Acquired Pericardial Pathologies: Imaging Features, Clinical Significance, and Management
Images
This article is accredited for one SA-CME credit. Visit appliedradiology.org/SAM2 for full SA-CME information.
Rapid and effective diagnosis of potentially life-threatening pericardial diseases remains a challenge due to their similarities of symptoms and signs, and complexity of their imaging findings.1 Imaging has some clinical value, but it also has important limitations, underscoring the importance of familiarity for radiologists.2 Understanding the management of pericardial abnormalities also helps radiologists to identify and convey important findings to referring clinicians.3 This article reviews common acquired pericardial diseases, with a particular focus on clinical and hemodynamic significance, imaging findings, and management of more commonly encountered pathologies.
Simple Pericardial Effusion
The pericardial space normally contains 15 to 50 mL of serous fluid acting as a lubricant between the visceral and parietal layers of the pericardium.4 Pericardial effusion results from a pathologic accumulation of fluid, which expands this potential space.5 Although most cases remain idiopathic, common causes are uremia, infections, connective tissue disorders like systemic lupus erythematosus (SLE), acute myocardial infarction, and cardiac intervention.5-7
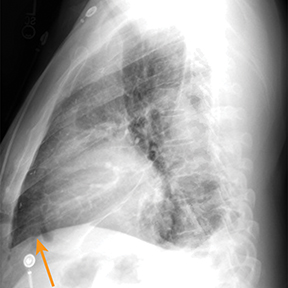
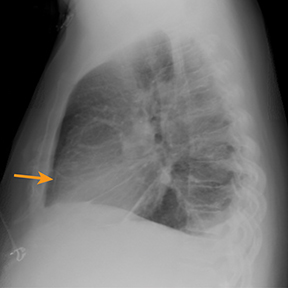
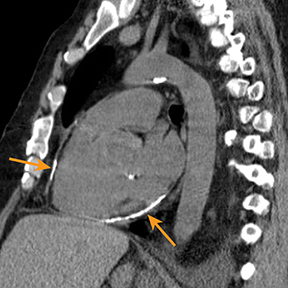
Chest radiography gives limited clues to the cause of pericardial effusion, depicting only an enlarged cardiac silhouette that may represent cardiomegaly or pericardial effusion.5 A large pericardial effusion can simulate a water bottle shape (“water bottle sign”) on frontal radiography.8 Furthermore, the “Oreo cookie sign” or “pericardial fat pad sign” (Figure 1) on lateral radiographs can indicate a pericardial effusion.9
Echocardiography represents a noninvasive, standard method for evaluating pericardial effusion.5 However, in addition to its innate limitation in field of view, the acoustic window may be further reduced by severe pulmonary emphysema or obesity.10 On echocardiography, pericardial fluid appears as a relatively echo-free space interposed between the epicardium and the pericardium.11
Computed tomography (CT) and magnetic resonance imaging (MRI) have several advantages over echocardiography, including anatomic assessment of a greater portion of the thorax with the potential to detect associated abnormalities.5 Furthermore, CT and MRI are far less operator dependent.5 MRI (as well as retrospective electrocardiography gated CT) may provide valuable information regarding cardiac function (Figure 1).5 CT attenuation of pericardial effusion can vary based on the transudative or exudative composition of the effusion, with higher attenuation in exudative pericardial effusions. However, there can be considerable overlap between the attenuation of exudative and transudative effusions.
Pericardial effusion management focuses on treatment of underlying etiologies; however, pericardiocentesis and pericardiectomy may be indicated in symptomatic cases with rapid or large fluid accumulation.5 Prognosis depends on the underlying etiology; patients with neoplastic pericardial effusion generally have a poor prognosis, while those with idiopathic/viral pericarditis usually have a very good prognosis.5
Pericardial Tamponade
Pericardial tamponade is a life-threatening mechanical condition occurring when pericardial effusion increases pericardial pressure; thus, impairing diastolic filling of the heart.12 This insufficiency leads to a decline in cardiac output and blood pressure.13,14 Presentation may be either acute (with chest discomfort, dyspnea, and hypotension) or subacute (asymptomatic initially with gradual progression of symptoms).12,13 A variety of pathologic conditions may result in pericardial tamponade, including infection, inflammatory processes, malignancy, and trauma.12,14
Echocardiography is considered a sensitive and rapid diagnostic tool.15 An increase in the right ventricle (RV) dimension and a decrease in the left ventricle (LV) dimension on inspiration is suggestive of pericardial tamponade.11,16 Diastolic collapse of the compliant RV signifies that pericardial pressure exceeds early diastolic RV pressure.11,14 Detection of RA collapse is 100% sensitive, but less specific, for tamponade.11,16 LV collapse is less common, owing to its wall thickness.11,16 Typical findings of pericardial tamponade on CT and MRI include flattening or inversion of the right atrial or RV wall with compression of these chambers, inversion of the interventricular septum, distention of the superior and inferior vena cavae, and reflux of contrast into the azygos vein and inferior vena cava.14 These findings are better appreciated on electrocardiographically gated cardiac studies.
