MDCT of the small bowel
Dr. Geraghty is Resident in Radiology, and Dr. Desser is an Associate Professor of Radiology, Department of Radiology, Stanford University School of Medicine, Stanford, CA.
Historically, the length and convoluted course of the small bowel have presented formidable diagnostic challenges to radiologists. The volumetric and multiplanar images that can now be obtained routinely with multidetector computed tomography (MDCT) scanners have greatly expanded the ability of CT to systematically evaluate the small bowel luminal contents, with the added bonus of being able to evaluate the bowel wall, mesentery, and associated vasculature. This article will summarize the use of MDCT in a variety of pathologic entities that involve the small bowel.
Technique
Small bowel MDCT imaging protocol
At our institution, all examinations of the abdomen are done on 8-, 16-, or 64-detector scanners. Sample MDCT scan parameters for small bowel studies on our 16-slice scanner (LightSpeed 16 scanner, GE Healthcare, Waukesha, WI) are shown in Table 1. The basic principle of MDCT protocol design is to scan with thin collimation, reconstruct thick slices for viewing, and reconstruct thin slices with overlap for multiplanar reformations.
Luminal contrast media
Positive oral contrast media that contain either dilute iodine or barium are useful for differentiating bowel segments from extraluminal collections, such as abscesses and fistulous tracts. But high-density media can mix unevenly with fluid bowel contents, creating pseudo-tumors, 1 and can also obscure features of the enhancing bowel wall. In 3-dimensional imaging studies of the vasculature, high-density segments must be painstakingly edited out. Thus, in our practice, we prefer to use water to distend the small bowel, and we do not routinely administer positive oral contrast media unless an abscess or fistula is suspected. We administer 750 mL of water 20 minutes prior to scanning, with an additional 250 mL given to the patient just before the scan to distend the stomach. Limitations of intraluminal water include rapid transit and quick absorption, which can result in poor distention at the time of imaging. VoLumen (E-Z-EM Inc., Lake Success, NY), an ultra-low-density (0.1%, 15 to 30 HU) barium suspension that contains sorbitol, has been reported to improve distention of distal bowel segments. 2
We routinely use intravenous contrast for evaluating enhancement patterns of the bowel wall and mesenteric vasculature, except when an intramural hemorrhage is suspected. An injection rate of 2 to 3 mL/sec is adequate for most indications. For angiographic studies, an injection rate of 4 to 5 mL/sec is required.
Anatomy
The duodenum is retroperitoneal, with the exception of the first 2 centimeters of the first portion (within the hepatoduodenal ligament) and the last few millimeters of the fourth part (at the ligament of Treitz). This transition of the fourth portion from a retroperitoneal to an intraperitoneal position creates the paraduodenal recesses of Landzert (on the left) and Waldeyer (on the right), which are potential sites of internal hernias. 3 The jejunum and ileum are entirely intraperitoneal.
Normal wall thickness of the fully distended small bowel should not exceed 2 mm. If the bowel is collapsed or if a slice is obtained within the plane of the valvulae conniventes, this can give a false appearance of focal concentric bowel wall thickening. However, true pathologic bowel wall thickening should be contiguous along a segment (Figure 1).
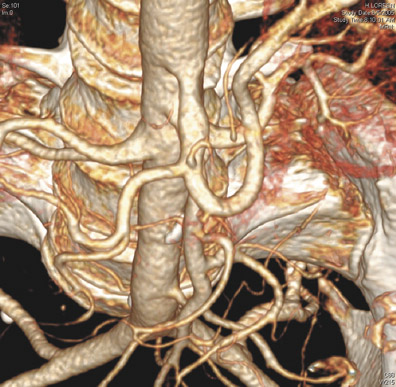
The gastroduodenal branches of the celiac axis supply the first and second portions of the duodenum, and the inferior pancreatoduodenal branches of the superior mesenteric artery (SMA) supply the third and fourth portions. The jejunum and ileum are supplied by, respectively, the jejunal and ileal branches of the SMA. Multidetector CT has largely supplanted catheter angiography as the principal method to evaluate the mesenteric vasculature. The mesenteric arcades and vasa recta are exquisitely seen in the coronal plane when thin collimation and volume rendering is utilized (Figure 2).
