Acute Acalculous Cholecystitis
Images
Case Summary
A child with acute myelogenous leukemia underwent a bone marrow transplantation that was complicated by graft versus host disease and progressively worsening abdominal pain and distention.
Imaging Findings
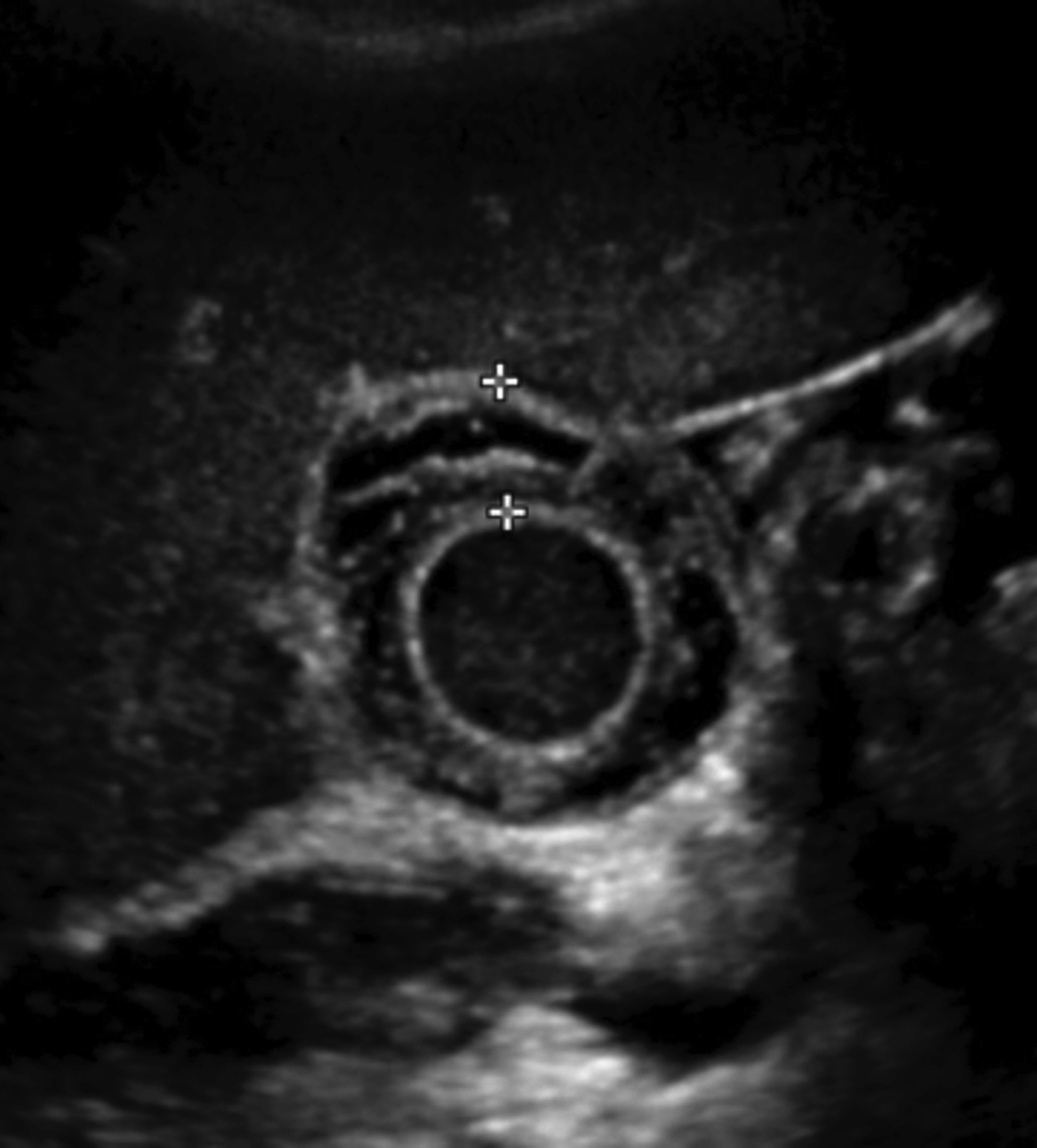
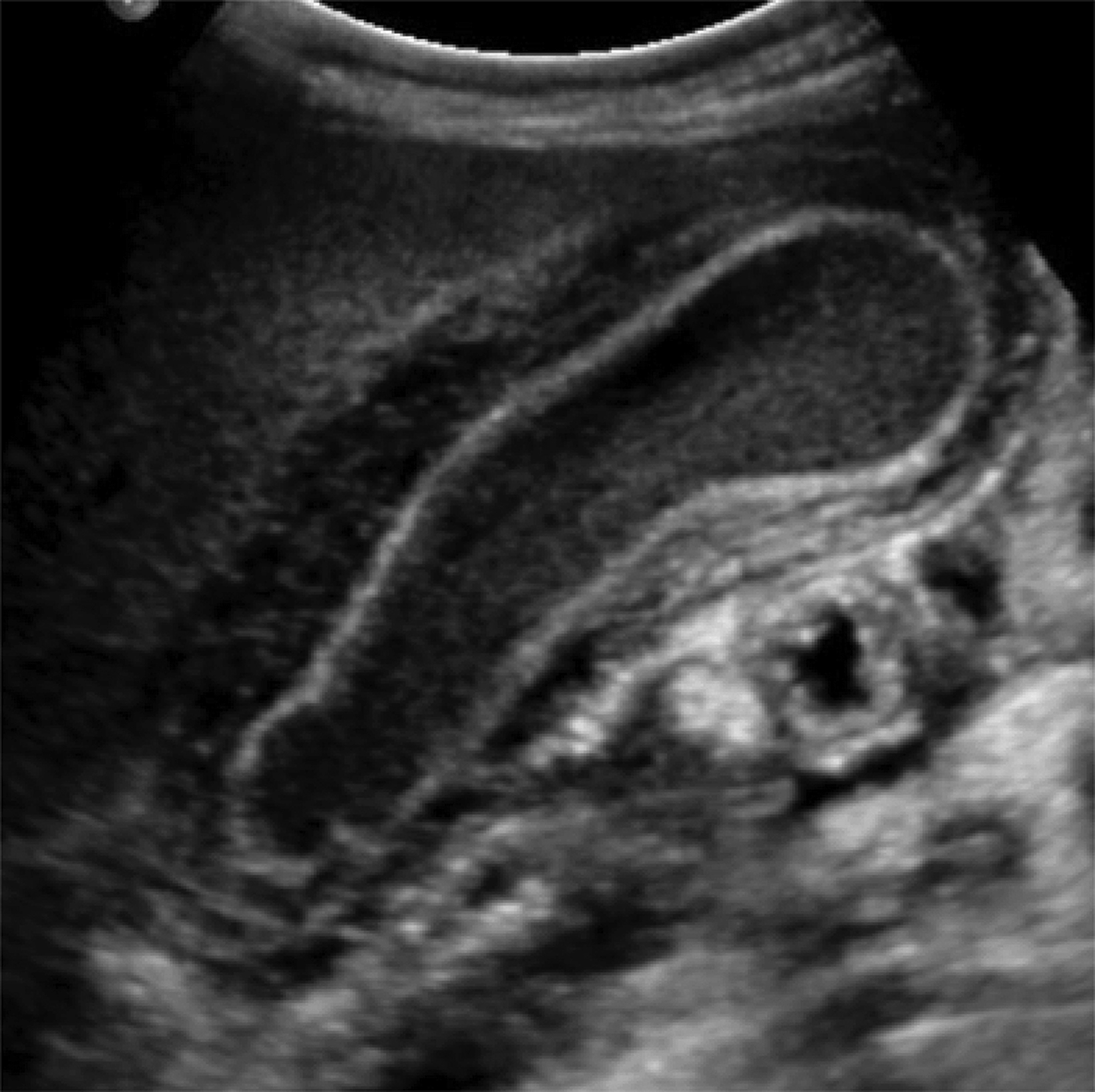
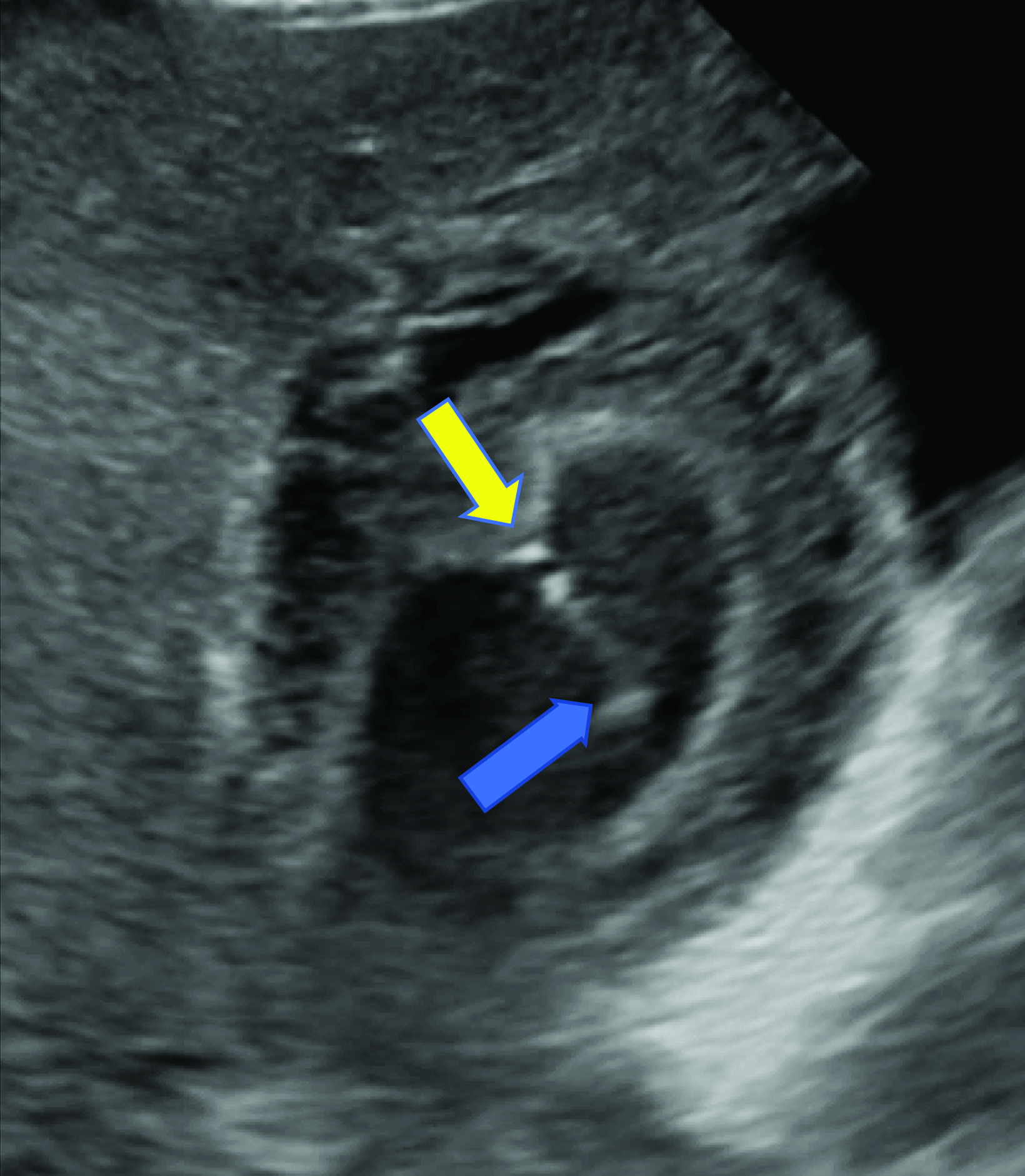
