An Overview of Acute Mesenteric Ischemia
Images




This article is accredited for one SA-CME credit. Visit appliedradiology.org/SAM2 for full SA-CME information.
Acute mesenteric ischemia (AMI), a surgical emergency, is an uncommon but serious condition that accounts for 1 in 1000 hospital admissions in the United States and Europe1 and has extremely high mortality rates, ranging between 50 and 69%.2 Mortality remains high despite advances in diagnostic and treatment options, including the advent and widespread use of multidetector computed tomography (MDCT). This is thought to be the result of the vague nature of AMI’s clinical presentation, poor correlation with biochemical markers, and resultant associated delays in diagnosis and management.3
Acute insufficiency of mesenteric blood flow accounts for 60-70% of cases of mesenteric ischemia4, with the remainder related to chronic mesenteric ischemia, which is not a focus of this review. The major causes of AMI include mesenteric arterial occlusion (embolism or thrombosis), non-occlusive mesenteric ischemia due to intestinal hypoperfusion, and mesenteric venous occlusion (thrombosis).5-7 Vasculitis is a rare risk factor for AMI, but this review will focus on the aforementioned etiologies.
The incidence of AMI is increasing, owing partly to increased recognition and diagnosis, but more so to the increase in the aging population, with its cardiovascular and/or systemic diseases that further accentuate AMI risk. Early diagnosis and management are of utmost importance given AMI’s prevalence in the elderly population, who often have worse prognoses due to comorbidities.
Biphasic MDCT has become the standard of care for investigating and diagnosing AMI, particularly given its easy availability in the emergency setting.3 With its capacity to identify mesenteric ischemia, its severity, and potential causes, MDCT can have very high sensitivity and specificity.8 The following review provides a summary of AMI, its causes and relevant anatomy, and an illustrated review of imaging findings.
Pathophysiology, Causes, and Anatomy of AMI
Development of intestinal ischemia depends on the following factors: systemic perfusion, number and caliber of affected vessels, available collateral circulation, and duration of the ischemic insult. Ischemic injury develops with prolonged mismatch between cellular metabolism and delivery of oxygen and nutrients. This mismatch can result from mesenteric arterial occlusion (MAO), mesenteric venous congestion/occlusion (MVO), or non-occlusive mesenteric ischemia (NOMI), as discussed below. In the gastrointestinal (GI) tract, mismatch with a 75% reduction in mesenteric blood flow can be tolerated for up to 12 hours without significant injury.9
It is also important to recognize that intestinal injury is caused by both tissue hypoxia and reperfusion. The latter occurs following blood flow restoration, with further damage caused by the release of free oxygen radicals, toxic ischemic injury byproducts, and neutrophil activation, which may lead to multisystem organ failure.10,11 Alternatively, after a prolonged period of ischemia, progressive vasoconstriction is established in the obstructed vascular territory, which can persist even after blood flow returns, leading to persistent ischemia, full-thickness necrosis of the bowel wall, and perforation.4,6
Based on AMI pathophysiology; ie, MAO, NOMI, or MVO, three subtypes of ischemic change can be identified that are relevant to imaging findings (Figure 1):
1. Pale ischemic type resulting from persistent arterial insufficiency without reperfusion;
2. Hemorrhagic type resulting from transient arterial insufficiency with subsequent reperfusion and associated injury; and,
3. Venous type resulting from impaired venous drainage (most commonly seen as the mechanism for ischemia in setting of mesenteric strangulation or venous thrombosis).
Ischemic damage can range from reversible ischemia, including mucosal infarction and mural infarction involving the mucosa and submucosa, to irreversible transmural infarction and perforation. The bowel demonstrates spastic contraction in the early phase of the disease, followed by adynamic ileus caused by ischemic tissue damage, including muscular and neurologic infarction.
Mesenteric artery occlusions account for most AMI cases, which can be related to cardioembolism, arterial thrombosis, occlusive atherosclerosis, arterial dissection, post-vascular interventions (eg, iatrogenic embolic showers),12 and, rarely, to vasculitides or fibromuscular dysplasia (Table).