The pericardial space in pericardial tamponade often requires urgent decompression.13 Pericardiocentesis is performed in cases of acute cardiac decompensation;17 the underlying cause should also be treated.18 Pericardiocentesis may be performed by surgically placing a drain in the subxiphoid space.13 Ultrasound-guided, percutaneous drainage with needle or catheter increases pericardiocentesis safety up to 20%.13 Radiologists who perform thoracic procedures should be familiar with CT-guided pericardial drain placement and imaging findings suggestive of pericardial tamponade.14
Pericarditis
Pericarditis is an inflammation of the pericardium.14 Its incidence is 0.1% in hospitalized patients and as high as 5% in emergency department patients with chest pain.19 A variety of conditions may cause primary and secondary pericarditis.14 Primary pericarditis presents as an isolated disorder of the pericardium, commonly with atypical chest pain (classically relieved by leaning forward) and variable constitutional symptoms.20 Although pericarditis is most often idiopathic, several secondary etiologies have been identified, including iinfection, acute myocardial infarction, drug toxicity/reaction, radiation therapy, cardiac surgery, and systemic inflammatory conditions such as rheumatoid arthritis.14,19,21 Patients presenting with acute pericarditis may describe recent symptoms suggestive of viral upper respiratory tract infection.19
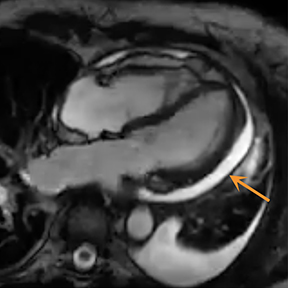
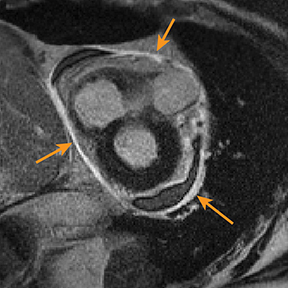
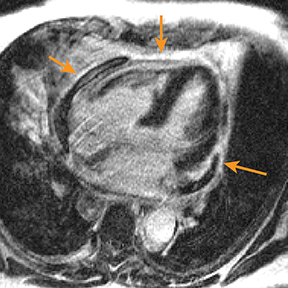
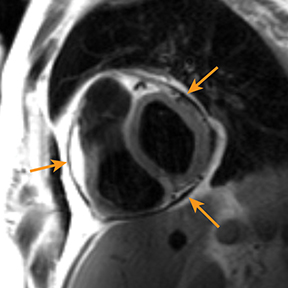
Radiography in the setting of acute pericarditis is often devoid of specific findings.19 Echocardiography, the modality of choice, demonstrates a characteristically thickened pericardium.14 CT and MRI can improve visualization and further characterization, particularly following contrast administration.14,22 Pericardial layers in acute cases show thickening and enhancement on CT, whereas irregularity or nodular thickening of the pericardium, with or without calcification, are highly suggestive of chronic pericarditis.14,22 MRI may show similar findings on T1 postcontrast and bright-blood sequences.14,23 T2 spin echo MRI demonstrates increased signal of the edematous pericardial tissues, attributable to increased fluid signal.14,23 The addition of pulse sequences such as short tau inversion-recovery (STIR) further improves pericardium evaluation by ing the signal from adjacent epicardial and pericardial fat, which also demonstrate high signal and may obscure the pericardium.14,23 Contrast-enhanced MRI is helpful in evaluating pericardial inflammation by demonstrating linear enhancement of the pericardial layers resulting from active inflammation (Figure 2).14,23
Most cases of acute pericarditis remit spontaneously.24 Treatment of noninfectious pericarditis largely involves systemic anti-inflammatory medications.24 Non-steroidal anti-inflammatory drugs are considered the mainstay of treatment for uncomplicated presentations.21 Glucocorticoids and colchicine may also be considered for those presenting with more severe and refractory cases.24,25
Pericardial Hematoma
Blood may accumulate between the visceral and parietal layers of the pericardium may occur following trauma; interventions include cardiac surgery and pericardiocentesis, due to neoplastic involvement and aortic dissection, with the latter having a dismal outcome.23,26 Clinical presentation typically depends on the cardiac structure being compressed by the hematoma.26 Although hemodynamic status may be preserved, acute pericardial hematoma may cause pericardial tamponade.15
