Essentials of scrotal ultrasound: A review of frequently encountered abnormalities
Images









Dr. Hebert is a Radiology Resident at the University of North Carolina, Durham, NC, and Dr. Chong is Chief of Diagnostic Ultrasound and a Professor of Radiology at the University of North Carolina, Chapel Hill, NC. Dr. Deurdulian is Chief of Imaging Services, W. G. Hefner VA Medical Center, Salisbury, NC.
Ultrasound is the primary modality for imaging palpable testicular lesions in the setting of acute traumatic injury/pain. Many scrotal ultrasounds are performed every year for these common conditions. Accurate interpretation of the findings is essential to guiding treatment and further intervention because a wide spectrum of pathologies can produce symptoms.
Technique
The scrotal ultrasound exam is performed with the patient in the supine position and a rolled towel or sheet placed between the legs to support the scrotum. The penis is positioned superiorly or superolaterally and draped with a towel. Scanning is usually performed with an 8-MHz to15-MHz transducer with sequential sagittal and transverse images. Higher-frequency transducers allow for greater resolution of the scrotal contents, but lower frequency transducers can be employed for an edematous scrotum. Transverse side-by-side images of both testes are obtained to compare flow symmetry, echogenicity, and scrotal wall thickness. Color and power Doppler ultrasound are also used to detect perfusion and verify abnormal flow patterns.
Normal anatomy
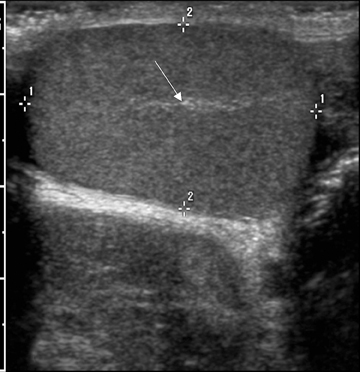
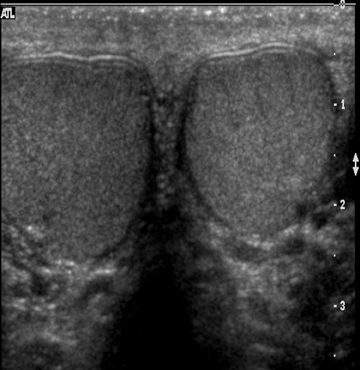
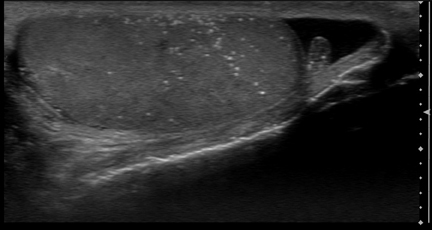
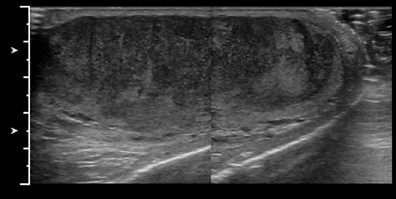
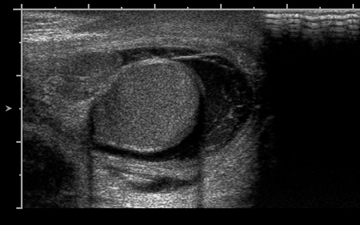
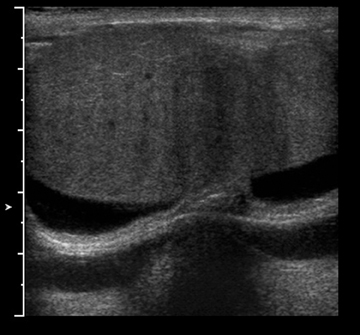
The normal testicle measures between 3 cm and 5 cm in length, 2 cm and 4 cm in width, and approximately 3 cm in AP dimension. The tunica vaginalis consists of visceral and parietal layers. The parietal layer lies against the scrotal wall; the visceral layer envelops all but the posterior portion of the testicle.1 The tunica albuginea surrounds the testicle; however, this is not normally visible under ultrasound unless fluid surrounds the testicle (Figure 1). The epididymis is an elongated, crescent-shaped structure that measures approximately 6-cm to 7-cm long and is usually isoechoic or hypoechoic relative to the testicle. The epididymis is usually located along the superior portion of the testicle, and the tail extends inferolaterally, eventually continuing as the vas deferens (Figure 2). The rete testis is formed by the convergence of seminiferous tubules within the mediastinum of the testicle.2 The paired testicular arteries, arising from the abdominal aorta, are the main blood supply to the testes. They enter the scrotum through the