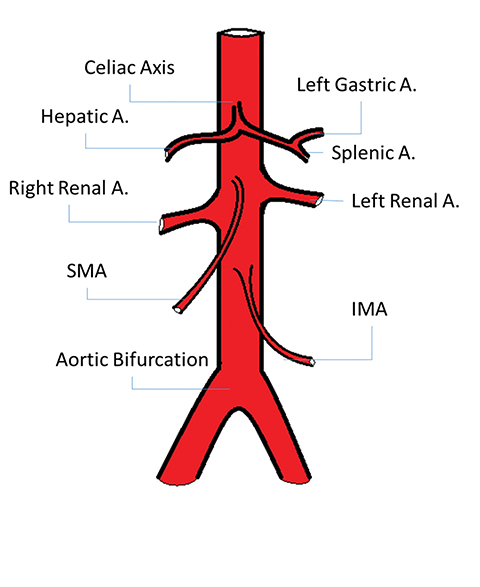
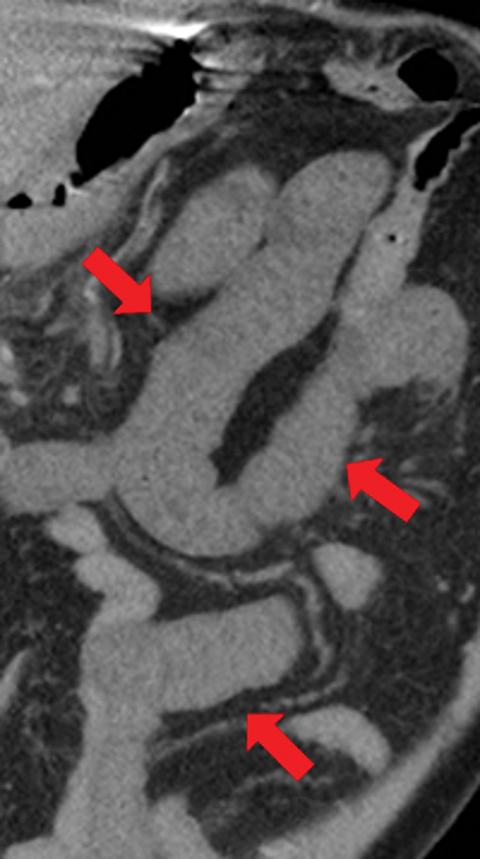
Given this common etiology, knowledge of arterial anatomy and territorial supply to the GI tract is key to AMI diagnosis, causality, and treatment planning. The GI tract is supplied by three major arteries: the celiac artery, which provides blood to the stomach and duodenum; the superior mesenteric artery (SMA), which supplies the jejunum, ileum, and proximal colon to the splenic flexure; and the inferior mesenteric artery (IMA), which supplies the distal colon, from the splenic flexure to the rectum (Figure 2).
The most common site of MAOs from thromboembolic events is the SMA, owing to its large diameter and narrow takeoff angle that lead to anatomical susceptibility to lodging of emboli. This phenomenon often happens 3 to 10 cm distal to the origin of SMA, in a segment that tapers distal to the origin of the middle colic artery, thereby sparing the first few tributary branches to the jejunum. It is noteworthy, however, that in 15% of thromboembolic events the embolus is lodged at the origin of the SMA.4 The middle segment of the jejunum is most often involved in the ischemic process as it is the farthest from the collateral circulation of the celiac axis and the inferior mesenteric artery, while the proximal jejunum is usually spared. The IMA is rarely affected by embolus due to its small caliber.5
The second most common etiology of AMI is NOMI. In this condition, the mesenteric vessels are patent but blood flow is very low, causing splanchnic vasoconstriction with subsequent multifocal infarction of solid and visceral organs. NOMI can therefore be challenging to diagnose and may result from shock (hemorrhagic, cardiogenic, or septic), heart failure (due to acute myocardial infarction or congestive heart failure), dehydration, stress (high-endurance athletes), chronic renal failure requiring hemodialysis,13 or drugs, illicit and therapeutic; eg, cocaine, digitalis, ergot derivatives, and norepinephrine.14 NOMI is often associated with the worst outcomes, reaching mortality rates of 58-70%.
The third-most prevalent cause of AMI is MVO, which results from venous congestion caused by mesenteric strangulation, venous thrombosis,15 phlebitis of intramural veins (rarely), or overdistention.
Imaging Technique
Prior to widespread availability of MDCT, catheter angiographic studies were used in suspected cases of AMI to identify and treat the underlying etiology.4 Today, MDCT is capable of diagnosing AMI, as well as potential other causes of “acute abdomen”, with excellent sensitivity and specificity.8 In a recent meta-analysis of six studies (3 prospective and 3 retrospective) with 142 positive AMI cases out of 619, MDCT was shown to have a pooled sensitivity and specificity of 93.3% and 95.9%, respectively, for AMI when analyzed by experienced radiologists.8
The optimal protocol for AMI detection is MDCT with unenhanced images followed by arterial and venous phases without oral contrast.16 Between 100 and 150 mL of nonionic iodinated contrast material is administered at a rate of 2.5–4 mL per second, with a scanning delay of 30 seconds for the arterial phase and 60–70 seconds for the venous phase. Axial sections of 5-mm thickness are acquired with coronal and sagittal reformations. Bolus tracking is preferred, as follows: arterial phase scanning commencing 2 seconds after trigger threshold of 150 Hounsfield units (HU) reaches the supraceliac aorta, with the portal venous phase scan acquired at a fixed delay of 45 seconds thereafter.
