Aneurysmal Bone Cyst
Images
Case Summary
A teenager presented with a history of an enlarging, painful mass in his right lower leg and increasing pain with ambulation during the past few months. On physical examination, there was tenderness to palpation of the right lateral lower leg.
Imaging Findings
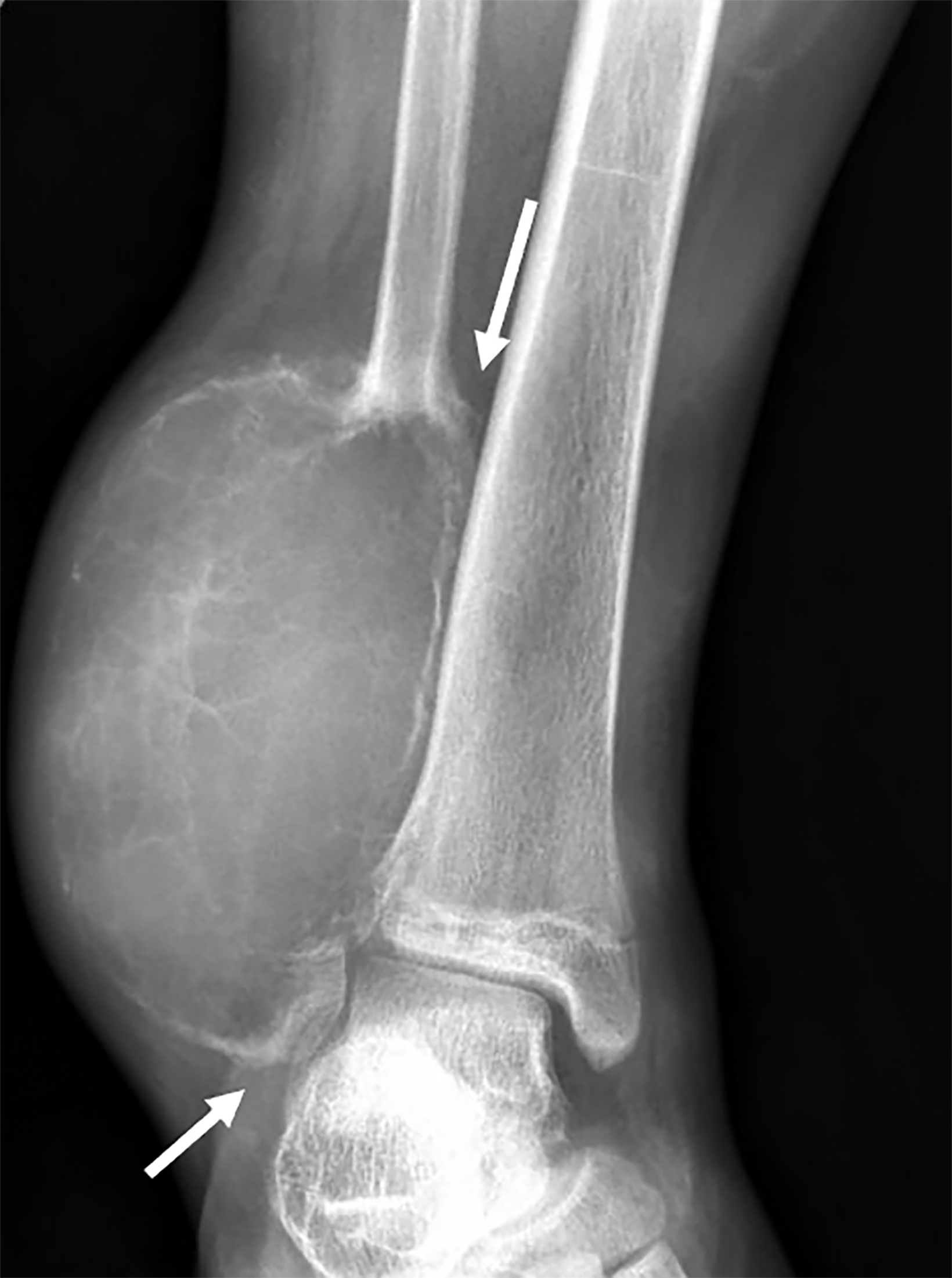
Radiography (Figure 1) demonstrated a large, expansile mass in the distal fibula with internal septations. There was extensive destruction of the distal diaphysis, metaphysis, physis, and epiphysis with marked cortical thinning and periosteal reaction at both the proximal and distal aspects of the mass.