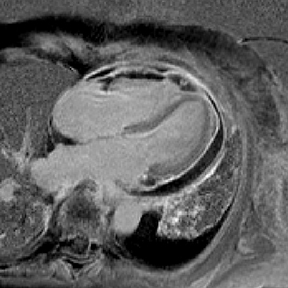
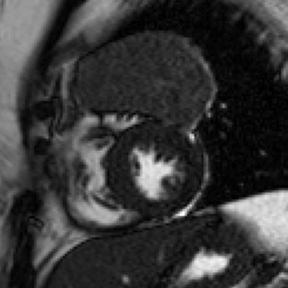
Although echocardiography provides limited tissue characterization, the modality can provide functional cardiac assessment.27,28 CT, showing blood product attenuation of the pericardial fluid, is sensitive but not specific (Figure 3).4 MRI provides ideal tissue characterization (Figure 3); the signal of the hematoma depends on the age of collection: it is homogeneously high on T2 and intermediate on T1 sequences in the acute phase; heterogeneously high on T1 and T2 sequences in the subacute phase; and low on T1 and T2 sequences with a dark rim in the chronic phase.23,29 No enhancement is expected on either CT or MR imaging.4,23
The management of pericardial hematoma involves either placing a percutaneous pericardial drain or emergent surgical drainage.26
Constrictive Pericarditis
Constrictive pericarditis should be considered when diastolic ventricular filling is impaired in the setting of thickened fibrotic pericardium.30 Although the condition is most often idiopathic or viral, a suspected inciting factor such as radiotherapy, intervention, bacterial or tuberculous infection, and metabolic disturbance (uremia) may be identified.31 Pericardial thickness greater than 4 mm is the hallmark of constrictive pericarditis. However, thicknesses less than 4 mm also can been seen in constrictive pericarditis.14 The extent of thickness has no direct relationship with the level of hemodynamic compromise.4 Constrictive pericarditis presents as potentially reversible heart failure with symptoms and signs including fatigue, orthopnea, and tachycardia.23,32 Differentiating constrictive pericarditis from restrictive cardiac pathologies is often challenging, owing to similarities in clinical presentation and echocardiography findings. Cardiac MRI can help to exclude restrictive cardiomyopathy diseases.4
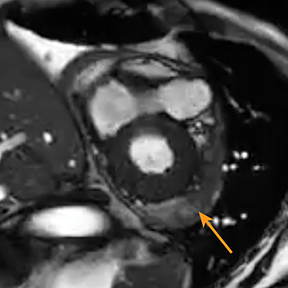
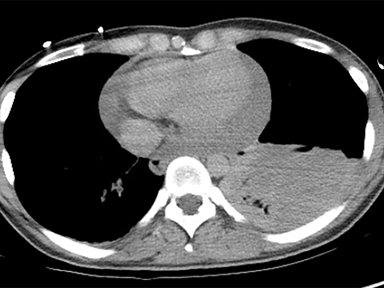
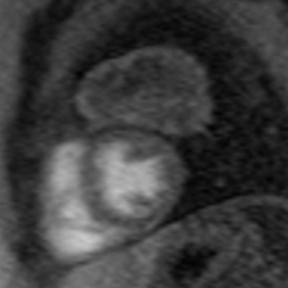
Although constrictive pericarditis is not directly visible on radiography, several indirect findings, such as pulmonary edema and pericardial calcifications, are suggestive (Figure 4).14 Echocardiography often serves as the first dedicated cardiac imaging modality.32 The pericardium typically appears abnormally thickened, potentially with regions of artifact due to calcification.32 Doppler echocardiography demonstrates signs of restrictive pathology such as impaired diastolic filling without augmentation of central venous return on inspiration.32,33 “Septal bounce,” a paradoxical bouncing motion of the interventricular septum on echocardiography or MRI is a hallmark of constrictive pericarditis and is highly specific and relatively sensitive.34 The thickened fibrotic and/or calcified pericardium has low signal intensity on T1 and T2 spin-echo MR images and at steady-state free precession imaging (Figure 4).14,23 CT is very sensitive to minimal amounts of pericardial calcification and thickness severity before surgery (Figure 4).14 Moreover, other abnormalities resulting from increased cardiac filling pressures, such as atrial enlargement, dilatation of caval and hepatic veins, pleural effusion, and ascites may be easily recognized on CT.4,14 Whereas pericardial enhancement suggests inflammation, no enhancement may appear in end-stage chronic pericardial fibrosing.14
Some cases with transient constrictive pericarditis may respond to medical therapy, but severely affected patients may be candidates for partial or total pericardiectomy.4,31