Pathology
Diverticula
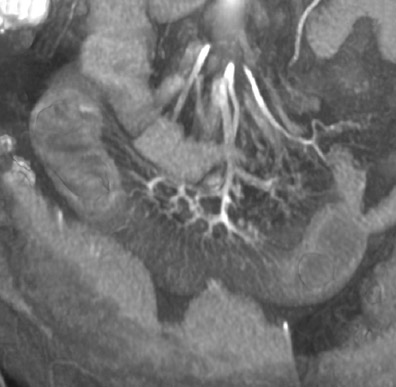
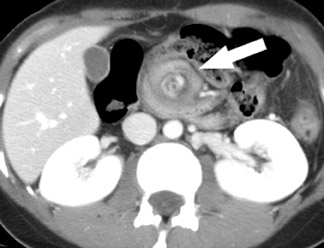
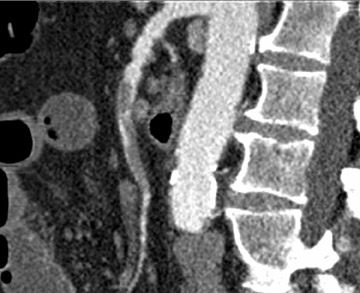
Small bowel diverticula are outpouchings that typically extend from the mesenteric side of the bowel wall. Duodenal diverticula are the most common, with most of these acquired and asymptomatic. Intraluminal "windsock" diverticula, by contrast, are congenital and may be symptomatic. Multidetector CT multiplanar rendering can clearly show the orientation of these lesions and can assist in surgical planning 4 (Figure 3).
Meckel's diverticula are the most common type of congenital abnormality of the small bowel, with a prevalence of approximately 2%. They arise from the antimesenteric side of the bowel wall typically within 100 cm of the ileocecal valve and result from incomplete closure of the intestinal end of the embryonic omphalomesenteric duct. On CT, they are most commonly found at or near the midline and range from 1 to 10 cm in length. They may present clinically with diverticulitis, intussusception, or bleeding. 5
Small bowel obstruction
Small bowel obstruction (SBO) accounts for 20% of surgical admissions for acute abdominal pain. The causes of SBO can be divided into 3 broad categories, including extrinsic, intrinsic, and intraluminal etiologies (Table 2). 6 In Western societies, the most common causes of SBO are adhesions, Crohn's disease, and neoplasia, while hernias are the most common cause of SBO in developing countries.
Prompt diagnosis is crucial because missed or delayed recognition can result in life-threatening complications, such as ischemia or perforation. Plain films are often equivocal, and further evaluation with CT is usually indicated to elucidate the site and cause of obstruction as well as to detect potential complications of closed-loop obstruction or strangulation. Exogenous oral contrast is usually not necessary because the fluid retained in obstructed bowel loops provides excellent distention. In fact, positive oral contrast decreases the conspicuity of bowel wall enhancement, making ischemia more difficult to detect.