Imaging Findings and Management
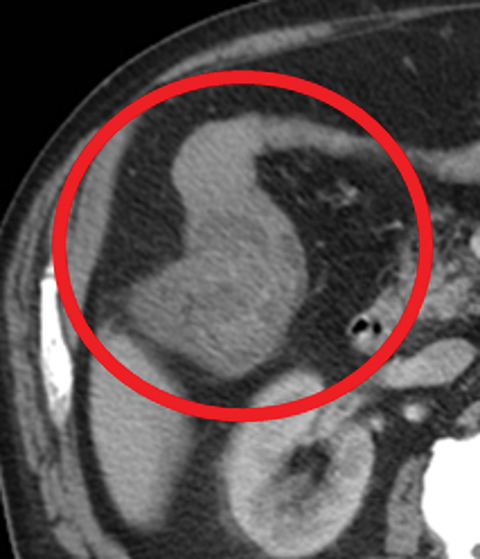
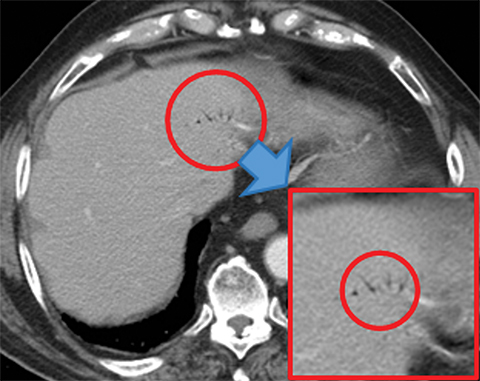
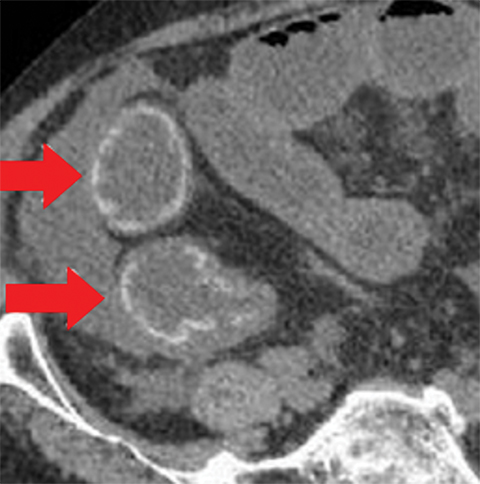
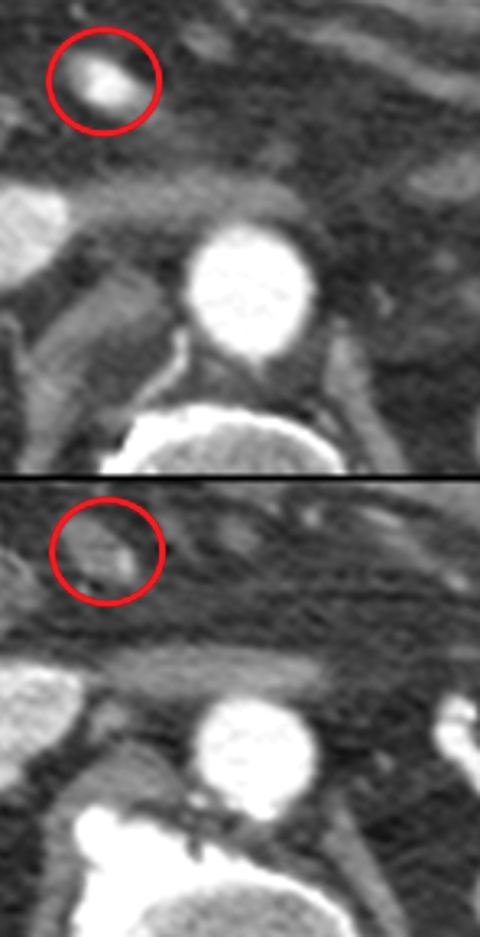
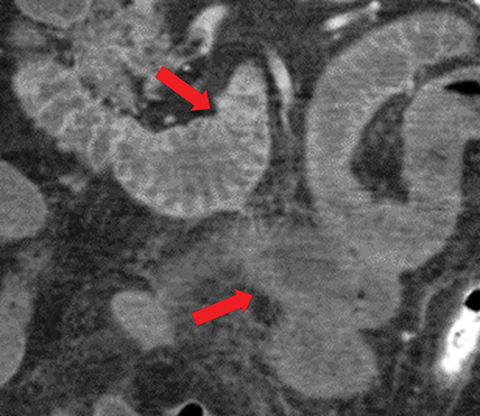
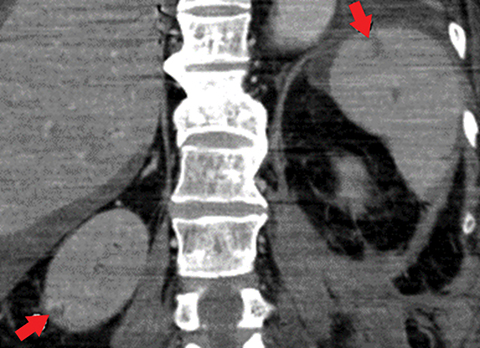
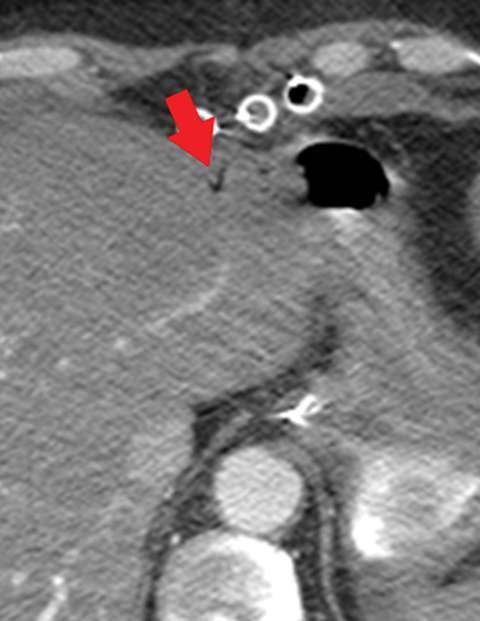
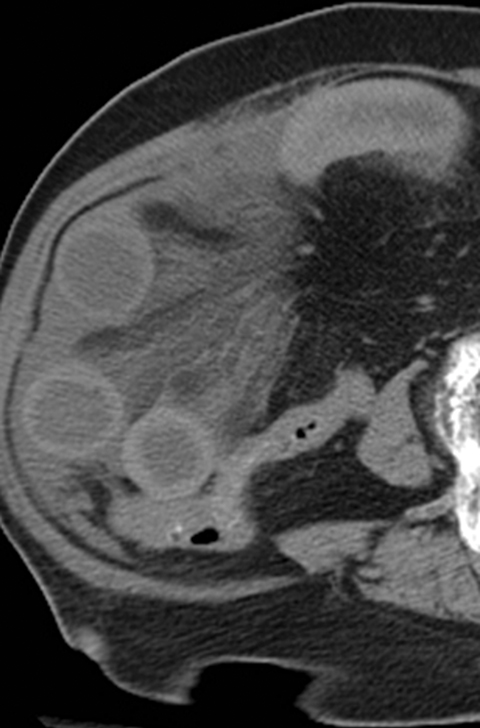
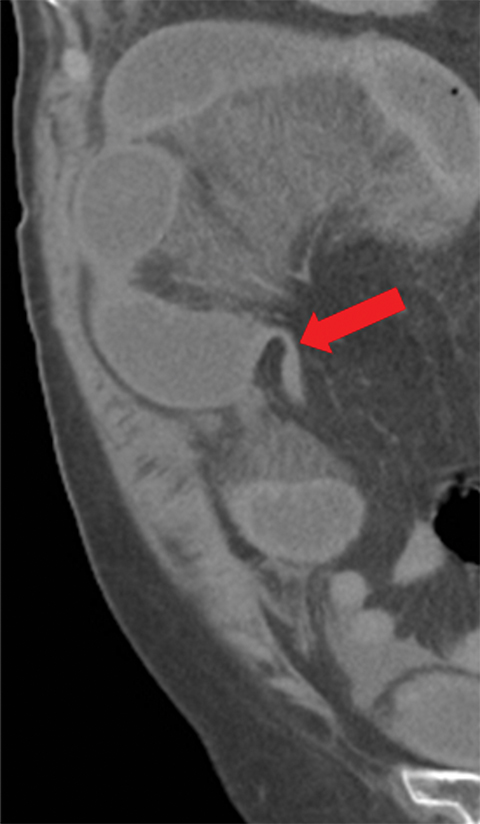
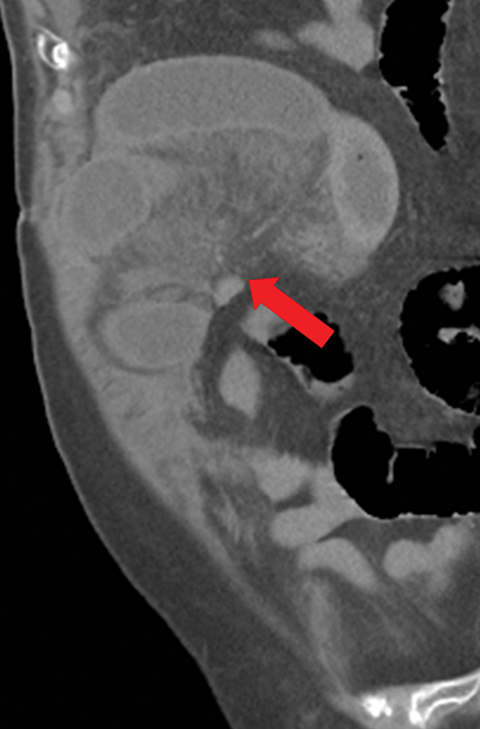
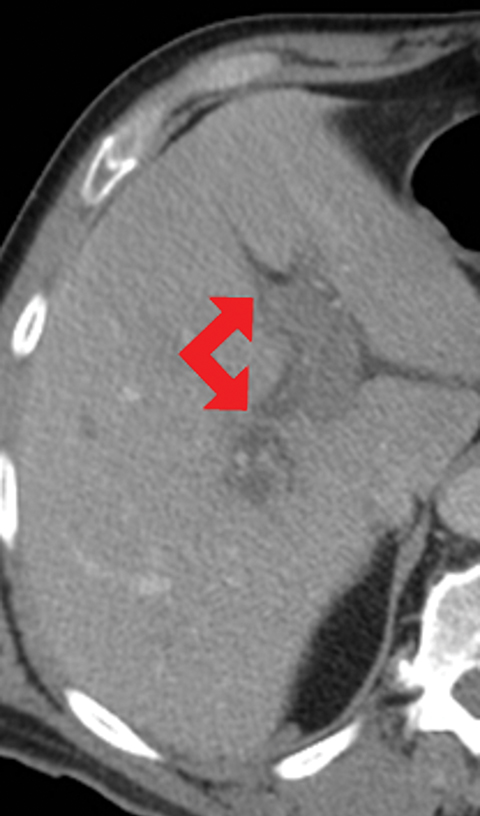
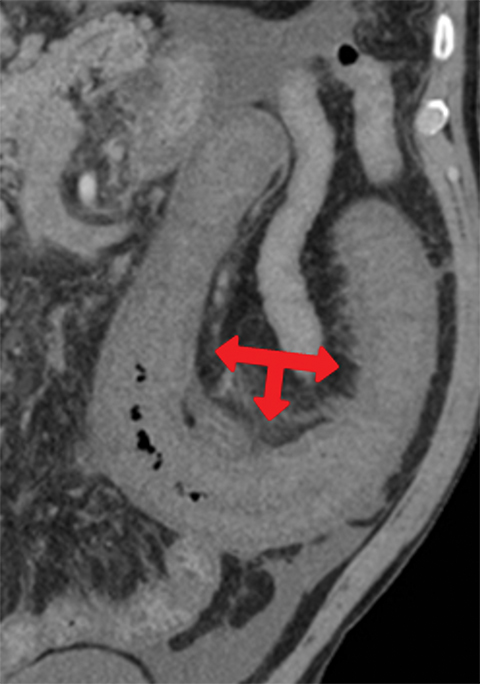
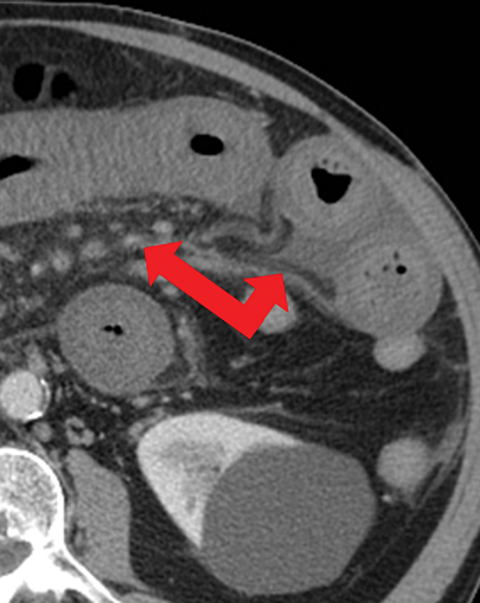
Many MDCT findings can be suggestive of AMI. They differ on the basis of pathogenesis (veno-occlusive or arterio-occlusive), severity and stage, location (small bowel or large bowel), and presence of hemorrhage. The features with the highest positive