Effusive Constrictive Pericarditis
Effusive constrictive pericarditis is a rare inflammatory syndrome in which constrictive pathology (specifically involving the visceral layer) and compressive pericardial effusion exist simultaneously.23 The diagnosis should be considered when intracardiac pressures remain elevated following pericardiocentesis.35 The effusive constrictive pericarditis has been reported in 1.2% of patients with effusive pericarditis, and 6.8% of patients who underwent pericardiocentesis.36 The mortality rate is high (~ 60%).36 Potential etiologies and manifestations of effusive constrictive pericarditis overlap with constrictive pericarditis.23,35 Underlying etiology has an important impact on patient outcomes.36
Optimal diagnosis requires measuring both intra-pericardial and right atrial/biventricular pressures before and after pericardiocentesis.35 Noninvasive approaches such as echocardiography, CT, and MR imaging may be useful in demonstrating thickened pericardium in constrictive pericarditis but not in effusive constrictive pericarditis.37 Doppler echocardiography and MRI can assess cardiac hemodynamics and confirm pericardial inflammatory changes, respectively.35,38
Although many patients will require visceral pericardiectomy, some patients may improve spontaneously or respond to more conservative medical therapy with anti-inflammatory agents and treatment of underlying conditions.23,35
Pericardial Tumor Invasion
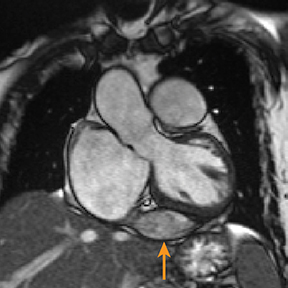
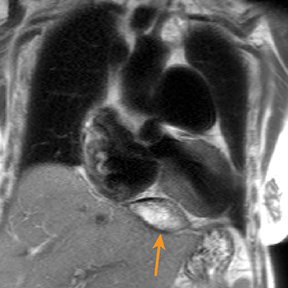
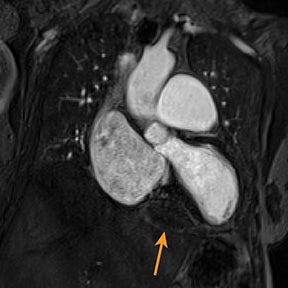
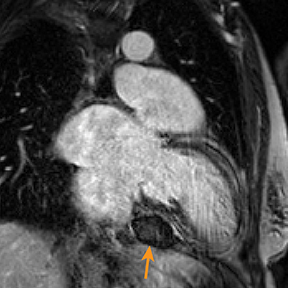
Metastatic pericardial diseases are more common than primary pericardial tumors.4 Approximately 10% of patients with malignancy have pericardial invasion at autopsy.4 Malignant cells from other organs may metastasize to the pericardium by lymphatic or hematogenous routes, in addition to direct, local extension.4 Common pericardial metastases include lung and breast cancers, melanoma, leukemia, and lymphoma.4,39 Primary pericardial tumors such as solitary fibrous tumor are less likely (Figure 5). Cardiac function is impaired in up to 30% of patients, owing to arrhythmias and compromised ventricular contraction.39 Clinical presentation is nonspecific and depends on the size and location of the lesions.39
Echocardiography may be limited by reduced tissue characterization.40 MRI and CT offer a greater field of view to evaluate the extent, and potentially the origin, of metastases.40,41 CT findings include enhancing pericardial nodules or masses.4,42 MRI has higher tissue characterization compared to echocardiography and CT, allowing better assessment of cardiac and extracardiac tumor extension.40,43 Most pericardial tumors enhance with contrast and demonstrate low signal intensity on T1 images and increased signal on T2 images (Figure 5).41 Positron emission tomography (PET) in combination with CT has also demonstrated particular utility in staging and follow-up of pericardial tumors.41,44
Neoplastic involvement of the pericardium may remain unknown until autopsy.41 The prognosis of pericardial tumor invasion is poor.43 Death from pericardial invasion usually occurs as the result of tamponade.4
Conclusion
Understanding the diagnostic imaging features, clinical significance, and management of acquired pericardial abnormalities will help radiologists improve their identification of these diseases and improve their communication with referring clinicians.