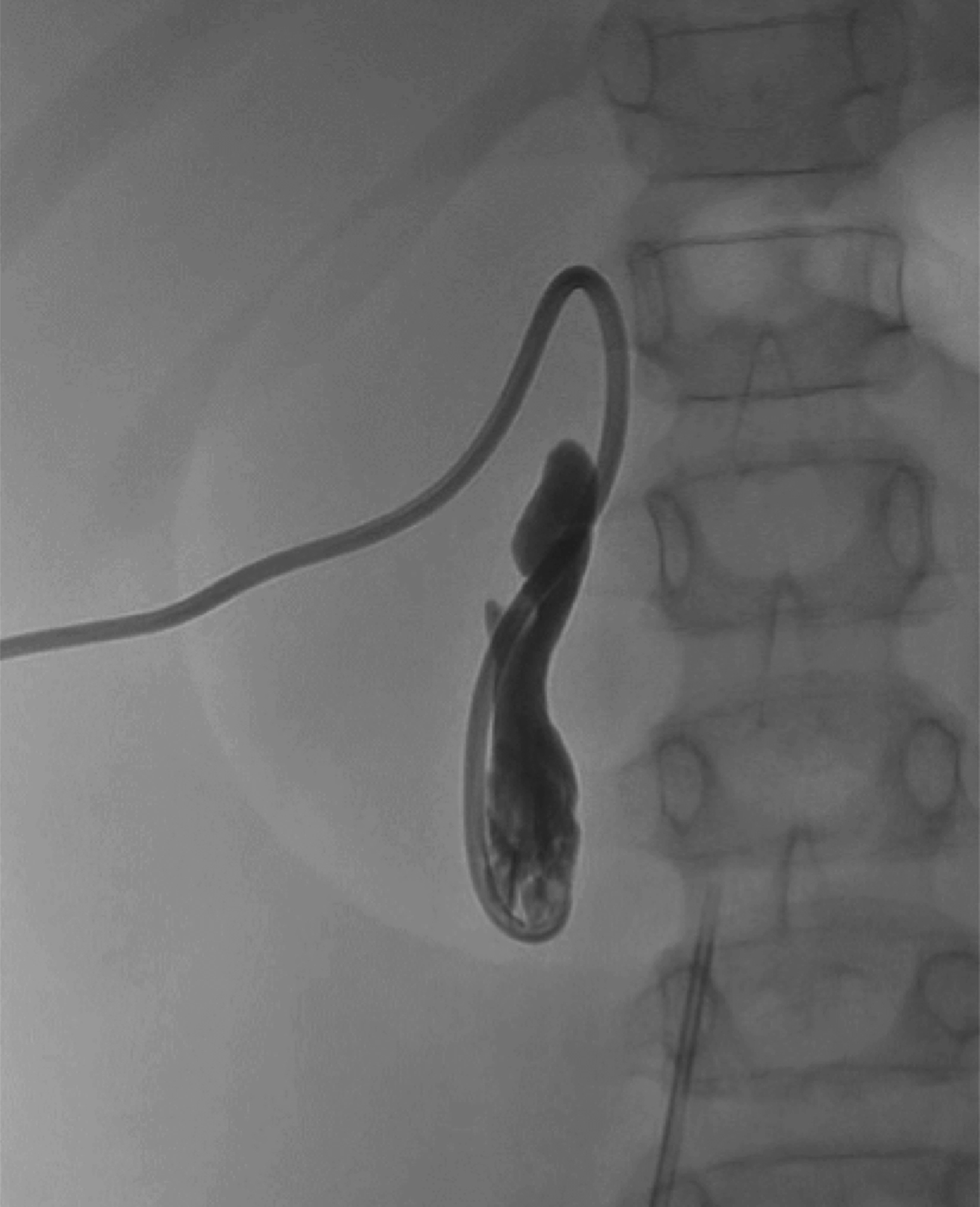
An ultrasound of the gallbladder (GB) demonstrated a sludge-filled and distended GB with marked wall thickness, but no significant pericholecystic fluid (Figure 1). The child was too unstable for surgical cholecystectomy so a percutaneous cholecystostomy (PC) was performed (Figure 2).
Diagnosis
Acute acalculous cholecystitis.