inguinal canal. There is collateral circulation from the deferential artery (a branch of the inferior vesical artery, which arises from the internaliliac) and the cremasteric artery (a branch of the inferior epigastric artery). The testicular arteries, as well as the pampiniform plexus, nerves, and lymphatics, converge within the spermatic cord and course toward the tunica albuginea. Once the testicular artery enters the tunica albuginea, it branches into capsular arteries and eventually into recurrent rami branches that course centrifugally into the mediastinum. The remaining portions of the scrotum receive arterial blood from the pudendal arteries, which arise from the internal iliac artery. A transmediastinal artery persists in ≤50% of patients and will appear as a prominent hypoechoic band within the testicle, with blood flow in the opposite direction of the recurrent rami arteries with Doppler imaging.3
The anatomic relationships of the vasculature to the spermatic cord are important because of several common conditions that can be related to changes in the normal relationships. For example, the bell-and-clapper deformity is a high insertion of the tunica vaginalis onto the spermatic cord, which leaves the testes free to rotate within the tunica and can predispose to intravaginal testicular torsion. Another anatomic correlate is that testicular varicoceles occur more commonly on the left side, likely because the left testicular vein drains via the left renal vein into the inferior vena cava (IVC), rather than directly into the IVC, as with the right testicular vein.3
Benign conditions
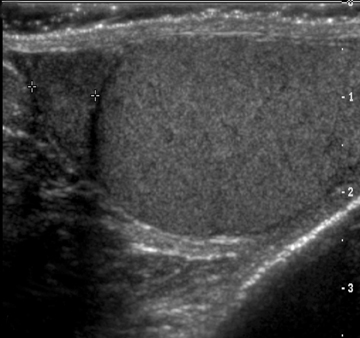
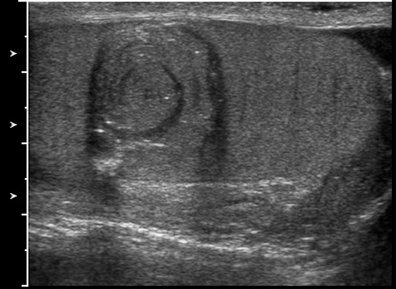
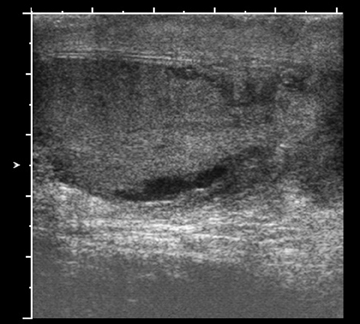
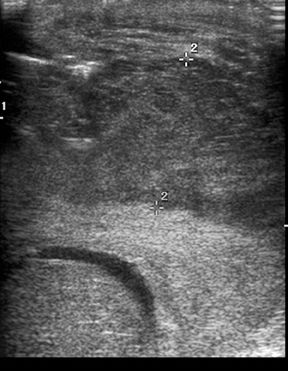
Ectasia of the rete testis is an idiopathic, benign condition that may be associated with partial or complete obstruction of the efferent ductules, leading to cystic dilatation. This condition occurs most frequently in men older than 55 and is frequently bilateral, although unilateral occurrences are also seen.4 The characteristic finding of this lesion is a peripheral, elongated structure composed of multiple small, cystic ortubular structures that replace the mediastinum without causing significant mass effect.5 There are no calcifications, solid components, or flow on Doppler imaging (Figure 3).6
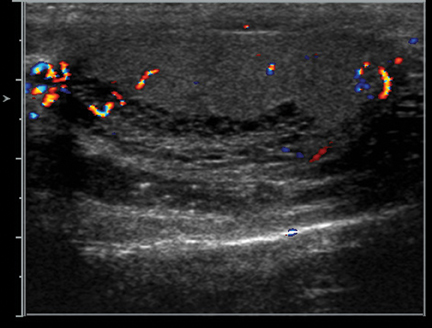
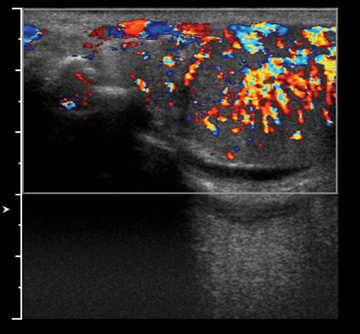
Varicoceles may be extratesticular or intratesticular. Of the extratesticular varieties, the left-side ones are more frequently involved because their venous drainage is indirect: The left testicular vein drains into the left renal vein, while the right testicular vein drains directly into the IVC. Vein diameter >3 mm and/or >1 mm of reflux during Valsalva are diagnostic of a varicocele (Figure 4).7 Reported infertility rates with varices are as high as 33%, although the size of varices does not correlate with likelihood of infertility.3