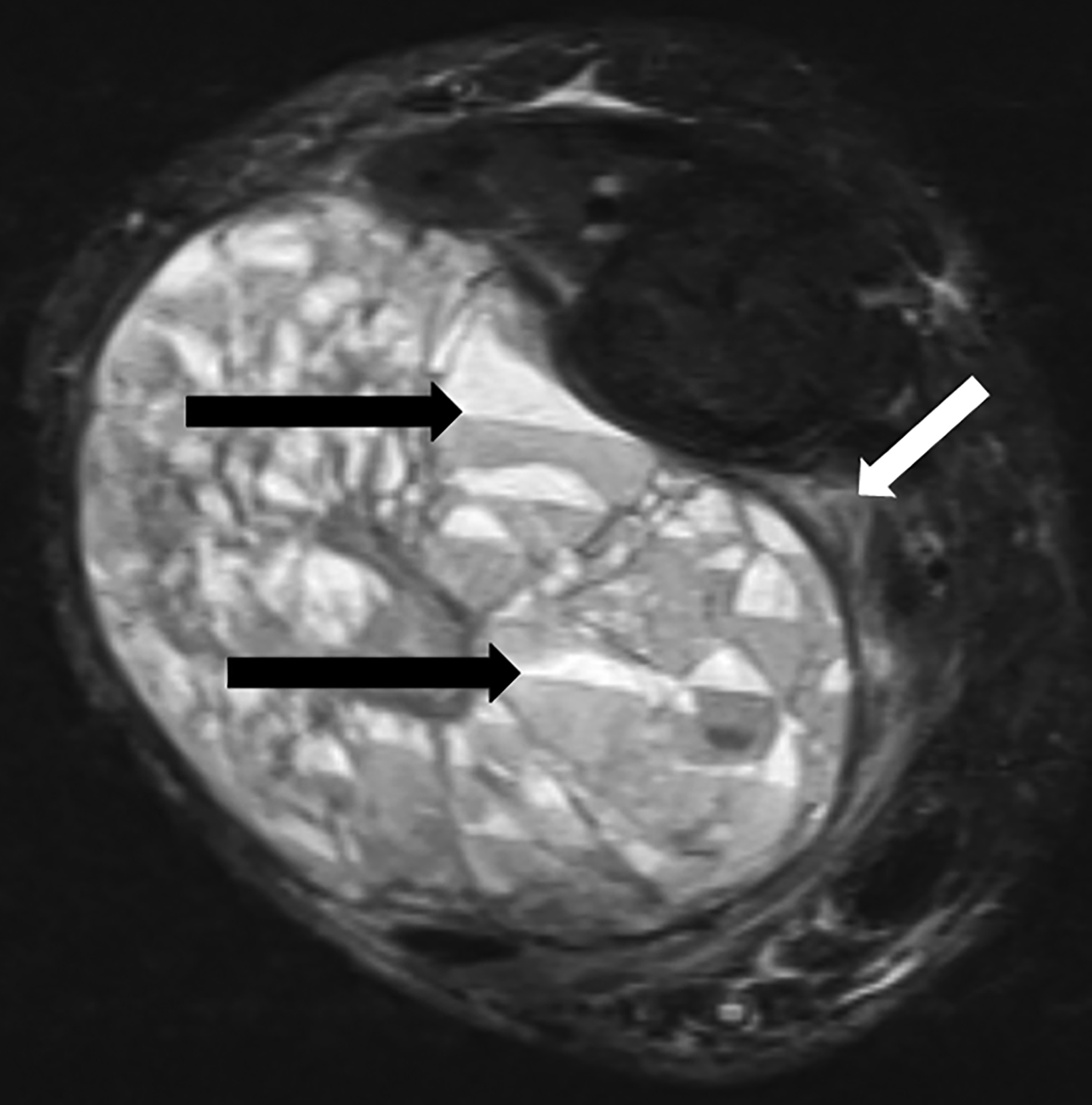
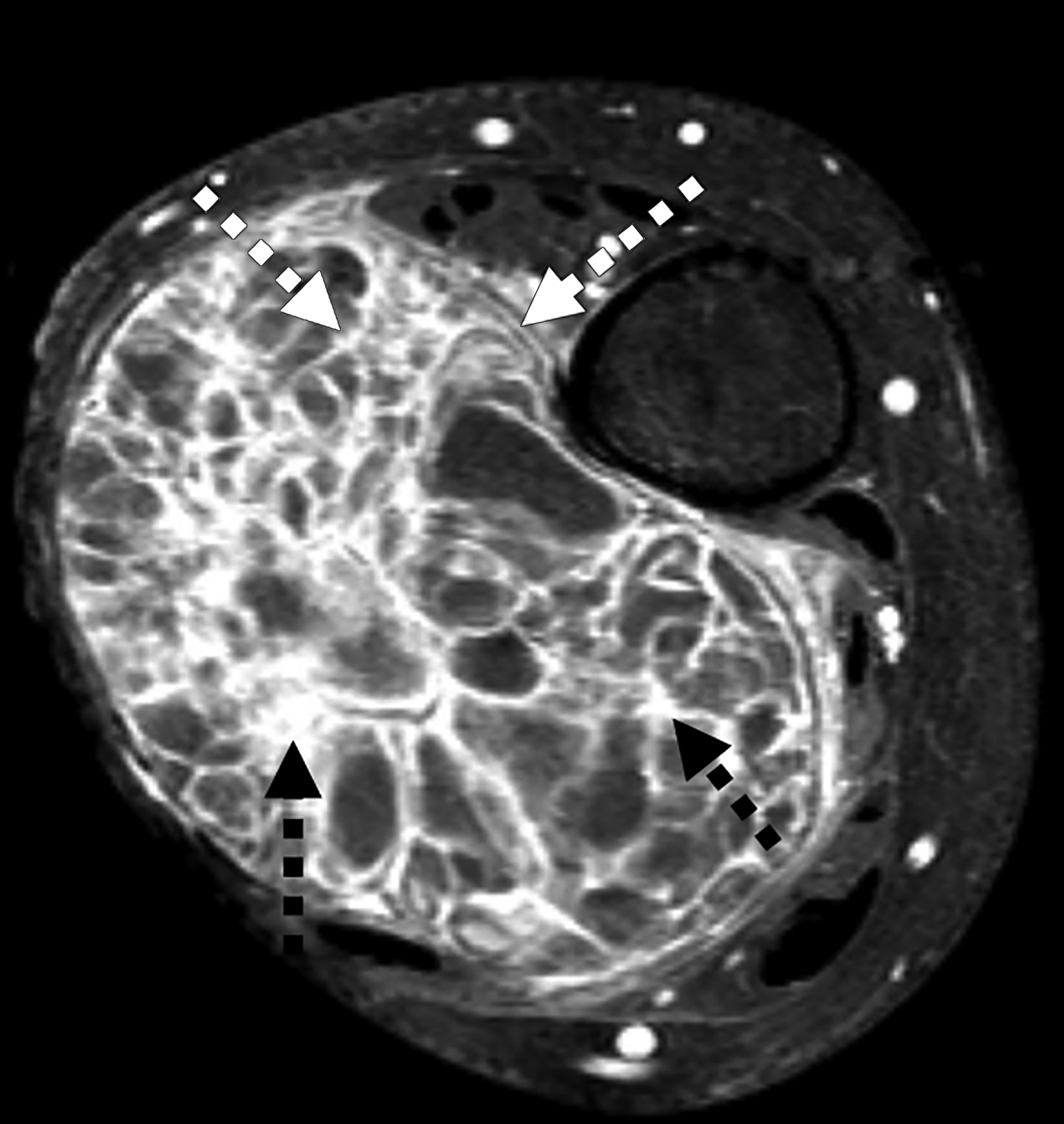
MRI (Figure 2) redemonstrated an expansile and markedly septated mass containing fluid-fluid levels with surrounding soft-tissue edema. Post-gadolinium imaging showed extensive peripheral and linear septal enhancement with scattered areas of more nodular septal enhancement.