In mechanical small bowel obstruction, CT shows dilated bowel segments (>25 mm diameter) proximal to collapsed loops distally. 7 The specific point of transition may be seen as a small triangular "beak" immediately beyond the dilated segment. A "small bowel feces" sign may also be seen immediately proximal to the transition zone and can be helpful in identifying the site of obstruction. This sign is defined as gas bubbles and particulate matter within dilated small bowel segments that mimic colonic fecal matter and that result from stasis and water absorption. 8 Although it is fairly specific for SBO, it has also been reported in cystic fibrosis and other entities. 9
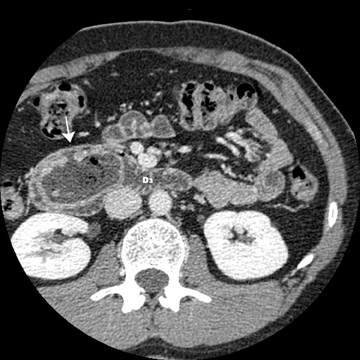
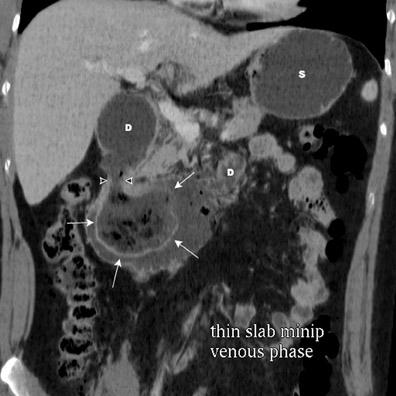
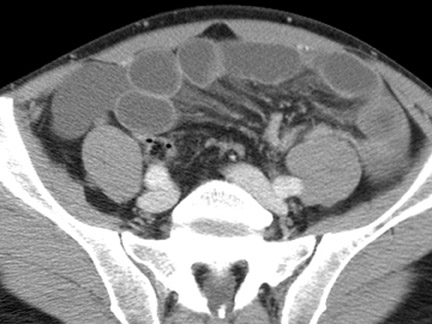
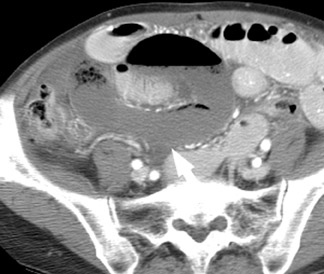
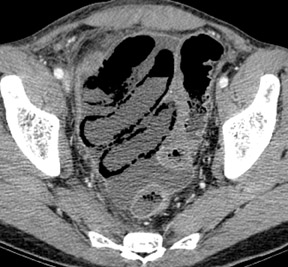
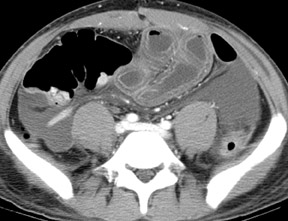
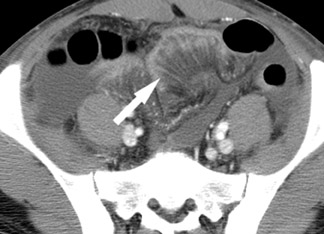
Identifying the complication of closed-loop obstruction is critical, as these patients are at high risk for developing ischemia (strangulation) as a consequence of impaired arterial or venous flow. CT findings of closed-loop obstruction depend on the orientation and length of the incarcerated segment and include a dilated, fluid-filled segment with a "C"- or "U"-shaped configuration, and a radial or "spoke-wheel" arrangement of mesenteric vessels (Figure 4). 7,10 With superimposed volvulus, CT may show engorged mesenteric vessels converging toward the abnormal segment, often with delayed enhancement. Often coronal images may exhibit these features when they are not apparent in the axial plane. A "whirl" sign (Figure 5), which represents rotation of the mesentery in small bowel volvulus, may be seen in the axial plane but is best appreciated in the coronal or sagittal planes, depending on the axis of rotation. Superimposed ischemia in SBO is usually due to impaired venous outflow in a closed-loop obstruction.
Mesenteric ischemia
Acute small bowel ischemia can result from occlusive (arterial or venous) causes as well as nonocclusive causes, such as hypoperfusion. Most cases of acute mesenteric ischemia are due to thromboembolic disease obstructing the proximal portion of the SMA. Embolic disease related to atrial fibrillation is the most common etiology for SMA occlusion. Venous occlusion may result from a hypercoagulable state and/or in the postsurgical setting but can also occur as a result of infiltrative, neoplastic, or inflammatory/infectious conditions. As described above, closed-loop obstruction may result in bowel ischemia if the veins are twisted, stretched, or compressed.