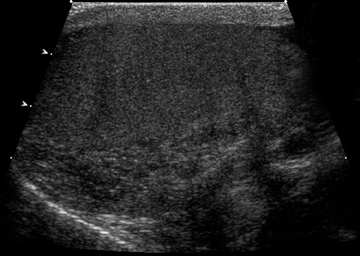
Intratesticular simple cysts are common and have the usual ultrasound characteristics of benign cysts. Cysts of the tunica are common after trauma and frequently are palpable by the patient. They may show calcification on ultrasound. Epidermoid cysts are also benign and account for1% to 2% of all resected testicular masses.8 These lesions show alternating rings of hyperechogenicity and hypoechogenicity on ultrasound,leading to the characteristic “onion ring” sign (Figure 5).5 Typically, epidermoid cysts show no flow on Doppler imaging, helping to differentiate them from the majority of other solid intratesticular masses.8
Tunical fibromas are benign, painless tumors that arise most frequently from the tunica vaginalis, but they can arise from the tunica albuginea, the spermatic cord, or even the testicular parenchyma in rare cases. On ultrasound, these lesions are usually well-defined hyperechoicmasses measuring 1 cm to 3 cm in diameter centered on the tunica, although they rarely can appear poorly defined and hypoechoic. Posterior acoustic shadowing often signifies a dominant fibrous component.9
Sperm granulomas result from a chronic inflammatory process against extravasated sperm from damage to the tubules, usually in a patient with history of trauma, infection, or surgery. These lesions are frequently solid, well-defined, and hypoechoic extratesticular masses measuring ≤1 cm. They are usually found within the epididymis or along the course of the vas deferens.9
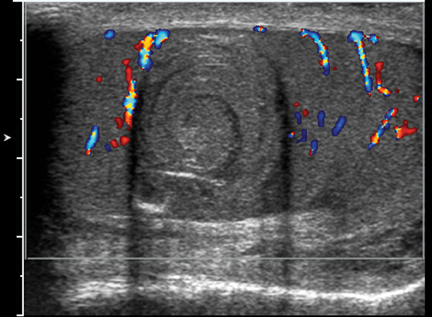
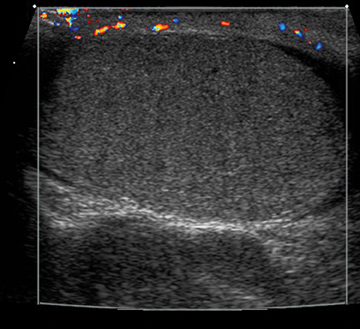
Adenomatoid tumors are the most common epididymal tumors; they comprise about 30% of paratesticular neoplasms. They are frequently well defined and can range from 3 mm to 5 cm in size. The lesion will show an echotexture similar to, or slightly greater than, the testis without increased flow on Doppler imaging (Figure 6).9
Scrotal tuberculosis most commonly presents with an enlarged, hypoechoic epididymis with or without calcifications. Testicular involvement can occur from epididymal extension and result in an enlarged hypoechoic testicle, sometimes with a nodular appearance. The appearance is often nonspecific, but the presence of bilateral epididymal involvement or heterogeneous hypoechoic epididymal enlargement with a testicular lesion favors tuberculosis over other infections.9
Scrotal sarcoid is a rare complication of sarcoidosis, but epididymal and testicular involvement do occur. On ultrasound, single or multiple hypoechoic masses or a solitary echogenic mass involving the epididymis or testis can appear.9