predictive value are decreased or absent bowel-wall enhancement (Figure 3A), increased attenuation (intramural hemorrhage) of the bowel wall on unenhanced CT (Figure 4); and filling defect in vessels representing thrombus (Figure 5B). Filling defects may be hyper-attenuating on pre-contrast images. Particular attention should be paid to filling defects in the SMA (Figure 5B), IMA, SMV, and small mesenteric arteries, as well as to ancillary findings of ischemia in the liver, spleen, and kidneys (Figure 6) which are commonly overlooked.17
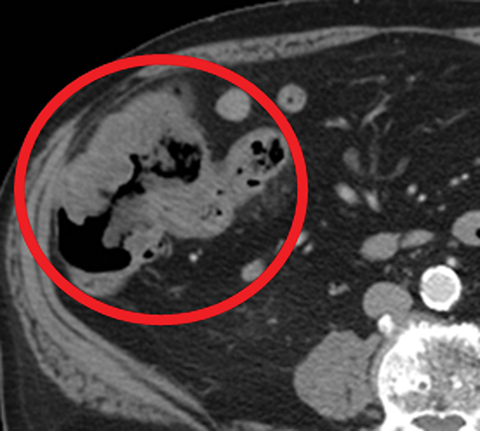
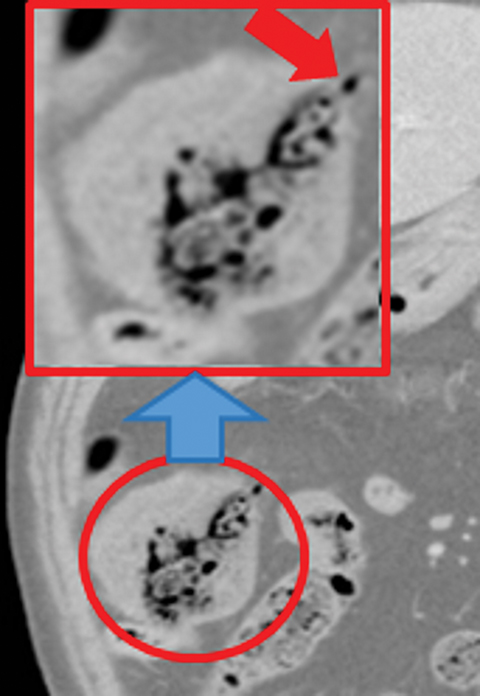
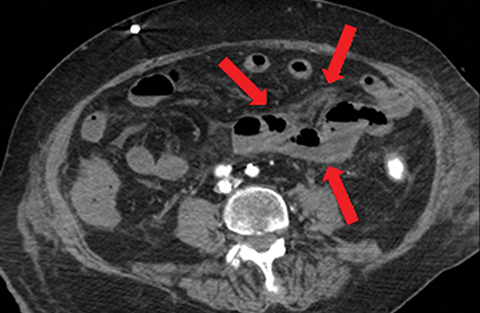
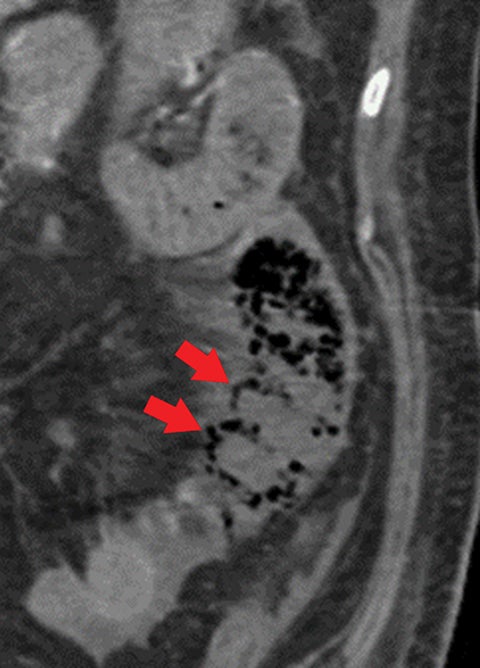
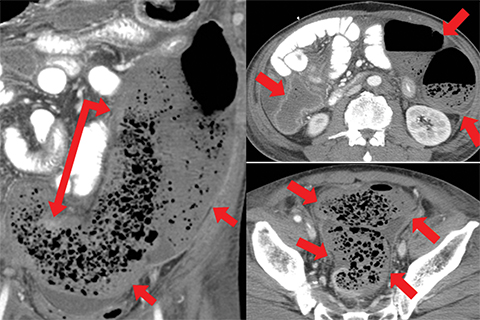