The use of routine MDCT protocols of the abdomen can often reveal small bowel ischemia in patients with nonspecific symptoms or suspected SBO. However, if bowel ischemia is the specific question, mesenteric CT angiography (CTA) is preferred at our institution to determine the presence, site, distribution, and cause. Three-dimensional volume-rendered processing of vascular structures is a helpful adjunct to primary interpretation of 2-dimensional images and allows immediate recognition of the vascular abnormalities, even in the presence of dense calcifications (Figure 6). The postprocessed volumetric images are often preferred by our clinicians, since one image presents a synthesis of the findings of hundreds of source images.
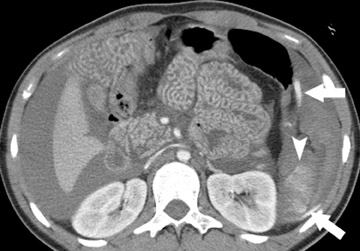
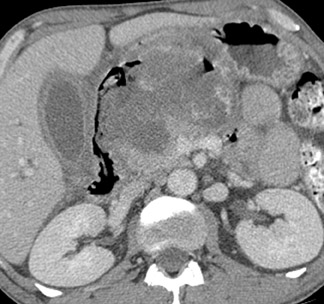
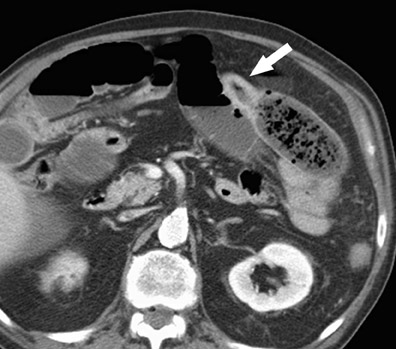
CT findings in acute mesenteric ischemia are variable, depending on the etiology and degree of damage to the bowel wall. 11 The most common CT finding in acute bowel ischemia is bowel wall thickening, which is present in 26% to 96% of reported cases, 12 due to mural edema, hemorrhage, and/or superinfection of the ischemic bowel wall. However, this is also the least specific finding, and the degree of bowel wall thickening does not correlate with the severity of ischemic bowel wall damage. 13 Acute arterial occlusions may actually produce thinning of bowel wall segments because of the loss of motor tone in the ischemic wall musculature. The key CT findings in mesenteric ischemia are summarized in Table 3 and include mesenteric fat stranding, mesenteric fluid, ascites, submucosal edema with low attenuation in the bowel wall, and increased attenuation due to hyperemia, intramural hematoma, or hemorrhagic infarction. Segmental absence of bowel enhancement is an especially helpful sign of focal ischemia (Figure 7). Hyperemia and hyperperfusion of an ischemic segment that primarily affect the mucosa and submucosa may produce a "target" sign. Pneumatosis and porto-mesenteric venous gas are less common and are usually late findings in acute ischemia (Figure 8), but they also have a variety of benign etiologies. 12,14-16
In mesenteric ischemia caused by a low-flow state (nonobstructive ischemia), CT may show diffusely narrowed arteries as well as distal branches that are fewer in number and of decreased caliber. "Shock bowel" is a reversible, nonocclusive ischemia that is thought to be a reperfusion injury related to resuscitation following severe hypotension. CT findings include diffuse small bowel wall thickening and hyperattenuation, with persistent increased enhancement and slow washout (Figure 9). 17
Chronic mesenteric ischemia results in a characteristic clinical syndrome of chronic abdominal pain, especially after meals, and is frequently associated with weight loss. It most frequently affects elderly patients and is secondary to long-standing atherosclerotic disease. Mesenteric CTA has become a common screening technique for this entity. 18 Findings may include arterial plaque formation, luminal narrowing, and collateral vessel formation.
Crohn's disease
Crohn's disease is a chronic granulomatous disease that can affect any portion of the gastrointestinal tract in a noncontiguous fashion (socalled skip lesions) and most frequently involves the terminal ileum. The recent introduction of wireless capsule endoscopy (WCE) into clinical practice has led to the detection of the early small bowel mucosal disease, such as aphthoid ulcers, even in the setting of a negative enteroclysis. 19 It is likely that the even the less-sensitive small bowel follow-through examination will diminish in importance in this new era of WCE. By contrast, CT can depict changes of Crohn's disease beyond the mucosa and beyond the scope of WCE.