The differential diagnosis includes acute calculous cholecystitis, acute pancreatitis, hepatic abscess, or right sided pyelonephritis.1
Discussion
Acute acalculous cholecystitis (AAC) may be suspected in pediatric patients presenting with abdominal pain, signs of sepsis without a clear source, or with jaundice. The condition is the most common form of acute cholecystitis in children, representing approximately 50-70% of all cases. Pediatric patients most commonly present with right upper quadrant or diffuse abdominal pain. Other symptoms and signs such as nausea, vomiting, fever, jaundice, anorexia, and weight loss may be present. Laboratory findings include leukocytosis as well as elevated bilirubin, alkaline phosphatase, aspartate aminotransferase, alanine aminotransferase, and γ-glutamyl transferase levels.2
Pediatric AAC is typically caused by bile stasis and/or GB ischemia. It most often occurs after a viral infection such as Epstein-Barr and hepatitis A.3 Other associations include burns, sepsis, immune compromise, immune-mediated disorders (eg, systemic lupus erythematosus), vasculitis (eg, Kawasaki disease or Henoch-Schoenlein purpura), malignancy, and genetic disorders (eg, cystic fibrosis or type 1 diabetes mellitus).4
Ultrasound is the preferred imaging modality for initial assessment owing to its greater availability, short examination time, and lack of ionizing radiation. Prior studies have shown that ultrasound has equal sensitivity and superior specificity compared to CT, MRI, and hepatobiliary iminodiacetic acid (HIDA) scanning.5 In the absence of gallstones, at least two of the following findings are required to diagnose cholecystitis: GB wall edema, increased wall thickness (>3.5-4 mm), pericholecystic fluid, intraluminal sludge, or GB distention.6 A sonographic Murphy sign, defined as maximal abdominal tenderness from pressure of the ultrasound probe over the GB, may also be present.
Contrast-enhanced CT of the abdomen and pelvis, MRI, MR cholangiopancreatography (MRCP), or HIDA scan are alternative diagnostic options. On computed tomography, AAC appears with increased GB wall thickness >3-4 mm, pericholecystic fluid, subserosal edema, intramural gas, GB sludge, and hydrops.7 Unfortunately, CT is not always sensitive to gallstones. Thus, excluding AAC from the differential diagnosis is difficult. MRI and MRCP findings are similar to those of CT. MRI is more sensitive than CT for cholelithiasis or choledocholithiasis; however, MRI takes longer and often requires sedation. This often precludes its use in critically ill, unstable patients.
AAC is diagnosed on HIDA when the GB is not visualized 1 hour postinjection with radiolabeled technetium. Imaging can be augmented with intravenous morphine. Morphine causes the sphincter of Oddi to contract and helps the GB to fill; AAC is diagnosed if the GB is not visualized after 30 minutes.
AAC is often initially treated with nasogastric suctioning, intravenous fluids, and parenteral antibiotics. Although a cholecystectomy may be performed, PC is preferred in critically ill patients who are unlikely to tolerate surgery. In addition, the GB may be salvaged. A PC is less invasive and effectively drains the GB, leading to improvement in clinical status and symptoms. If necessary, open cholecystectomy can be performed electively in patients who are not critically ill.8
Conclusion
AAC most commonly presents in the setting of viral infection but may also be seen in immunocompromised children and those with vasculitis and autoimmune and genetic diseases. The most common presenting symptom is abdominal pain, with other signs and symptoms including nausea, vomiting, jaundice, fever, and anorexia. Laboratory results supporting a diagnosis of AAC include leukocytosis and elevation of markers suggestive of cholestasis and hepatic dysfunction. Ultrasound is the preferred initial imaging modality and often reveals GB wall edema, increased wall thickness (>3.5-4 mm), pericholecystic fluid, mucosal membrane sludge, and GB distention.
Percutaneous cholecystostomy is the preferred treatment for critically ill patients who are unlikely to tolerate a cholecystectomy.
References
Citation
PC C, RB T, CM S, AJ T. Acute Acalculous Cholecystitis. Appl Radiol. 2023;(6):36-38.
November 3, 2023