodenal papilla.
Choledochoceles are rare, making up fewer than 5% of choledochal cysts, and are more commonly diagnosed in adults than children.1,2,3,5 Choledochoceles differ from other choledochal cysts in that they affect males and females equally (other choledochal cysts have a well-established 3:1 female to male ratio).1,3,5 Patients with a prior biliary procedure are thought to have a higher risk of acquiring a choledochocele. This is supported by the literature, which reports prior cholecystectomy or CBD exploration in 44-60% of adult patients with choledochoceles.2,7 In children, these are generally considered congenital abnormalities.
Various theories have been put forth to explain the etiology of choledochal cysts. The prevailing theory is that an anomalous pancreaticobiliary junction (APBJ) leads to pancreaticobiliary reflux, which ultimately causes inflammation, cystic dilation, and an increased risk for malignancy.8 However, an APBJ is not found in all patients with choledochoceles.2 Other congenital etiologies include failed regression of a rudimentary CBD during embryological development or a congenital intramural cyst or diverticulum.1,3,5,6 Evidence supporting a congenital etiology of choledochoceles includes the presence of duodenal mucosa in nearly two-thirds of cysts.2,3 An acquired choledochocele may result from any situation that causes inflammation and chronically elevated pressure within the ampulla of Vater, with resulting obstructive intramural dilation of the CBD.1,2,3,6
Patients with a choledochocele most commonly present with abdominal pain, jaundice, nausea, vomiting, or a palpable mass.2 These entities can also be encountered incidentally on imaging performed for unrelated indications, as in this patient. In contrast to patients with other types of choledochal cysts, those with a choledochocele are more likely to present with acute pancreatitis and less likely to develop biliary tract symptoms and cholangitis.2,3,4
Ultrasound is often the first-line imaging modality for patients presenting with signs and symptoms of an acute or subacute biliary abnormality. In patients with a choledochocele, ultrasound can identify the intraduodenal cystic mass.2,3 Additionally, it can help to determine the extent of a pancreaticobiliary abnormality and the presence or absence of intraductal biliary stones. MRCP is considered the noninvasive test of choice as it provides detailed anatomic information and helps to assess size, location, and nature of the cystic mass. ERCP is considered the diagnostic gold-standard, but it is an invasive intervention with associated risk factors.2,3 Currently, the major role of ERCP is to confirm the MRCP findings and to guide cystostomy and biliary sphincterostomy.2,3
Studies report a risk of malignancy around 2.5%.1,3,4,5 In contrast, the risk of cancer with other choledochal cysts has been reported to range from 10-15%.1,2,4 It is surmised that the lower risk of malignancy is related to choledochoceles etiology. Because an APBJ is not routinely found in patients with a choledochocele, the risk of reflux and resulting cancer is thought to be decreased. An intraoperative biopsy is performed to help determine malignancy risk.
The lining of a choledochocele can be either intestinal or biliary epithelium.7 Some researchers hypothesize that cysts lined with intestinal epithelium do not carry an increased risk of malignancy, while cysts lined with biliary epithelium may carry an increased risk of developing cholangiocarcinoma.
Treatment depends on malignancy risk and size of the choledochocele.5 Endoscopic sphincterotomy and/or surgical marsupialization have been used if intestinal epithelium is present.5,7, 11 Endoscopic sphincterotomy with cystotomy is the most widely used option; the procedure relieves any biliary obstruction and allows for cyst drainage. Endoscopic sphincterotomy outcomes are favorable, with quick, postsurgical resolution of symptoms.2,3 Alternatively, transmural excision of the choledochocele is required if biliary or dysplastic epithelium is found or if the cyst is causing gastric outlet obstruction.4,5
Conclusion
Choledochoceles are rare abnormalities of the biliary tree, with an etiology that is not well understood. Imaging often detects a cystic mass in the descending duodenum, and MRCP elucidates the anatomy and confirms the diagnosis. Choledochoceles affect adults most often, with no male or female preference.
Symptomatic patients present with abdominal pain, jaundice, and/or nausea/vomiting. Treatment consists most commonly of endoscopic sphincterotomy and cystotomy. Surgical excision may be necessary if there are findings of gastric outlet obstruction or dysplasia.
References
- Singham J, Yoshida EM, Scudamore CH. Choledochal cysts: part 1 of 3: classification and pathogenesis. Can J Surg. 2009;52(5):434‐440.
- Ziegler KM, Zyromski NJ. Choledochoceles: are they choledochal cysts? Adv Surg. 2011;45:211‐224. doi:10.1016/j.yasu.2011.03.019
- Law R, Topazian M. Diagnosis and treatment of choledochoceles. Clin Gastroenterol Hepatol. 2014;12(2):196‐203. doi:10.1016/j.cgh.2013.04.037
- Soares KC, Arnaoutakis DJ, Kamel I, et al. Choledochal cysts: presentation, clinical differentiation, and management. J Am Coll Surg. 2014;219(6):1167‐1180. doi:10.1016/j.jamcollsurg.2014.04.023
- Lobeck IN, Dupree P, Falcone RA Jr, et al. The presentation and management of choledochocele (type III choledochal cyst): A 40-year systematic review of the literature. J Pediatr Surg. 2017;52(4):644‐649. doi:10.1016/j.jpedsurg.2016.10.008
- Scholz FJ, Carrera GF, Larsen CR. The choledochocele: correlation of radiological, clinical and pathological findings. Radiology. 1976;118(1):25‐28. doi:10.1148/118.1.25
- Schimpl G, Sauer H, Goriupp U, Becker H. Choledochocele: importance of histological evaluation. J Pediatr Surg. 1993;28(12):1562‐1565. doi:10.1016/0022-3468(93)90097-5
- Cha SW, Park MS, Kim KW, et al. Choledochal cyst and anomalous pancreaticobiliary ductal union in adults: radiological spectrum and complications [published correc
- tion appears in J Comput Assist Tomogr. 2008 Sep-Oct;32(5):827]. J Comput Assist Tomogr. 2008;32(1):17‐22. doi:10.1097/RCT.0b013e318064e723
- Kamisawa T, Tu Y, Nakajima H, Egawa N, Tsuruta K, Okamoto A. The presence of a common channel and associated pancreaticobiliary diseases: a prospective ERCP study. Dig Liver Dis. 2007;39(2):173‐179. doi:10.1016/j.dld.2006.09.020
- Matsushita M, Uchida K, Nishio A, Okazaki K. Differential diagnosis of intraduodenal cystic lesions: choledochocele, duodenal duplication cyst, or intraluminal duodenal diverticulum? Gastrointest Endosc. 2010;71(1):219‐220. doi:10.1016/j.gie.2009.04.032
- Lobeck IN, Dupree P, Falcone RA Jr, Lin TK, Trout AT, Nathan JD, Tiao GM. The presentation and management of choledochocele (type III choledochal cyst): A 40-year systematic review of the literature. J Pediatr Surg. 2017;52(4):644-649. doi:10.1016/j.jpedsurg.2016.10.008
References
Citation
AR S, RB T, Y L, DS V, AJ T. Choledochocele. Appl Radiol. 2021;(6):4-7.
November 5, 2021