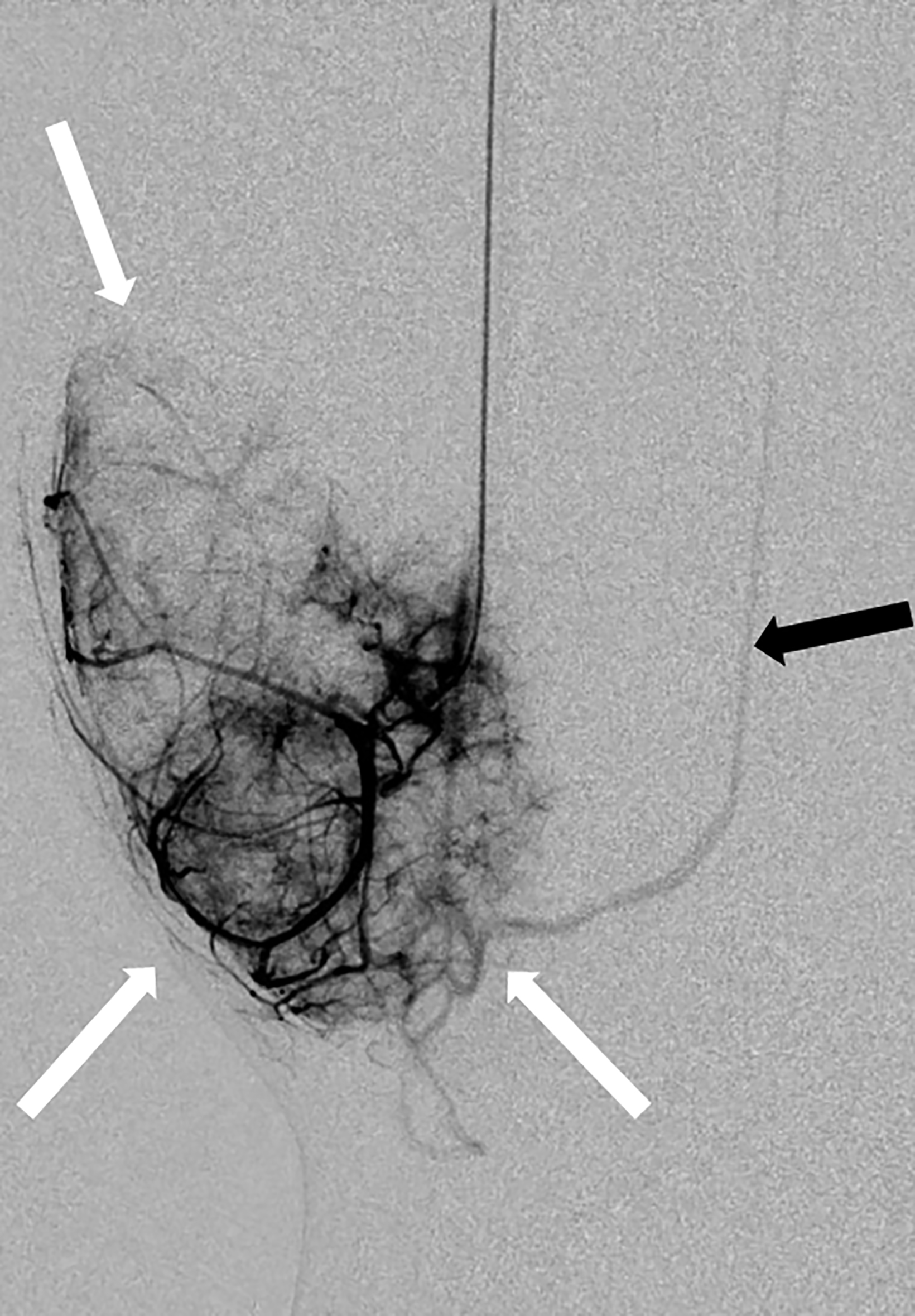
Catheter angiography performed during preoperative embolization (Figure 3) showed the mass to be hypervascular, primarily supplied by the peroneal artery, with feeding branches from the anterior tibial and posterior tibial arteries.