References
- Khandaker MH, Espinosa RE, Nishimura RA, et al. Pericardial disease: diagnosis and management. Mayo Clin Proc. 2010;85:572-593.
- Alter P, Figiel JH, Rupp TP, Bachmann GF, Maisch B, Rominger MB. MR, CT, and PET imaging in pericardial disease. Heart Fail Rev. 2013;18(3):289-306.
- Yared K, Baggish AL, Picard MH, Hoffmann U, Hung J. Multimodality imaging of pericardial diseases. JACC Cardiovasc Imaging. 2010;3(6):650-660.
- Rajiah P, Kanne JP. Computed tomography of the pericardium and pericardial disease. J Cardiovasc Comput Tomogr. 2010;4(1):3-18.
- Sagristà-Sauleda J, Mercé AS, Soler-Soler J. Diagnosis and management of pericardial effusion. World J Cardiol. 2011;3(5):135.
- Bezwada P, Quadri A, Shaikh A, Ayala-Rodriguez C, Green S. Myopericarditis and Pericardial Effusion as the Initial Presentation of Systemic Lupus Erythematosus. Case Rep Med. 2017;2017:1-4.
- Imazio M, Adler Y. Management of pericardial effusion. Eur Heart J. 2013;34(16):1186-1197.
- Foley J, Tong LP, Ramphul N. Message in a bottle. The use of chest radiography for diagnosis of pericardial effusion. Afr J Emerg Med. 2016;6(3):148-150.
- Herren J. Pericardial Effusion. In: Eltora A, Hymen C, Healey T. eds. Essential Radiology Review. Springer: Cham; 2019:223-224.
- Jung H-O. Pericardial effusion and pericardiocentesis: role of echocardiography. Korean Circ J. 2012;42(11):725-734.
- Goodman A, Perera P, Mailhot T, Mandavia D. The role of bedside ultrasound in the diagnosis of pericardial effusion and cardiac tamponade. J Emerg Trauma Shock. 2012;5(1):72.
- Schiavone WA. Cardiac tamponade: 12 pearls in diagnosis and management. Cleve Clin J Med. 2013;80(2):109-116.
- Ristić AD, Imazio M, Adler Y, et al. Triage strategy for urgent management of cardiac tamponade: a position statement of the European Society of Cardiology Working Group on Myocardial and Pericardial Diseases. Eur Heart J. 2014;35(34):2279-2284.
- Bogaert J, Francone M. Pericardial disease: value of CT and MR imaging. Radiology. 2013;267(2):340-356.
- Bodson L, Bouferrache K, Vieillard-Baron A. Cardiac tamponade. Curr Opin Crit Care. 2011;17(5):416-424.
- Pérez-Casares A, Cesar S, Brunet-Garcia L, Sanchez-de-Toledo J. Echocardiographic evaluation of pericardial effusion and cardiac tamponade. Front Pediatr. 2017;5:79.
- Petcu C, Dilof R, Bătăiosu C, Petcu P. Purulent pericardial effusions with pericardial tamponade–diagnosis and treatment issues. Curr Health Sci J. 2013;39(1):53.
- Kumar S, Iuga A, Jean R. Cardiac tamponade in a patient with dengue fever and lupus nephritis: a case report. J Intensive Care med. 2010;25(3):175-178.
- Snyder MJ, Bepko J, White M. Acute pericarditis: diagnosis and management. Am Fam Physician. 2014;89(7):553-560.
- Imazio M, Gaita F. Diagnosis and treatment of pericarditis. Heart. 2015;101(14):1159-1168.
- LeWinter MM. Acute pericarditis. N Engl J Med. 2014;371(25):2410-2416.
- Belgour A, Christiaens L, Varroud-Vial N, Vialle R, Tasu J. Chronic pericarditis: CT and MR imaging features. J Radiol. 2010;91(5 Pt 2):615-622.