Other important but less specific MDCT findings include bowel-wall thickening (Figure 3A), the most common and least specific sign; dilated lumen with paper-thin wall, due to interruption of peristalsis reflex due to ischemic injury and/or irreversible and transmural ischemic damage to bowel wall; mesenteric stranding, and free fluid. Late findings of AMI are ominous and often associated with worse outcomes and irreversible ischemia; these include extraluminal (free or focal), intravenous (mesenteric or portal venous), or mural (pneumatosis intestinalis) gas, which all indicate transmural injury with or without perforation (Figures 3,7). The etiologies of pneumatosis intestinalis (Figure 8) and portal venous gas in ischemic colitis are debated, but a popular theory suggests transmural ischemia and subsequent bacterial translocation. In cases of intestinal pneumatosis associated with suspected bowel ischemia, detection of hepatic portal or portomesenteric venous gas increases the likelihood of transmural bowel infarction (Figures 3,7,8).
Characteristic Imaging Findings and Management for the Three Main Causes of AMI
Imaging findings in arterial occlusion bowel ischemia include:
• A bowel wall that may be thinned and/or featureless (Figures 5,7), unchanged, or thickened (specifically in reperfusion) (Figure 3).
• Proximal occlusions, which cause greater injury (Figure 5).
• Increased bowel-wall attenuation in hemorrhagic infarction on unenhanced CT (Figure 4).
• Absent or diminished attenuation on contrast-enhanced CT (CECT, Figure 5), target appearance, or increased attenuation with reperfusion.
• Fat stranding and hazy mesentery in the setting of established ischemia (Figure 5).