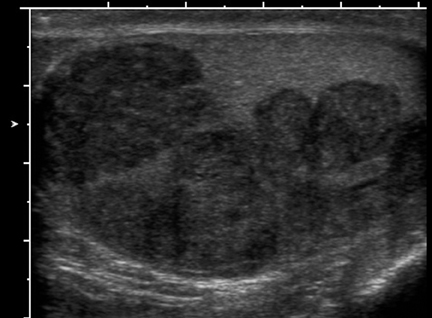
Polyorchidism is rare and can range from testicular duplication alone to duplication of the epididymis or the spermatic cord. These duplications are thought to be due to abnormal division of the embryonic genital ridge. On ultrasound, duplicated testicles can be recognized by their resemblance to a normal testis; they will have a mediastinum testis. The patients have an increased risk of cryptorchidism, inguinal hernia, and testicular malignancy.9
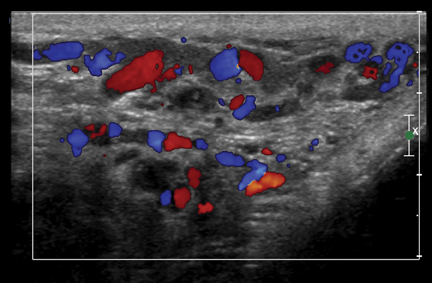
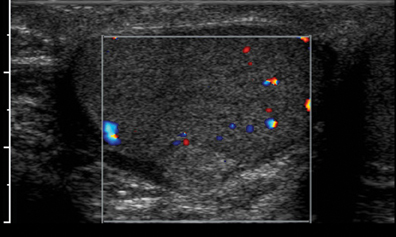
Congenital adrenal hyperplasia (CAH) refers to a group of inherited disorders that cause defects in the adrenal glands’ ability to produce cortisol from cholesterol, leading to increased levels of adrenocorticotropic hormone (ACTH). In patients with CAH, ectopic adrenal rest tissue is frequently identified within the testicles. In response to elevated levels of ACTH, this rest tissue enlarges and can present as a palpable mass.10 On ultrasound, these lesions are typically bilateral, hypoechoic, predominantly located peripherally, and show minimal mass effect (Figure 7).11 Additionally, a spoke-like pattern of vascularity may be present within the mass on color Doppler, but many of the masses will appear hypovascular or avascular.11
Testicular microlithiasis is defined as >5 punctate, nonshadowing, intratesticular calcifications (Figure 8). Although there is an association between microlithiasis and testicular cancer, microlithiasis is very common – the prevalence is 5.6% (14.1% of African Americans).Longitudinal studies have shown that very few patients with microlithiasis will develop testicular cancer.12 Consequently, surveillance ultrasound for patients with microlithiasis is impractical and no longer recommended.
Testicular malignancy
Testicular malignancy is the most common cancer in young men. Ultrasound’s sensitivity to testicular neoplasms is reported to be between 87.5% and 100%, with specificities between 55% and 66.7%.13 The overwhelming majority of primary testicular cancers are germ cell tumors. The commonest germ cell tumors are seminomas, mixed germ cell tumors, and teratomas. The ultrasound appearance of seminomas is usually a solid, hypoechoic mass; however, they can appear heterogeneous with microlithiasis or necrosis (Figure 9).14 Twenty-five percent of patients will have distant spread of the disease via lymphatic or hematogenous routes.14 The primary tumor may outgrow its blood supply and involute in patients with systemic metastases. In this situation, known as the “burnt out” tumor, the primary lesion appears disproportionately small. Ultrasound examination may show a nonspecific hypoechoic or hyperechoic testicular lesion, which may be calcified, in the presence of large, systemic metastases.