- Rajiah P. Cardiac MRI: part 2, pericardial diseases. AJR Am J Roentgenol. 2011;197(4): W621-W634.
- Lilly LS. Treatment of acute and recurrent idiopathic pericarditis. Circulation. 2013;127(16):1723-1726.
- Imazio M, Brucato A, Cemin R, et al. Colchicine for recurrent pericarditis (CORP): a randomized trial. Ann Intern Med. 2011;155(7):409-414.
- Cafri C, Shalev A, Zahger D. ST-elevation myocardial infarction caused by coronary artery compression due to localized pericardial hematoma caused by coronary perforation. J Invasive Cardiol. 2010;22(11):E189-192.
- Nagai M, Kurokawa S, Ozaki M, Nomura M. Intrathoracic hemorrhage by deep pericardial sutures as detected by transesophageal echocardiography: report of two cases. JA Clin Rep. 2019;5(1):1-4.
- Veress G, Feng D, Oh JK. Echocardiography in pericardial diseases: new developments. Heart Fail Rev. 2013;18(3):267-275.
- Wang ZJ, Reddy GP, Gotway MB, Yeh BM, Hetts SW, Higgins CB. CT and MR imaging of pericardial disease. Radiographics. 2003;23(suppl_1):S167-S180.
- Syed FF, Schaff HV, Oh JK. Constrictive pericarditis—a curable diastolic heart failure. Nat Rev Cardiol. 2014;11(9):530.
- Imazio M, Brucato A, Maestroni S, et al. Risk of constrictive pericarditis after acute pericarditis. Circulation. 2011;124(11):1270-1275.
- Welch TD, Ling LH, Espinosa RE, et al. Echocardiographic diagnosis of constrictive pericarditis: Mayo Clinic criteria. Circ Cardiovasc Imaging. 2014;7(3):526-534.
- Kusunose K, Dahiya A, Popović ZB, et al. Biventricular mechanics in constrictive pericarditis comparison with restrictive cardiomyopathy and impact of pericardiectomy. Circ Cardiovasc Imaging. 2013;6(3):399-406.
- Gupta SK, Parakh N. Septal bounce illustrated on coronary angiogram. BMJ Case Rep. 2019;12(4) e230179.
- Syed FF, Ntsekhe M, Mayosi BM, Oh JK. Effusive-constrictive pericarditis. Heart Fail Rev. 2013;18(3):277-287.
- NTSEKhE M, Commerford PJ, Mayosi BM, Wiysonge CS. The prevalence and outcome of effusive constrictive pericarditis: a systematic review of the literature. Cardiovasc J Afr. 2012; 23(5):281.
- Salami M, Adeoye P, Adegboye V, Adebo O. presentation pattern and management of effusive–constrictive pericarditis in ibadan. Cardiovasc J Afr. 2012;23(4):206.
- Kim KH, Miranda WR, Sinak LJ, et al. Effusive-constrictive pericarditis after pericardiocentesis: incidence, associated findings, and natural history. JACC: Cardiovasc Imaging. 2018;11(4):534-541.
- Lichtenberger III JP, Reynolds DA, Keung J, Keung E, Carter BW. Metastasis to the heart: a radiologic approach to diagnosis with pathologic correlation. AJR Am J Roentgenol. 2016;207(4):764-772.
- Goldberg AD, Blankstein R, Padera RF. Tumors metastatic to the heart. Circulation. 2013;128(16):1790-1794.
- Van Beek EJ, Stolpen AH, Khanna G, Thompson BH. CT and MRI of pericardial and cardiac neoplastic disease. Cancer Imaging. 2007; 7(1):19.
- Prakash P, Kalra MK, Stone JR, Shepard J-AO, Digumarthy SR. Imaging findings of pericardial metastasis on chest computed tomography. J Comput Assist Tomogr. 2010;34(4): 554-558.
- Randhawa K, Ganeshan A, Hoey ET. Magnetic resonance imaging of cardiac tumors: part 2, malignant tumors and tumor-like conditions. Curr Probl Diagn Radiol. 2011;40(4):169-179.
- Makis W, Ciarallo A, Hickeson M, et al. Spectrum of malignant pleural and pericardial disease on FDG PET/CT. AJR Am J Roent. 2012;198(3):678-685.
Citation
P K, M H, AM I, B M, A B, HP M, H C.Acquired Pericardial Pathologies: Imaging Features, Clinical Significance, and Management. Appl Radiol. 2021; (2):10-15.
March 11, 2021