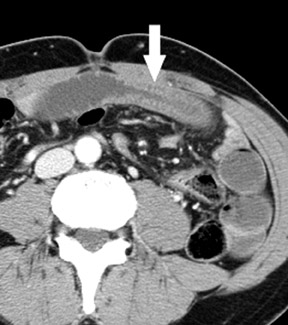
CT is useful in determining the presence, extent, and severity of Crohn's disease, to assess disease activity, and to detect extraintestinal complications. 20 Findings vary depending on whether the disease is in its acute or chronic phase. The most common finding is wall thickening (up to 2 cm), which is often associated with luminal narrowing. During the acute stage, the mural stratification of the bowel wall is maintained, often with a target (or "double halo") sign produced by concentric rings that represent enhancing mucosa, lower attenuation submucosa, and higher attenuation muscularis propria (Figure 10). The outer contours of the bowel wall are often irregular during the acute stage, and disease activity has been correlated with the intensity of contrast enhancement in the inflamed mucosa and serosa. 20,21 With disease progression, mural stratification is lost and there is homogenous wall enhancement in association with fibrosis. Both inflammatory and fibrotic changes tend to occur on the antimesenteric side of the bowel.
Inflammatory hyperemia of the mesenteric vasculature creates the "comb" sign, which also suggests active disease (Figure 11). 22 The engorged vasa recta creates a series of parallel opacities that mimic the teeth of a comb. Coronal images often beautifully display the mesenteric hypervascularity. In Crohn's disease, local mesenteric lymph nodes are increased in number but small in size (usually <1 cm). Both of these findings can be used to help distinguish Crohn's disease from neoplastic processes. 23 The separation of bowel loops caused by fibrofatty proliferation of the central mesentery ("creeping fat") and perirectal fat deposition are other common signs. Because of edema and infiltration with inflammatory cells, attenuation measurements of the involved fat are often increased.
Complications of Crohn's disease (such as fistulae, abscesses, and extramural extension to adjacent structures) often dictate patient management and are well depicted with MDCT. 24 Three-dimensional reconstructions of fistulous tracts can be particularly helpful in surgical planning.
Neoplasms
Primary tumors
Small bowel neoplasms are rare, representing <25% of all gastrointestinal neoplasms. Adenocarcinoma, carcinoid tumor, lymphoma, and gastrointestinal stromal tumor (GIST) are the most common tumors of the small bowel and may be distinguished by location and imaging characteristics. Most small bowel tumors tend to be small and present with nonspecific symptoms, such as abdominal pain or bleeding. For detecting and evaluating small bowel endoluminal masses, the distension with neutral or negative contrast agents is key. Thin collimation and coronal or sagittal reformations can help resolve uncertain findings and bring additional findings to light.
Adenocarcinoma, the most common primary malignancy of the small bowel, usually appears as asymmetric focal wall thickening or as an enhancing intraluminal mass. The duodenum is the most common site, followed by the jejunum. Associated luminal narrowing can lead to obstruction. Local infiltration of the mesenteric fat can be seen. Less commonly, an "apple-core" type lesion can occur. Tumors of the duodenum can invade the adjacent pancreas, obstructing the pancreatic duct, creating an appearance that can be indistinguishable from pancreatic cancer.
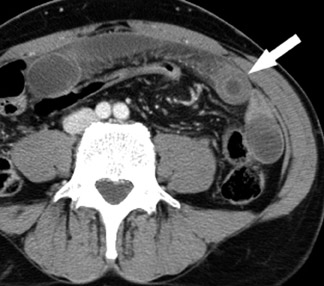
Carcinoid tumors are of neuroendocrine origin and are the second most common small bowel malignancy. Unlike adenocarcinoma, these more commonly affect the distal small bowel. The primary tumors are often very small and are slow-growing, appearing as small hypervascular areas of nodular bowel wall thickening or submucosal masses. In approximately 30% of cases, multiple sites are demonstrated. 25 A characteristic desmoplastic response can occur, with soft tissue infiltration and retraction of the adjacent mesentery. Calcifications are frequently present. Bulky lymphadenopathy can occur, with central low attenuation signifying necrosis. Pulmonary, hepatic, and osteoblastic skeletal metastases are common.