Nonseminomatous germ cell tumors are the most common testicular cancer, usually presenting in patients <30 years old. In general, they are more aggressive than seminomas. On ultrasound imaging, they are often heterogeneous, and they can show areas of necrosis, hemorrhage, or calcification.14
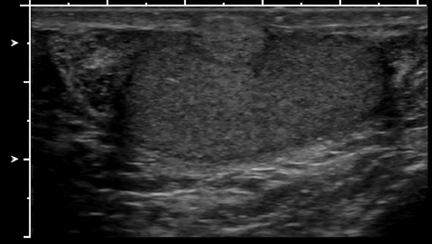
Lymphoma is the most common secondary tumor of the testes. It most commonly occurs in patients >60 years, with non-Hodgkin’slymphoma being the most frequent. It may appear on ultrasound as a diffuse, hypoechoic enlargement of the testis or as an avascular, hypoechoic, and intratesticular mass (Figure 10).14
Leukemic involvement is also common, as the blood-gonad barrier shields the testis from systemic chemotherapy. The ultrasound appearance often shows diffusely enlarged, hypoechoic testicles, frequently indistinguishable from lymphoma.14
Acute scrotal pain
Ultrasound is essential to the evaluation of acute scrotal pain, but the exam must be correlated with clinical findings. Ultrasound is excellent at differentiating surgical causes from medical causes of testicular pain.15
Testicular trauma is usually related to blunt force, with athletic injuries being the most frequent cause. The right side is more likely tobe injured due to trapping of the testicle against the pubis. Ultrasound is used to evaluate the integrity of the tunica and the testicular blood supply. Testicular rupture is defined as disruption of the tunica albuginea and requires surgical exploration. Ultrasound findings include irregular testicular contour and decreased or absent vascularity (Figure 11). Recognition of these findings is important because surgery within 72 hours of the injury results in testicular salvage in approximately 80% of cases.16 Testicular fracture without disruption of the tunica albuginea can be managed conservatively if perfusion is preserved (Figure 12). The integrity of the testicular blood supply can be confirmed with Doppler imaging.2 Either injury can result in hematoceles that eventually transform into post-traumatichydroceles.
Testicular torsion and epididymo-orchitis are the most common causes of nontraumatic testicular pain. Differentiating between them can be clinically difficult. Color Doppler ultrasound is highly sensitive and specific (85%-100%) in torsion. As with rupture,early diagnosis is critical. The rate of successful testicular salvage is between 80% and 100% within 6 hours of symptom onset, butit decreases to 20% after 12 hours.14 On Doppler ultrasound the blood flow within the symptomatic testicle will be decreased or absent(Figure 13). Grayscale appearance of the testicle may be normal at this point. Grayscale abnormalities, such as heterogeneous echo texture, occur late and usually reflect a testicle that is no longer viable.17 Care must be taken to perform the ultrasound with low-pulse repetition frequency and high Doppler gain to effectively demonstrate the slow flow inherent to testicular vessels.14 Because the findings can sometimes be subtle, obtaining side-by-side comparison images of both testicles with grayscale and spectral Doppler imaging to assess for symmetry is very important. The presence of blood flow documented with color Doppler imaging alone cannot completely exclude torsion. In partial torsion or torsion/detorsion, flow can be present while the exam is performed; sometimes, compensatory hyperemia is actually present.3 In partial torsion, flow may be present on color Doppler, but spectral imaging can show high resistance waveforms within the testicular artery.
Torsion of the appendix testis occurs less frequently than testicular torsion. Ultrasound will show a hyperechoic avascular mass adjacent to the testis or epididymis. Blood flow within the testicle will be normal (Figure 14). Torsion of the appendix testis is self-limiting and does not require surgical intervention.3 Segmental testicular infarction is also a rare complication, usually following surgery for primary inguinal hernia repair, although the risk increases up to as much as 5% in patients undergoing repair of recurrent hernia.18
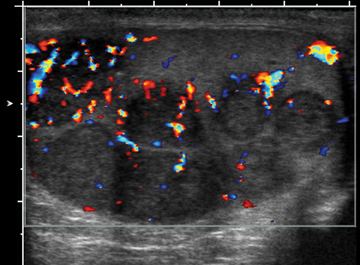
Early epididymo-orchitis will show increased blood flow within both the testicle and the epididymis, often with an enlarged, hypoechoic testicle and epididymis on the symptomatic side (Figure 15).14 Again, obtaining side-by-side comparison images of both testicles is very important for accurate evaluation. Patients presenting in the advanced stages of epididymo-orchitis may show decreased flow due to ischemic changes.Complications from epididymo-orchitis include abscess formation with complex hydrocele or testicular ischemia due to epididymal edema compressing the testicular venous outflow.19 Fournier gangrene is a necrotizing infection that involves the soft tissues of the male genitalia.Findings include a thickened, edematous scrotal wall with gas, seen as hyperechoic linear foci with reverberation artifacts. Gas may be seen on ultrasound prior to the finding of crepitus on clinical examination (Figure 16). Reactive hydroceles can also be present, but the testes and epididymides often appear normal due to separate blood supplies.20 Differentiate Fournier’s from epididymo-orchitis is critical because Fournier’s requires surgical debridement and more aggressive antibiotic therapy.