Management of AMI resulting from arterial occlusion bowel includes anticoagulation, endovascular treatment (EVT) by an interventional radiologist, and surgical intervention, such as bowel resection.3,18,19
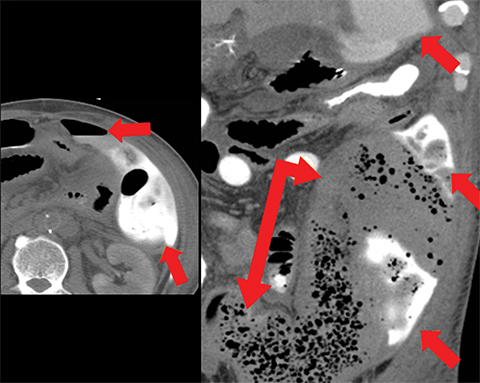
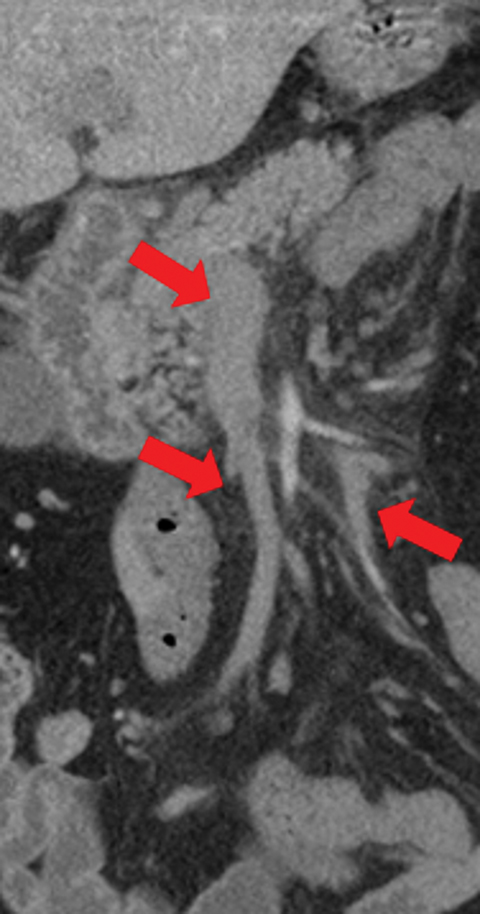
Veno-occlusive causes of AMI can appear similar to arterial occlusion, with bowel-wall thickening and variable attenuation on unenhanced CT (Figures 1,4, 9), absent or diminished attenuation on the contrast-enhanced phase or a target-sign appearance with reperfusion. Additional imaging findings include:
• Moderate or prominent dilatation of bowel with fluid retention.
• Bowel strangulation and obstruction (Figure 8).
• Portomesenteric venous thromboses with venous engorgement (Figure 9).
• Hazy mesentery and ascites resulting from increased back-pressure (Figure 8).
Systemic anticoagulation is the first-line therapy for veno-occlusive AMI;20 however, rapid recanalization with systemic thrombolytic therapy and EVT may help improve outcomes in patients with large thrombotic burden and clinical deterioration.21
NOMI, the most difficult entity to identify early, is characterized by segmental and discontinuous involvement unrelated to a specific arterial territory. The most reliable early finding is differential enhancement, which is difficult to recognize owing to its discontinuous and segmental distribution. Treatment is often supportive, with attempts to reverse the insulting factors; eg, reversal of shock by addressing myocardial injury or hypovolemia. However, mesenteric contraction and vasospasm often persist, even after addressing precipitating events, and lead to worse prognosis.
Developing Technologies and Future Directions
Although CECT angiography has shown high sensitivity to acute bowel ischemia,8 the most common and readily recognized signs detailed above, including bowel-wall thickening, bowel dilatation, mesenteric stranding, and fluid, remain nonspecific. Conversely, bowel-wall hypoenhancement, the CT sign with the highest specificity (93–100%), is not as easily recognized.22-26 Difficulty recognizing hypo-enhancement may be technique related; eg, the use of low energy levels, low volume/concentration of contrast agent, inadequate delay time or case related, as in the presence of intramural hemorrhage with high attenuation of the bowel wall on unenhanced CT or luminal distention with thinned bowel wall.
In recent years, strides made by dual-energy CT (DECT) technology toward improved recognition of differential bowel-wall enhancement have garnered interest.16DECT has been shown to help identify such differences more readily than conventional MDCT.16,27 However, DECT is not widely available and remains in its infancy with respect to clinical application. Further studies are required to consolidate the degree of improved performance and specificity for bowel-wall hypoenhancement detection with DECT.
Conclusion
Acute mesenteric ischemia is a rare but life-threatening condition that requires timely recognition and intervention. The role of radiologists is pivotal to early identification of AMI; hence, considering AMI as a differential diagnosis when protocolling and identifying the subtle image findings detailed in this review remains vital to delivering the best patient care and outcomes.
References
- Stoney RJ, Cunningham CG. Acute mesenteric ischemia. Surgery. 1993; 114:489-490.
- Adaba F, Askari A, Dastur J et al. Mortality after acute primary mesenteric infarction: a systematic review and meta-analysis of observational studies. Colorectal disease: the official journal of the Association of Coloproctology of Great Britain and Ireland. 2015; 17:566-577.