Small bowel lymphoma can occur either as a primary disease or secondary to nodal spread of extraintestinal lymphoma. Four major patterns of small bowel lymphoma have been described: 1) a focal intramural or intraluminal mass; 2) multiple nodules; 3) diffuse involvement of a small bowel segment with wall thickening and aneurysmal dilatation of the lumen; and, 4) rarely, an exophytic mass with ulceration. 25 Lymphoma is a soft tumor, rarely leading to obstruction even with marked luminal narrowing. Focal masses may act as lead points in intussusception. Bulky lymphomatous masses in the mesentery may encase mesenteric vessels, producing the characteristic "sandwich" sign.
Gastrointestinal stromal tumors, formerly known as leiomyomas or leiomyosarcomas, are mesenchymal tumors that arise from the smooth muscle cells within the small bowel wall. They may be submucosal, subserosal, or intraluminal, occurring as solitary or multiple lesions. While GISTs can be benign or malignant, imaging features do not easily distinguish their behavior unless obvious metastatic disease is present. Submucosal GISTs often appear as smoothly marginated, round filling defects, but the more common subserosal GISTs are typically large, bulky, exophytic masses with ulceration that displace adjacent structures (Figure 12).
Metastases
Metastatic disease may spread to the small bowel via intraperitoneal seeding, hematogenous dissemination, lymphatic channels, or direct extension. The most common malignancies that seed the small bowel intraperitoneally are gastrointestinal (GI) primaries, and, in women, ovarian or uterine carcinoma. Hematogenous spread to the small bowel usually manifests as multiple small nodules along the mesenteric border at the insertion of the vasa recta. Melanoma, lung, and breast are the most common primary cancers (Figure 13). 26
Trauma and perforation
Contusion, intramural hematoma, or small bowel perforation may result from blunt abdominal trauma. As MDCT use becomes routine for assessing trauma patients, these injuries are being recognized with increasing frequency. Findings on CT are typically subtle and include focal bowel wall thickening and adjacent mesenteric stranding. The attenuation of any interloop fluid should be measured. Water-attenuation fluid suggests bowel perforation, whereas high attenuation (>30 HU) suggests hemorrhage. Penetrating injuries to the small bowel typically produce extraluminal air. Hypotension and subsequent resuscitation may give rise to a "shock bowel" pattern of dramatic mucosal hyperenhancement, as described above.
Foreign body ingestion is an uncommon but fascinating cause of small bowel perforation. Most foreign bodies are thought to pass unimpeded through the intestine, but sharp bodies (such as toothpicks, or small fish or chicken bones) may lead to perforation. Clinical manifestations vary from pain, nausea, and vomiting to fever and peritonitis with local abscess formation. CT diagnosis may be extremely difficult because the object ingested is typically unknown. Bowel wall thickening, adjacent fat stranding, and localized extraluminal gas are common-but nonspecific-findings. In a recent series described by Coulier et al, 27 direct visualization of the calcified foreign body was the most definitive sign and was greatly facilitated by thin, overlapping sections and multiplanar reformations, which allowed the shape of the object to be recognized. 27
Conclusion
Multidetector CT facilitates the evaluation of a variety of small bowel processes. Water is preferred for luminal distention in conjunction with intravenous contrast administration. For the highest-quality multiplanar reformations, imaging should be performed with narrow detector collimation and overlapping reconstruction. Imaging in both the arterial and venous phases of enhancement increases total radiation dose but may be helpful for the assessment of vascular pathology. Coronal images may be particularly helpful in assessing small bowel obstruction and inflammatory bowel disease.