Diagnosis
Aneurysmal bone cyst.
Differential diagnosis includes telangiectatic osteosarcoma, chondroblastoma, giant cell tumor, metastatic disease, and multiple myeloma.
Discussion
Aneurysmal bone cysts (ABCs) are rare, highly vascularized, expansile benign lytic lesions. They most frequently occur at the metaphysis of the long bones within the first two decades of life and constitute 1-2% of primary bone tumors.1 Once believed to be non-neoplastic reactive lesions, ABCs have recently been shown to have gene rearrangements, and 70% are considered true neoplasms.2 They can also be associated with preexisting bony lesions, as they share characteristics such as histology and location but without underlying genetic mutations.3 ABCs can thus be considered either primary neoplasms or secondary to a preexisting bone tumor. Radiography demonstrates an expansile, septated, lucent lesion with cortical thinning. They can have irregular margins with periosteal reaction, mimicking more aggressive malignant lesions.4 Cross-sectional imaging with CT or MRI reveals internal fluid-fluid levels, along with improved visualization of septations and cortex.
Magnetic resonance imaging plays a uniquely important role, as the modality can depict the involvement of adjacent soft tissues, presence of solid components, and extent of surrounding edema, which is frequently underestimated on radiographs and CT scans. The presence of enhancing solid components on MRI raise concern for an associated malignant lesion; however, solid variants of ABC have been described.5
Triple-phase bone scan can be helpful in cases of suspected malignancy to evaluate for osteoblastic activity outside the primary site. Otherwise, for primary ABC, a bone scan will show nonspecific increased radiotracer activity, which reflects osteoblastic activity, hyperemia, and/or new bone formation.6
Catheter angiography can be useful vascular mapping and potentially embolization. Overall, imaging plays an important role evaluating expansile lytic lesions like ABCs. However, biopsy and histologic evaluation are required for definitive diagnosis.
Treatment depends on lesion size and region of involvement. Options include curettage with or without bone graft and en bloc resection. Curettage is the standard of care, but it has a variable recurrence rate.7 En bloc resection is associated with lower rates of recurrence but also with higher morbidity; thus, it is primarily reserved for cases where it will not compromise function.7 Since ABCs are highly vascular structures, preoperative selective arterial embolization with catheter angiography can minimize the risk of intraoperative hemorrhaging of larger lesions, as well as serve as an alternative treatment for those that are difficult to access surgically.8
The differential diagnosis for ABC includes telangiectatic osteosarcoma (TOS), chondroblastoma, giant cell tumor, metastatic disease, and multiple myeloma. As a rare, aggressive malignant tumor, TOS tends to show thickened, nodular internal septations with wide zones of transition. They frequently infiltrate adjacent soft tissue and contain solid enhancing components on MRI.9
Chondroblastomas are benign lesions that usually do not feature a soft-tissue component and often present with intact cortical bone. They typically arise from the epiphysis or apophysis of long bones in young patients. Giant cell tumors are benign bony neoplasms that occur at the epiphysis of long bones after growth plate closure. These lesions can show locally aggressive features such as wide zones of transition, cortical thinning, and soft-tissue extension.10 Despite being considered benign, they can metastasize, most often to the lungs.10 Metastatic disease and multiple myeloma commonly present with multiple lytic lesions that can be expansile. These should be included in the differential diagnosis for patients over age 40, with the most common expansile metastatic lesions arising from thyroid carcinoma and renal cell carcinoma.
Conclusion
Aneurysmal bone cysts are rare, hypervascular, expansile, benign lytic lesions often presenting within the first two decades of life. The differential diagnosis includes telangiectatic osteosarcomas, giant cell tumors, chondroblastomas, metastatic disease, and multiple myeloma. Prognosis and management differ among these lesions, and their imaging features often overlap. Imaging plays an important role in assessing their size, location, aggressiveness, degree of soft-tissue extension, and presence of solid components to help guide optimal management.
References
Citation
R W, K Y, S N, G S.Aneurysmal Bone Cyst. Appl Radiol. 2023; (3):43-44.
May 5, 2023