Conclusion
Ultrasound is the preferred imaging modality for the scrotum because of its low cost, lack of ionizing radiation, and ability to image in real time. The clinical history and examination of patients presenting with scrotal symptoms frequently overlap among multiple etiologies, and a thorough understanding of the usual findings to aid in differentiating surgical from medical, and benign from malignant, etiologies is required to accurately guide patient care.
References
- Garriga V, Serrano A, Marin A, et al. US of the tunica vaginalis testis: Anatomic relationships and pathologic conditions. Radiographics. 2009;29:2017-2032.
- Deurdulian C, Mittelstaedt C, Chong WK, Fielding JR. US of acute scrotal trauma: Optimal technique, imaging findings, and management. Radiographics. 2007;27:357-369.
- Chen P, John S. Ultrasound of the acute scrotum. Applied Radiology. 2006;35:8-17.
- Tartar MV, Trambert MA, Balsara ZN, Mattrey RF. Tubular ectasia of the testicle: Sonographic and MR imaging appearance. AJR Am J Roentgenol. 1993;160:539-542.
- Dogra VS, Gottlieb RH, Rubens DJ, Liao L. Benign intratesticular cystic lesions: US features. Radiographics. 2001;21:S273-S281.
- Kenny D, Batra K, Nancy M, et al. Radiological case: Tubular ectasia of rete testis with spermatocele. Applied Radiology. 2008;37:38-39.
- Dudea SM, Ciurea A, Chiorean A, Botar-Jid C. Doppler applications in testicular and scrotal disease. Med Ultrason. 2010;12:43-51.
- Langer JE, Ramchandani P, Siegelman ES, Banner MP. Epidermoid cysts of the testicle: Sonographic and MR imaging features. AJR Am J Roentgenol. 1999;173:1295-1299.
- Deurdulian C, Chong WK, Mittlestaedt CA, et al. Unusual scrotal masses: Thinking beyond germ cell tumors. Poster.
- Maizlin ZV, Strauss S. Testicular adrenal rest tumors. Isr Med Assoc J. 2005;7:206-207.
- Avila NA, Premkumar A, Merke DP. Testicular adrenal rest tissue in congential adrenal hyperplasia: Comparison of MR imaging and sonographic findings. AJR Am J Roentgenol. 1999;172: 1003-1006.
- Costabile RA. How worrisome is testicular microlithiasis? Curr Opin Urol. 2007;17:419-423.
- Rizvi SA, Ahamd I, Siddiqui MA, et al. Role of color Doppler ultrasonography in evaluation of scrotal swellings: Pattern of disease in 120 patients with review of literature. J Urology. 2011;8:60-65.
- Cokkinos DD, Antypa E, Tserotas P, et al. Emergency ultrasound of the scrotum: A review of the commonest pathologic conditions. Curr Probl Diagn Radiology. 2011;40: 1-14.
- Guichard G, El Ammari J, Del Coro C, et al. Accuracy of ultrasonography in diagnosis of testicular rupture after blunt scrotal trauma. Urology. 2008;71:52-56.
- Bhatt S, Dogra VS. Testicular and scrotal trauma. Radiographics. 2008;28:1617-1629.
- Kaye JD, Shapiro EY, Levitt SB, et al. Parenchymal echotexture predicts testicular salvage after torsion: Potential impact on the need for emergent exploration. J Urology. 2008; 180(Suppl):1733-1736.
- Holloway BJ, Belcher HE, Letourneau JG, Kunberger LE. Scrotal sonography: A valuable tool in the evaluation of complications following inguinal hernia repair. J Clin Ultrasound . 1998;26: 341-44.
- Horstman WG, Middleton WD, Melson GL, Siegel BA. Color Doppler US of the scrotum. Radiographics. 1991;11:941-957.
- Levenson RB, Singh AK, Novelline RA. Fournier gangrene: Role of imaging. Radiographics. 2008;28:519-528.