- Kanasaki S, Furukawa A, Fumoto K et al. Acute Mesenteric Ischemia: Multidetector CT Findings and Endovascular Management. Radiographics.2018; 38:945-961.
- McKinsey JF, Gewertz BL. Acute mesenteric ischemia. The Surgical clinics of North America. 1997; 77:307-318.
- Cappell MS. Intestinal (mesenteric) vasculopathy. I. Acute superior mesenteric arteriopathy and venopathy. Gastroenterology Clinics of North America. 1998; 27:783-825, vi.
- Reinus JF, Brandt LJ, Boley SJ. Ischemic diseases of the bowel. Gastroenterology Clinics of North America. 1990; 19:319-343.
- Acosta S, Ogren M, Sternby NH et al. Clinical implications for the management of acute thromboembolic occlusion of the superior mesenteric artery: autopsy findings in 213 patients. Ann Surg. 2005; 241:516-522.
- Menke J. Diagnostic accuracy of multidetector CT in acute mesenteric ischemia: systematic review and meta-analysis. Radiology. 2010; 256:93-101.
- Boley SJ, Frieber W, Winslow PR, al e. Circulatory responses to acute reduction of superior mesenteric arterial flow. Physiologist. 1969; 12:180.
- Granger DN, Rutili G, McCord JM. Superoxide radicals in feline intestinal ischemia. Gastroenterology. 1981; 81:22-29.
- Zimmerman BJ, Granger DN. Reperfusion injury. The Surgical clinics of North America. 1992; 72:65-83.
- Perry RJ, Martin MJ, Eckert MJ et al. Colonic ischemia complicating open vs endovascular abdominal aortic aneurysm repair. J Vasc Surg. 2008; 48:272-277.
- John AS, Tuerff SD, Kerstein MD. Nonocclusive mesenteric infarction in hemodialysis patients. J Am Coll Surg. 2000; 190:84-88.
- Brandt LJ, Feuerstadt P, Longstreth GF, Boley SJ. ACG clinical guideline: epidemiology, risk factors, patterns of presentation, diagnosis, and management of colon ischemia (CI). Am J Gastroenterol. 2015; 110:18-44; quiz 45.
- Abu-Daff S, Abu-Daff N, Al-Shahed M. Mesenteric venous thrombosis and factors associated with mortality: a statistical analysis with five-year follow-up. J Gastro Surg. 2009; 13:1245-1250.
- Lourenco PDM, Rawski R, Mohammed MF et al. Dual-Energy CT Iodine Mapping and 40-keV Monoenergetic Applications in the Diagnosis of Acute Bowel Ischemia. AJR. Am J Roentgenol. 2018; 211:564-570.
- Firetto MC, Lemos AA, Marini A et al. Acute bowel ischemia: analysis of diagnostic error by overlooked findings at MDCT angiography. Emergency Radiology. 2013; 20:139-147.
- Yasuhara H, Niwa H, Takenoue T, Naka S. Factors influencing mortality of acute intestinal infarction associated with SIRS. Hepato-gastroenterology. 2005; 52:1474-1478.
- Karkkainen JM, Lehtimaki TT, Saari P et al. Endovascular Therapy as a Primary Revascularization Modality in Acute Mesenteric Ischemia. Cardiovasc Intervent Radiol. 2015; 38:1119-1129.
- Tilsed JV, Casamassima A, Kurihara H et al. ESTES guidelines: acute mesenteric ischaemia. Eur J Trauma Emerg Surg. 2016; 42:253-270.
- Acosta S, Bjorck M. Modern treatment of acute mesenteric ischaemia. Brit J Surg. 2014; 101:e100-108.
- Taourel PG, Deneuville M, Pradel JA et al. Acute mesenteric ischemia: diagnosis with contrast-enhanced CT. Radiology. 1996; 199:632-636.
- Zalcman M, Sy M, Donckier V et al. Helical CT signs in the diagnosis of intestinal ischemia in small-bowel obstruction. AJR. Am J Roentgenol. 2000; 175:1601-1607.
- Kirkpatrick ID, Kroeker MA, Greenberg HM. Biphasic CT with mesenteric CT angiography in the evaluation of acute mesenteric ischemia: initial experience. Radiology. 2003; 229:91-98.
- Sheedy SP, Earnest Ft, Fletcher JG et al. CT of small-bowel ischemia associated with obstruction in emergency department patients: diagnostic performance evaluation. Radiology. 2006; 241:729-736.
- Geffroy Y, Boulay-Coletta I, Julles MC et al. Increased unenhanced bowel-wall attenuation at multidetector CT is highly specific of ischemia complicating small-bowel obstruction. Radiology. 2014; 270:159-167.
- Potretzke TA, Brace CL, Lubner MG et al. Early small-bowel ischemia: dual-energy CT improves conspicuity compared with conventional CT in a swine model. Radiology. 2015; 275:119-126.
Citation
A M, C G, H G,. An Overview of Acute Mesenteric Ischemia. Appl Radiol. 2021;(1):10-18.
January 19, 2021